Yaz (Drospirenone/Ethinyl Estradiol Tablets) Is Available in Blister Packs
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Androgen Excess in Breast Cancer Development: Implications for Prevention and Treatment
26 2 Endocrine-Related G Secreto et al. Androgen excess in breast 26:2 R81–R94 Cancer cancer development REVIEW Androgen excess in breast cancer development: implications for prevention and treatment Giorgio Secreto1, Alessandro Girombelli2 and Vittorio Krogh1 1Epidemiology and Prevention Unit, Fondazione IRCCS – Istituto Nazionale dei Tumori, Milano, Italy 2Anesthesia and Critical Care Medicine, ASST – Grande Ospedale Metropolitano Niguarda, Milano, Italy Correspondence should be addressed to G Secreto: [email protected] Abstract The aim of this review is to highlight the pivotal role of androgen excess in the Key Words development of breast cancer. Available evidence suggests that testosterone f breast cancer controls breast epithelial growth through a balanced interaction between its two f ER-positive active metabolites: cell proliferation is promoted by estradiol while it is inhibited by f ER-negative dihydrotestosterone. A chronic overproduction of testosterone (e.g. ovarian stromal f androgen/estrogen balance hyperplasia) results in an increased estrogen production and cell proliferation that f androgen excess are no longer counterbalanced by dihydrotestosterone. This shift in the androgen/ f testosterone estrogen balance partakes in the genesis of ER-positive tumors. The mammary gland f estradiol is a modified apocrine gland, a fact rarely considered in breast carcinogenesis. When f dihydrotestosterone stimulated by androgens, apocrine cells synthesize epidermal growth factor (EGF) that triggers the ErbB family receptors. These include the EGF receptor and the human epithelial growth factor 2, both well known for stimulating cellular proliferation. As a result, an excessive production of androgens is capable of directly stimulating growth in apocrine and apocrine-like tumors, a subset of ER-negative/AR-positive tumors. -

Ocps) Used in the NM Family Planning Program (FPP
The following slides are intended to familiarize nurses and clinicians with oral contraceptive pills (OCPs) used in the NM Family Planning Program (FPP). The FPP does not intend for this information to supersede the Family Planning Protocol particularly on the requirement for Public Health Nurses in the Public Health Offices to consult a clinician as stated in the Protocol when in doubt or if it is necessary to switch the client’s OCP type. 1 2 Combined oral contraceptives (COCs) contain two hormones; estrogen and progestin. In general, any combined OCP is good for most women who are eligible to take estrogen according to the CDC U.S. Medical Eligibility Criteria (MEC). Once again, refer to the US MEC chart to find out if OCP is a suitable choice for clients with specific health conditions. To learn a little bit about what each hormone does, the FPP is providing the following summary: Estrogen: provides endometrial stability = menstrual cycle control. A higher estrogen dose increases the venous thromboembolism (VTE) or clot risk but OCP clot risk is still less harmful than the clot risk related to pregnancy and giving birth. Progestin: provides most of the contraceptive effect by ‐Preventing luteinizing hormone (LH) surge /ovulation ‐Thickening the cervical mucus to prevent sperm entry. Two major OCP formations are available. Monophasic: There is only one dose of estrogen and progestin in each active pill in the packet; and Multiphasic: There are varying doses of hormones, particularly progestin in the active pills. 3 Section 3 of the FPP Protocol contains the OCP Substitute Table, which groups OCPs into 6 classes according to the estrogen dosage, the type of progestin and the formulations. -

Comparing the Effects of Combined Oral Contraceptives Containing Progestins with Low Androgenic and Antiandrogenic Activities on the Hypothalamic-Pituitary-Gonadal Axis In
JMIR RESEARCH PROTOCOLS Amiri et al Review Comparing the Effects of Combined Oral Contraceptives Containing Progestins With Low Androgenic and Antiandrogenic Activities on the Hypothalamic-Pituitary-Gonadal Axis in Patients With Polycystic Ovary Syndrome: Systematic Review and Meta-Analysis Mina Amiri1,2, PhD, Postdoc; Fahimeh Ramezani Tehrani2, MD; Fatemeh Nahidi3, PhD; Ali Kabir4, MD, MPH, PhD; Fereidoun Azizi5, MD 1Students Research Committee, School of Nursing and Midwifery, Department of Midwifery and Reproductive Health, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 2Reproductive Endocrinology Research Center, Research Institute for Endocrine Sciences, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 3School of Nursing and Midwifery, Department of Midwifery and Reproductive Health, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 4Minimally Invasive Surgery Research Center, Iran University of Medical Sciences, Tehran, Islamic Republic Of Iran 5Endocrine Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran Corresponding Author: Fahimeh Ramezani Tehrani, MD Reproductive Endocrinology Research Center Research Institute for Endocrine Sciences Shahid Beheshti University of Medical Sciences 24 Parvaneh Yaman Street, Velenjak, PO Box 19395-4763 Tehran, 1985717413 Islamic Republic Of Iran Phone: 98 21 22432500 Email: [email protected] Abstract Background: Different products of combined oral contraceptives (COCs) can improve clinical and biochemical findings in patients with polycystic ovary syndrome (PCOS) through suppression of the hypothalamic-pituitary-gonadal (HPG) axis. Objective: This systematic review and meta-analysis aimed to compare the effects of COCs containing progestins with low androgenic and antiandrogenic activities on the HPG axis in patients with PCOS. -
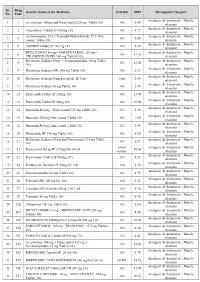
PMBJP Product.Pdf
Sr. Drug Generic Name of the Medicine Unit Size MRP Therapeutic Category No. Code Analgesic & Antipyretic / Muscle 1 1 Aceclofenac 100mg and Paracetamol 325 mg Tablet 10's 10's 8.00 relaxants Analgesic & Antipyretic / Muscle 2 2 Aceclofenac Tablets IP 100mg 10's 10's 4.37 relaxants Acetaminophen 325 + Tramadol Hydrochloride 37.5 film Analgesic & Antipyretic / Muscle 3 4 10's 8.00 coated Tablet 10's relaxants Analgesic & Antipyretic / Muscle 4 5 ASPIRIN Tablets IP 150 mg 14's 14's 2.70 relaxants DICLOFENAC 50 mg+ PARACETAMOL 325 mg+ Analgesic & Antipyretic / Muscle 5 6 10's 11.30 CHLORZOXAZONE 500 mg Tablets 10's relaxants Diclofenac Sodium 50mg + Serratiopeptidase 10mg Tablet Analgesic & Antipyretic / Muscle 6 8 10's 12.00 10's relaxants Analgesic & Antipyretic / Muscle 7 9 Diclofenac Sodium (SR) 100 mg Tablet 10's 10's 6.12 relaxants Analgesic & Antipyretic / Muscle 8 10 Diclofenac Sodium 25mg per ml Inj. IP 3 ml 3 ml 2.00 relaxants Analgesic & Antipyretic / Muscle 9 11 Diclofenac Sodium 50 mg Tablet 10's 10's 2.90 relaxants Analgesic & Antipyretic / Muscle 10 12 Etoricoxilb Tablets IP 120mg 10's 10's 33.00 relaxants Analgesic & Antipyretic / Muscle 11 13 Etoricoxilb Tablets IP 90mg 10's 10's 25.00 relaxants Analgesic & Antipyretic / Muscle 12 14 Ibuprofen 400 mg + Paracetamol 325 mg Tablet 10's 15's 5.50 relaxants Analgesic & Antipyretic / Muscle 13 15 Ibuprofen 200 mg film coated Tablet 10's 10's 1.80 relaxants Analgesic & Antipyretic / Muscle 14 16 Ibuprofen 400 mg film coated Tablet 10's 15's 3.50 relaxants Analgesic & Antipyretic -

Contraception and Misconceptions
CONTRACEPTION AND MISCONCEPTIONS CONTRACEPTION IN WOMEN WITH MENTAL ILLNESS OVERVIEW Hormones and mood How mental illness impacts on contraceptive choice Ideal contraception Pro and cons of contraceptive methods in women with mental illness Hormonal contraception and mood ESTROGENS anti-inflammatory neuroprotective effects of estradiol modulation of the limbic processing memory of emotionally-relevant information. estradiol “beneficially” modulates pathways implicated in the pathophysiology of depression, including serotonin and norepinephrine pathways PROGESTROGENS Previously thought to be anxiogenic, depressogenic Breakdown products Depression – alpha hydroxy progestrogen breakdown products pro – inflammatory, anxiogenic HORMONES AND MOODS – COMPLEX INTERPLAY MENTAL ILLNESS AND CONTRACEPTIVE CHOICE Effect of mental illness on contraceptive choice Effect of contraceptive choice on mental illness Drug interactions EFFECT MENTAL ILLNESS ON CONTRACEPTIVE CHOICE Impulsive Poor planning Cognitive and problem judgement Poor adherence IMPULSIVITY COGNITION POOR JUDGEMENT LETS NOT FORGET SUBSTANCES…… TYPES OF CONTRACEPTION No method Non hormonal Condom/Femidom Diaphragm Non hormone containing IUCD (Copper T) Hormonal COC POP Mirena Evra Patch Nuva Ring Implant ORAL CONTRACEPTIVES POP – avoid Same time every day COC Pros Effective Reduction PMS – monophasic estrogen dominant pill eg Femodene, Yaz, Nordette Cons Drug interactions Pill burden Daily dose EVRA PATCH Weekly patch Transdermal system: 150 mcg/day -

Estradiol (E2), Estriol (E3), Ethinylestradiol (EE2), Testosterone (TEST), Androstenedione (AND), and Progesterone
UNIVERSITY OF CINCINNATI Date: 13-Aug-2010 I, Ruth Marfil Vega , hereby submit this original work as part of the requirements for the degree of: Doctor of Philosophy in Environmental Science It is entitled: Abiotic Transformation of Estrogens in Wastewater Student Signature: Ruth Marfil Vega This work and its defense approved by: Committee Chair: Makram Suidan, PhD Makram Suidan, PhD George Sorial, PhD George Sorial, PhD Margaret Kupferle, PhD, PE Margaret Kupferle, PhD, PE Marc Mills, PhD Marc Mills, PhD 11/8/2010 1,041 Abiotic Transformation of Estrogens in Wastewater A Dissertation submitted to the Graduate School of the University of Cincinnati in partial fulfillment of the requirements for the degree of Doctor of Philosophy In the School of Energy, Environmental, Biological and Medical Engineering By Ruth Marfil-Vega B.S. Chemistry, University of Valladolid, Spain, 2001 Committee Chair: Makram T. Suidan, Ph.D. ABSTRACT The fate of seven steroids: estrone (E1), estradiol (E2), estriol (E3), ethinylestradiol (EE2), testosterone (TEST), androstenedione (AND), and progesterone (PROG), in the presence of synthetic wastewater was studied in order to establish the role abiotic processes play in the elimination of these chemicals from the environment. Comprehension of these mechanisms will foster the optimization of the existing wastewater treatment technologies and the development of sustainable alternatives. Distinctive behavior was encountered for the target compounds in accordance with their chemical structure, hence, different physico-chemical properties and reactivity. Estrogenic compounds, comprising E1, E2, E3 and EE2, were found to undergo a catalytic transformation when contacted with a model vegetable material present in the synthetic wastewater. -

209627Orig1s000
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 209627Orig1s000 MULTI-DISCIPLINE REVIEW Summary Review Office Director Cross Discipline Team Leader Review Clinical Review Non-Clinical Review Statistical Review Clinical Pharmacology Review Reviewers of Multi-Disciplinary Review and Evaluation SECTIONS OFFICE/ AUTHORED/ ACKNOWLEDGED/ DISCIPLINE REVIEWER DIVISION APPROVED Mark Seggel, Ph.D. OPQ/ONDP/DNDP2 Authored: Section 4.2 Digitally signed by Mark R. Seggel -S CMC Lead DN: c=US, o=U.S. Government, ou=HHS, ou=FDA, ou=People, cn=Mark R. Signature: Mark R. Seggel -S Seggel -S, 0.9.2342.19200300.100.1.1=1300071539 Date: 2018.08.08 16:29:15 -04'00' Frederic Moulin, DVM, PhD OND/ODE3/DBRUP Authored: Section 5 Pharmacology/ Digitally signed by Frederic Moulin -S Toxicology DN: c=US, o=U.S. Government, ou=HHS, ou=FDA, ou=People, Reviewer Signature: Frederic Moulin -S 0.9.2342.19200300.100.1.1=2001708658, cn=Frederic Moulin -S Date: 2018.08.08 15:26:57 -04'00' Kimberly Hatfield, PhD OND/ODE3/DBRUP Approved: Section 5 Pharmacology/ Toxicology Digitally signed by Kimberly P. Hatfield -S DN: c=US, o=U.S. Government, ou=HHS, ou=FDA, ou=People, Team Leader Signature: Kimberly P. Hatfield -S 0.9.2342.19200300.100.1.1=1300387215, cn=Kimberly P. Hatfield -S Date: 2018.08.08 14:56:10 -04'00' Li Li, Ph.D. OCP/DCP3 Authored: Sections 6 and 17.3 Clinical Pharmacology Dig ta ly signed by Li Li S DN c=US o=U S Government ou=HHS ou=FDA ou=People Reviewer cn=Li Li S Signature: Li Li -S 0 9 2342 19200300 100 1 1=20005 08577 Date 2018 08 08 15 39 23 04'00' Doanh Tran, Ph.D. -

Desogestrel-Only Pill (Cerazette)
J Fam Plann Reprod Health Care: first published as 10.1783/147118903101197593 on 1 July 2003. Downloaded from Faculty of Family Planning and Reproductive Health Care Clinical Effectiveness Unit A unit funded by the FFPRHC and supported by the University of Aberdeen and SPCERH to provide guidance on evidence-based practice New Product Review (April 2003) Desogestrel-only Pill (Cerazette) Journal of Family Planning and Reproductive Health Care 2003; 29(3): 162–164 Evidence from a randomised trial has shown that a 75 mg (microgrammes) desogestrel pill inhibits ovulation in 97% of cycles. Thus, on theoretical grounds, we would expect the desogestrel pill to be more effective than existing progestogen- only pills (POPs). However, Pearl indices from clinical trials comparing it to a levonorgestrel POP were not significantly different. Therefore an evidence-based recommendation cannot be made that the desogestrel pill is different from other POPs in terms of efficacy, nor that it is similar to combined oral contraception (COC) in this respect. An evidence-based recommendation can be made that the desogestrel-only pill is similar to other POPs in terms of side effects and acceptability. The desogestrel-only pill is not recommended as an alternative to COC in routine practice, but provides a useful alternative for women who require oestrogen-free contraception. In clinical trials: l Ovulation was inhibited in 97% of cycles at 7 and 12 months after initiation. l The Pearl index was 0.41 per 100 woman-years, which was not significantly different from a levonorgestrel-only pill. However, the trial providing these data was too small to detect a clinically important difference. -

Estrogen and Progestin Hormone Doses in Combined Birth Control Pills
Estrogen and Progestin Hormone Doses in Combined Birth Control Pills Estrogen level Pill Brand Name Progestin Dose (mg) ethinyl estradiol (micrograms) 20 mcgm Alesse® levonorgestrel 0.10 Levlite® levonorgestrel 0.10 Loestrin 1/20® Fe norethindrone 1.00 acetate Mircette® desogestrel 0.15 Ortho Evra® norelgestromin 0.15 (patch) (norgestimate metabolite) phasic Estrostep® Fe norethindrone 1.0/1.0/1.0 20/30/35 mcgm acetate 30 mcgm Levlen® levonorgestrel 0.15 Levora® levonorgestrel 0.15 Nordette® levonorgestrel 0.15 Lo/Ovral® norgestrel 0.30 Desogen® desogestrel 0.15 Ortho-Cept® desogestrel 0.15 Loestrin® 1.5/30 norethindrone 1.50 acetate Yasmin® drospirenone 3.0 phasic Triphasil® levonorgestrel 0.05/0.075/0.125 30/40/30 mcgm Tri-Levlen® levonorgestrel 0.05/0.075/0.125 Trivora® levonorgestrel 0.05/0.075/0.125 35 mcgm Ortho-Cyclen® norgestimate 0.25 Ovcon-35® norethindrone 0.40 Brevicon® norethindrone 0.50 Modicon® norethindrone 0.50 Necon® norethindrone 1.00 Norethin® norethindrone 1.00 Norinyl® 1/35 norethindrone 1.00 Ortho-Novum® 1/35 norethindrone 1.00 Demulen® 1/35 ethynodiol diacetate 1.00 Zovia® 1/35E ethynodiol diacetate 1.00 phasic Ortho-Novum® norethindrone 0.50/1.00 35/35 mcgm 10/11 Jenest® norethindrone 0.50/1.00 phasic Ortho-Tri-Cyclen® norgestimate 0.15/0.215/0.25 35/35/35 mcgm Ortho-Novum® norethindrone 0.50/0.75/1.00 7/7/7 Tri-Norinyl® norethindrone 0.50/1.00/0.50 50 mcgm Necon® 1/50 norethindrone 1.00 Norinyl® 1/50 norethindrone 1.00 Ortho-Novum® 1/50 norethindrone 1.00 Ovcon-50® norethindrone 1.00 Ovral® norgestrel 0.50 Demulen® 1/50 ethynodiol diacetate 1.00 Zovia® 1/50E ethynodiol diacetate 1.00 Which pills have higher progestin side efects or cause more acne and hair growth? Each progestin has a diferent potency, milligram per milligram, in terms of progesterone efect to stop menstrual bleeding or androgen efect to stimulate acne and hair growth. -

Loette (Levonorgestrel and Ethinylestradiol) Tablets
AUSTRALIAN PRODUCT INFORMATION - LOETTE (LEVONORGESTREL AND ETHINYLESTRADIOL) TABLETS 1. NAME OF THE MEDICINE Levonorgestrel and Ethinylestradiol 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each pink tablet contains levonorgestrel 100 g and ethinylestradiol 20 g. Each white tablet contains no active ingredients. Excipients with known effect Lactose monohydrate For the full list of excipients, see section 6.1, List of Excipients. 3. PHARMACEUTICAL FORM Tablet, film coated. Pink tablet: Round, pink, biconvex, film coated tablets with "W" debossed on one side and "912" debossed on the other side. White tablet: White film-coated round tablet with convex faces. 4. CLINICAL PARTICULARS 4.1 Therapeutic indications LOETTE is indicated for: The prevention of pregnancy. The treatment of moderate acne vulgaris not controlled with topical preparations in post-menarchal, pre-menopausal women who accept contraception. Version:pfploett11219 Supersedes: pfploett11018 Page 1 of 29 4.2 Dose and method of administration How to Take LOETTE Each package of LOETTE contains 21 active pink tablets and 7 white inactive tablets. To achieve maximum contraceptive effectiveness, LOETTE must be administered as directed and at the same time every day, at intervals not exceeding 24 hours. The recommended dose for the prevention of pregnancy and the treatment of acne is the same. Where poor compliance is a concern, an oral contraceptive with higher levels of estrogens and progestogens should be considered (see section 5.1, Pharmacodynamic Properties, Clinical Trials). How to Start LOETTE No Preceding Hormonal contraceptive Use (in the Past Month) On the first day of the menstrual cycle, i.e. the first day of bleeding, the woman is instructed to take a pink active tablet corresponding to that day of the week from the pink shaded section of the LOETTE pack. -

U.S. Medical Eligibility Criteria for Contraceptive Use, 2016
Morbidity and Mortality Weekly Report Recommendations and Reports / Vol. 65 / No. 3 July 29, 2016 U.S. Medical Eligibility Criteria for Contraceptive Use, 2016 U.S. Department of Health and Human Services Centers for Disease Control and Prevention Recommendations and Reports CONTENTS Introduction ............................................................................................................1 Methods ....................................................................................................................2 How to Use This Document ...............................................................................3 Keeping Guidance Up to Date ..........................................................................5 References ................................................................................................................8 Abbreviations and Acronyms ............................................................................9 Appendix A: Summary of Changes from U.S. Medical Eligibility Criteria for Contraceptive Use, 2010 ...........................................................................10 Appendix B: Classifications for Intrauterine Devices ............................. 18 Appendix C: Classifications for Progestin-Only Contraceptives ........ 35 Appendix D: Classifications for Combined Hormonal Contraceptives .... 55 Appendix E: Classifications for Barrier Methods ..................................... 81 Appendix F: Classifications for Fertility Awareness–Based Methods ..... 88 Appendix G: Lactational -

Recommendations for Contraceptive Use, 2013 Adapted from the World Health Organization Selected Practice Recommendations for Contraceptive Use, 2Nd Edition
Morbidity and Mortality Weekly Report Early Release / Vol. 62 June 14, 2013 U.S. Selected Practice Recommendations for Contraceptive Use, 2013 Adapted from the World Health Organization Selected Practice Recommendations for Contraceptive Use, 2nd Edition Continuing Education Examination available at http://www.cdc.gov/mmwr/cme/conted.html. U.S. Department of Health and Human Services Centers for Disease Control and Prevention Early Release CONTENTS CONTENTS (Continued) Introduction ............................................................................................................1 Appendix A: Summary Chart of U.S. Medical Eligibility Criteria for Methods ....................................................................................................................2 Contraceptive Use, 2010 .................................................................................. 47 How To Use This Document ...............................................................................3 Appendix B: When To Start Using Specific Contraceptive Summary of Changes from WHO SPR ............................................................4 Methods .............................................................................................................. 55 Contraceptive Method Choice .........................................................................4 Appendix C: Examinations and Tests Needed Before Initiation of Maintaining Updated Guidance ......................................................................4 Contraceptive Methods