And Perca Fluviatilis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Do Some Atlantic Bluefin Tuna Skip Spawning?
SCRS/2006/088 Col. Vol. Sci. Pap. ICCAT, 60(4): 1141-1153 (2007) DO SOME ATLANTIC BLUEFIN TUNA SKIP SPAWNING? David H. Secor1 SUMMARY During the spawning season for Atlantic bluefin tuna, some adults occur outside known spawning centers, suggesting either unknown spawning regions, or fundamental errors in our current understanding of bluefin tuna reproductive schedules. Based upon recent scientific perspectives, skipped spawning (delayed maturation and non-annual spawning) is possibly prevalent in moderately long-lived marine species like bluefin tuna. In principle, skipped spawning represents a trade-off between current and future reproduction. By foregoing reproduction, an individual can incur survival and growth benefits that accrue in deferred reproduction. Across a range of species, skipped reproduction was positively correlated with longevity, but for non-sturgeon species, adults spawned at intervals at least once every two years. A range of types of skipped spawning (constant, younger, older, event skipping; and delays in first maturation) was modeled for the western Atlantic bluefin tuna population to test for their effects on the egg-production-per-recruit biological reference point (stipulated at 20% and 40%). With the exception of extreme delays in maturation, skipped spawning had relatively small effect in depressing fishing mortality (F) threshold values. This was particularly true in comparison to scenarios of a juvenile fishery (ages 4-7), which substantially depressed threshold F values. Indeed, recent F estimates for 1990-2002 western Atlantic bluefin tuna stock assessments were in excess of threshold F values when juvenile size classes were exploited. If western bluefin tuna are currently maturing at an older age than is currently assessed (i.e., 10 v. -

Review and Meta-Analysis of the Environmental Biology and Potential Invasiveness of a Poorly-Studied Cyprinid, the Ide Leuciscus Idus
REVIEWS IN FISHERIES SCIENCE & AQUACULTURE https://doi.org/10.1080/23308249.2020.1822280 REVIEW Review and Meta-Analysis of the Environmental Biology and Potential Invasiveness of a Poorly-Studied Cyprinid, the Ide Leuciscus idus Mehis Rohtlaa,b, Lorenzo Vilizzic, Vladimır Kovacd, David Almeidae, Bernice Brewsterf, J. Robert Brittong, Łukasz Głowackic, Michael J. Godardh,i, Ruth Kirkf, Sarah Nienhuisj, Karin H. Olssonh,k, Jan Simonsenl, Michał E. Skora m, Saulius Stakenas_ n, Ali Serhan Tarkanc,o, Nildeniz Topo, Hugo Verreyckenp, Grzegorz ZieRbac, and Gordon H. Coppc,h,q aEstonian Marine Institute, University of Tartu, Tartu, Estonia; bInstitute of Marine Research, Austevoll Research Station, Storebø, Norway; cDepartment of Ecology and Vertebrate Zoology, Faculty of Biology and Environmental Protection, University of Lodz, Łod z, Poland; dDepartment of Ecology, Faculty of Natural Sciences, Comenius University, Bratislava, Slovakia; eDepartment of Basic Medical Sciences, USP-CEU University, Madrid, Spain; fMolecular Parasitology Laboratory, School of Life Sciences, Pharmacy and Chemistry, Kingston University, Kingston-upon-Thames, Surrey, UK; gDepartment of Life and Environmental Sciences, Bournemouth University, Dorset, UK; hCentre for Environment, Fisheries & Aquaculture Science, Lowestoft, Suffolk, UK; iAECOM, Kitchener, Ontario, Canada; jOntario Ministry of Natural Resources and Forestry, Peterborough, Ontario, Canada; kDepartment of Zoology, Tel Aviv University and Inter-University Institute for Marine Sciences in Eilat, Tel Aviv, -

Can Underwater Refuges Protect Fish Populations Against Cormorant
1 Can underwater refuges protect fish populations against cormorant 2 predation? Evidence from a large scale multiple pond experiment 3 Pieter Lemmens1*, Luc De Meester1 and Steven A.J. Declerck2 4 5 1 Laboratory of Aquatic Ecology, Evolution and Conservation, KU Leuven, Ch. Deberiotstraat 6 32, 3000 Leuven, Belgium; 7 2 Department of Aquatic Ecology, Netherlands Institute of Ecology (NIOO-KNAW), 8 Droevendaalsesteeg 10, 6708 PB Wageningen, The Netherlands. 9 10 * Corresponding author: [email protected] 11 12 ABSTRACT 13 Artificial structures can protect fish against predation by cormorants. However, their 14 effectiveness in larger water bodies with different fish communities in the presence of 15 natural vegetation still needs to be explored. Using a large scale field experiment with 16 twenty-four ponds stocked with differently composed fish communities, the present study 17 investigates the extent to which the effect of artificial refuges on fish is species-specific and 18 determined by the characteristics of the fish community. This study provides strong 19 experimental evidence for artificial refuges protecting fish against predation by cormorants, 20 even in the presence of submerged vegetation. The effect of the refuges was, however, 21 highly species-specific and depended on the composition of the fish community. Strong 22 positive effects of refuges on rudd and roach populations were observed, especially in ponds 23 where these species dominated from the start of the experiment. Overall, the total biomass 24 of young-of-the-year, one-year-old and adult rudd and roach was on average 500, 7 and 15 25 times lower in ponds without than in ponds with refuges, respectively. -

Beyond Fish Edna Metabarcoding: Field Replicates Disproportionately Improve the Detection of Stream Associated Vertebrate Specie
bioRxiv preprint doi: https://doi.org/10.1101/2021.03.26.437227; this version posted March 26, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. 1 2 3 Beyond fish eDNA metabarcoding: Field replicates 4 disproportionately improve the detection of stream 5 associated vertebrate species 6 7 8 9 Till-Hendrik Macher1, Robin Schütz1, Jens Arle2, Arne J. Beermann1,3, Jan 10 Koschorreck2, Florian Leese1,3 11 12 13 1 University of Duisburg-Essen, Aquatic Ecosystem Research, Universitätsstr. 5, 45141 Essen, 14 Germany 15 2German Environmental Agency, Wörlitzer Platz 1, 06844 Dessau-Roßlau, Germany 16 3University of Duisburg-Essen, Centre for Water and Environmental Research (ZWU), Universitätsstr. 17 3, 45141 Essen, Germany 18 19 20 21 22 Keywords: birds, biomonitoring, bycatch, conservation, environmental DNA, mammals 23 1 bioRxiv preprint doi: https://doi.org/10.1101/2021.03.26.437227; this version posted March 26, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. 24 Abstract 25 Fast, reliable, and comprehensive biodiversity monitoring data are needed for 26 environmental decision making and management. Recent work on fish environmental 27 DNA (eDNA) metabarcoding shows that aquatic diversity can be captured fast, reliably, 28 and non-invasively at moderate costs. -

Changing Communities of Baltic Coastal Fish Executive Summary: Assessment of Coastal fi Sh in the Baltic Sea
Baltic Sea Environment Proceedings No. 103 B Changing Communities of Baltic Coastal Fish Executive summary: Assessment of coastal fi sh in the Baltic Sea Helsinki Commission Baltic Marine Environment Protection Commission Baltic Sea Environment Proceedings No. 103 B Changing Communities of Baltic Coastal Fish Executive summary: Assessment of coastal fi sh in the Baltic Sea Helsinki Commission Baltic Marine Environment Protection Commission Editor: Janet Pawlak Authors: Kaj Ådjers (Co-ordination Organ for Baltic Reference Areas) Jan Andersson (Swedish Board of Fisheries) Magnus Appelberg (Swedish Board of Fisheries) Redik Eschbaum (Estonian Marine Institute) Ronald Fricke (State Museum of Natural History, Stuttgart, Germany) Antti Lappalainen (Finnish Game and Fisheries Research Institute), Atis Minde (Latvian Fish Resources Agency) Henn Ojaveer (Estonian Marine Institute) Wojciech Pelczarski (Sea Fisheries Institute, Poland) Rimantas Repečka (Institute of Ecology, Lithuania). Photographers: Visa Hietalahti p. cover, 7 top, 8 bottom Johnny Jensen p. 3 top, 3 bottom, 4 middle, 4 bottom, 5 top, 8 top, 9 top, 9 bottom Lauri Urho p. 4 top, 5 bottom Juhani Vaittinen p. 7 bottom Markku Varjo / LKA p. 10 top For bibliographic purposes this document should be cited as: HELCOM, 2006 Changing Communities of Baltic Coastal Fish Executive summary: Assessment of coastal fi sh in the Baltic Sea Balt. Sea Environ. Proc. No. 103 B Information included in this publication or extracts thereof is free for citing on the condition that the complete reference of the publication is given as stated above Copyright 2006 by the Baltic Marine Environment Protection Commission - Helsinki Commission - Design and layout: Bitdesign, Vantaa, Finland Printed by: Erweko Painotuote Oy, Finland ISSN 0357-2994 Coastal fi sh – a combination of freshwater and marine species Coastal fish communities are important components of Baltic Sea ecosystems. -
Labidesthes Sicculus
Version 2, 2015 United States Fish and Wildlife Service Lower Great Lakes Fish and Wildlife Conservation Office 1 Atherinidae Atherinidae Sand Smelt Distinguishing Features: — (Atherina boyeri) — Sand Smelt (Non-native) Old World Silversides Old World Silversides Old World (Atherina boyeri) Two widely separated dorsal fins Eye wider than Silver color snout length 39-49 lateral line scales 2 anal spines, 13-15.5 rays Rainbow Smelt (Non -Native) (Osmerus mordax) No dorsal spines Pale green dorsally Single dorsal with adipose fin Coloring: Silver Elongated, pointed snout No anal spines Size: Length: up to 145mm SL Pink/purple/blue iridescence on sides Distinguishing Features: Dorsal spines (total): 7-10 Brook Silverside (Native) 1 spine, 10-11 rays Dorsal soft rays (total): 8-16 (Labidesthes sicculus) 4 spines Anal spines: 2 Anal soft rays: 13-15.5 Eye diameter wider than snout length Habitat: Pelagic in lakes, slow or still waters Similar Species: Rainbow Smelt (Osmerus mordax), 75-80 lateral line scales Brook Silverside (Labidesthes sicculus) Elongated anal fin Images are not to scale 2 3 Centrarchidae Centrarchidae Redear Sunfish Distinguishing Features: (Lepomis microlophus) Redear Sunfish (Non-native) — — Sunfishes (Lepomis microlophus) Sunfishes Red on opercular flap No iridescent lines on cheek Long, pointed pectoral fins Bluegill (Native) Dark blotch at base (Lepomis macrochirus) of dorsal fin No red on opercular flap Coloring: Brownish-green to gray Blue-purple iridescence on cheek Bright red outer margin on opercular flap -

Transcriptome Sequencing and Analysis of Wild Amur Ide (Leuciscus Waleckii) Inhabiting an Extreme Alkaline-Saline Lake Reveals Insights Into Stress Adaptation
Transcriptome Sequencing and Analysis of Wild Amur Ide (Leuciscus waleckii) Inhabiting an Extreme Alkaline- Saline Lake Reveals Insights into Stress Adaptation Jian Xu1, Peifeng Ji1, Baosen Wang1,2, Lan Zhao1, Jian Wang1, Zixia Zhao1, Yan Zhang1, Jiongtang Li1, Peng Xu1*, Xiaowen Sun1* 1 Centre for Applied Aquatic Genomics, Chinese Academy of Fishery Sciences, Beijing, China, 2 College of Life Sciences, Tianjin Normal University, Tianjin, China Abstract Background: Amur ide (Leuciscus waleckii) is an economically and ecologically important species in Northern Asia. The Dali Nor population inhabiting Dali Nor Lake, a typical saline-alkaline lake in Inner Mongolia, is well-known for its adaptation to extremely high alkalinity. Genome information is needed for conservation and aquaculture purposes, as well as to gain further understanding into the genetics of stress tolerance. The objective of the study is to sequence the transcriptome and obtain a well-assembled transcriptome of Amur ide. Results: The transcriptome of Amur ide was sequenced using the Illumina platform and assembled into 53,632 cDNA contigs, with an average length of 647 bp and a N50 length of 1,094 bp. A total of 19,338 unique proteins were identified, and gene ontology and KEGG (Kyoto Encyclopedia of Genes and Genomes) analyses classified all contigs into functional categories. Open Reading Frames (ORFs) were detected from 34,888 (65.1%) of contigs with an average length of 577 bp, while 9,638 full-length cDNAs were identified. Comparative analyses revealed that 31,790 (59.3%) contigs have a significant similarity to zebrafish proteins, and 27,096 (50.5%), 27,524 (51.3%) and 27,996 (52.2%) to teraodon, medaka and three- spined stickleback proteins, respectively. -

Beyond Fish Edna Metabarcoding: Field Replicates Disproportionately Improve the Detection of Stream Associated Vertebrate Species
Metabarcoding and Metagenomics 5: 59–71 DOI 10.3897/mbmg.5.66557 Research Article Beyond fish eDNA metabarcoding: Field replicates disproportionately improve the detection of stream associated vertebrate species Till-Hendrik Macher1, Robin Schütz1, Jens Arle2, Arne J. Beermann1,3, Jan Koschorreck2, Florian Leese1,3 1 University of Duisburg-Essen, Aquatic Ecosystem Research, Universitätsstr. 5, 45141 Essen, Germany 2 German Environment Agency, Wörlitzer Platz 1, 06844 Dessau-Roßlau, Germany 3 University of Duisburg-Essen, Centre for Water and Environmental Research (ZWU), Universitätsstr. 3, 45141 Essen, Germany Corresponding author: Till-Hendrik Macher ([email protected]) Academic editor: Pieter Boets | Received 26 March 2021 | Accepted 10 June 2021 | Published 13 July 2021 Abstract Fast, reliable, and comprehensive biodiversity monitoring data are needed for environmental decision making and management. Recent work on fish environmental DNA (eDNA) metabarcoding shows that aquatic diversity can be captured fast, reliably, and non-invasively at moderate costs. Because water in a catchment flows to the lowest point in the landscape, often a stream, it can col- lect traces of terrestrial species via surface or subsurface runoff along its way or when specimens come into direct contact with water (e.g., when drinking). Thus, fish eDNA metabarcoding data can provide information on fish but also on other vertebrate species that live in riparian habitats. This additional data may offer a much more comprehensive approach for assessing vertebrate diversity at no additional costs. Studies on how the sampling strategy affects species detection especially of stream-associated communities, however, are scarce. We therefore performed an analysis on the effects of biological replication on both fish as well as (semi-)terrestrial species detection. -

Identification of Priority Areas for the Conservation of Stream Fish Assemblages: Implications for River Management in France A
Identification of Priority Areas for the Conservation of Stream Fish Assemblages: Implications for River Management in France A. Maire, P. Laffaille, J.F. Maire, L. Buisson To cite this version: A. Maire, P. Laffaille, J.F. Maire, L. Buisson. Identification of Priority Areas for the Conservation of Stream Fish Assemblages: Implications for River Management in France. River Research and Applications, Wiley, 2016, 33 (4), pp.524-537. 10.1002/rra.3107. hal-01426354 HAL Id: hal-01426354 https://hal.archives-ouvertes.fr/hal-01426354 Submitted on 2 Jul 2021 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Distributed under a Creative Commons Attribution| 4.0 International License IDENTIFICATION OF PRIORITY AREAS FOR THE CONSERVATION OF STREAM FISH ASSEMBLAGES: IMPLICATIONS FOR RIVER MANAGEMENT IN FRANCE A. MAIREa*,†, P. LAFFAILLEb,c, J.-F. MAIREd AND L. BUISSONb,e a Irstea; UR HYAX, Pôle Onema-Irstea Hydroécologie des plans d’eau; Centre d’Aix-en-Provence, Aix-en-Provence, France b CNRS; UMR 5245 EcoLab, (Laboratoire Ecologie Fonctionnelle et Environnement), Toulouse, France c Université de Toulouse, INP, UPS; EcoLab; ENSAT, Castanet Tolosan, France d ONERA, The French Aerospace Lab Composites Department, Châtillon, France e Université de Toulouse, INP, UPS; EcoLab, Toulouse, France ABSTRACT Financial and human resources allocated to biodiversity conservation are often limited, making it impossible to protect all natural places, and priority areas for protection must be identified. -
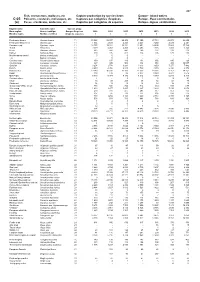
Fish, Crustaceans, Molluscs, Etc Capture Production by Species Items Europe
467 Fish, crustaceans, molluscs, etc Capture production by species items Europe - Inland waters C-05 Poissons, crustacés, mollusques, etc Captures par catégories d'espèces Europe - Eaux continentales (a) Peces, crustáceos, moluscos, etc Capturas por categorías de especies Europa - Aguas continentales English name Scientific name Species group Nom anglais Nom scientifique Groupe d'espèces 2009 2010 2011 2012 2013 2014 2015 Nombre inglés Nombre científico Grupo de especies t t t t t t t Freshwater bream Abramis brama 11 33 823 35 937 29 408 31 390 27 734 28 978 28 655 Freshwater breams nei Abramis spp 11 1 934 2 029 2 223 2 243 3 250 2 439 2 241 Common carp Cyprinus carpio 11 12 755 13 381 13 351 13 951 14 695 17 883 17 729 Tench Tinca tinca 11 3 019 4 053 2 455 2 253 1 686 1 491 1 328 Bleak Alburnus alburnus 11 682 547 441 269 231 213 219 Barbel Barbus barbus 11 300 195 412 224 205 170 188 Mediterranean barbel Barbus meridionalis 11 0 0 0 - - - - ...A Barbus cyclolepis 11 ... 0 0 - - - - Common nase Chondrostoma nasus 11 159 137 189 134 156 195 123 Crucian carp Carassius carassius 11 367 345 506 332 351 305 19 057 Goldfish Carassius auratus 11 3 254 2 778 3 293 3 613 3 653 7 277 7 482 Roach Rutilus rutilus 11 4 259 4 956 4 915 3 789 3 670 6 328 6 378 Roaches nei Rutilus spp 11 13 243 16 331 17 160 17 553 18 361 17 757 14 151 Rudd Scardinius erythrophthalmus 11 108 139 96 8 077 9 380 8 977 7 516 Orfe(=Ide) Leuciscus idus 11 5 040 5 374 5 330 4 812 5 979 6 240 5 316 Common dace Leuciscus leuciscus 11 4 .. -

Applied Freshwater Fish Biology an Introduction to Methods of Research and Management
How to preserve and how to exploit natural populations to be sustained for the future. Plain questions without equally plain answers Applied freshwater fish biology An introduction to methods of research and management Arne N. Linløkken, ass. professor Inland Norway University of Applied Sciences Arne N. 1 CONTENT INTRODUCTION .................................................................................................................................................. 3 Prehistory and evolution ......................................................................................................................................... 3 Short on construction and function ......................................................................................................................... 4 Morphology ........................................................................................................................................................ 4 Anatomy and physiology .................................................................................................................................... 5 European freshwater fish species ............................................................................................................................ 6 Immigration and distribution of freshwater fish in western Scandinavia ........................................................... 7 Western immigrants ........................................................................................................................ 8 -

Monthly Highlights | No.7/2018
M onthly Highlights No. 7 / 2018 In this issue Contents HighlightsIn May 2018, first−sales value and volume increased in Denmark, Estonia, Norway and Sweden over May 2017. First sales in Europe No.In 4,the 2014 same period, they dropped in Belgium, Latvia, European perch and pike-perch in Lithuania, Poland, and the UK. Denmark, Estonia, and Poland Over the past three years, average prices of European Extra-EU imports perch were the highest in Denmark (2,50 EUR/kg), Weekly average EU import prices for followed by Estonia (2,28 EUR/kg) and Poland selected products from selected (1,94 EUR/kg). Average price of pike-perch in Denmark countries of origin (8,33 EUR/kg) was 106% higher than that in Estonia (4,05 EUR/kg), and 99% higher than in Poland Consumption (4,18 EUR/kg). Gilthead seabream in France, Italy, Extra-EU import prices of Ecuadorian tropical shrimp Portugal, and Spain continued to fall amid a global supply glut. Frozen tilapia from China continued a long-run rise in price. Case studies In January–April 2018, the average retail price of fresh Atlantic mackerel in the EU gilthead seabream for household consumption was the Fisheries and aquaculture in highest in France (11,39 EUR/kg) and the lowest in Madagascar Portugal (6,41 EUR/kg). Global Highlights EU consumption of mackerel was 1,07 kg per capita in 2016, which places mackerel in seventh place on the list Macroeconomic context of top fish species consumed in the EU. Marine fuel, consumer prices, exchange rates In 2017, Malagasy exports to the EU reached EUR 142 million and 21.318 tonnes, mostly frozen shrimp and canned tuna.