Get Consistent and Reliable Results
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Bile Esculin Agar
BILE ESCULIN AGAR INTENDED USE Remel Bile Esculin Agar is a solid medium recommended for use in qualitative procedures for the presumptive identification of group D streptococci and enterococci. SUMMARY AND EXPLANATION Rochaix first demonstrated the value of esculin hydrolysis for identification of enterococci.1 Meyer et al. found that 61 of 62 strains of enterococci hydrolyzed esculin in a medium containing bile.2 Swan determined that positive results obtained on esculin agar containing 40% bile correlated well with serologically confirmed group D streptococci.3 Using Swan’s formula, Facklam and Moody tested over 700 strains of streptococci and enterococci representing all known serological groups and found all strains to be bile-resistant and esculin-positive. PRINCIPLE Group D streptococci and enterococci hydrolyze esculin in the presence of bile to form esculetin and dextrose. Esculetin reacts with ferric ions supplied by ferric ammonium citrate to form brown-black colonies on Bile Esculin Agar. Oxgall in a concentration of 4% (equivalent to 40% bile) inhibits most strains of streptococci and enterococci other than group D. REAGENTS (CLASSICAL FORMULA)* Oxgall (40% Bile) ............................................................ 40.0 g Esculin...............................................................................1.0 g Gelatin Peptone ................................................................ 5.0 g Ferric Ammonium Citrate...................................................0.5 g Beef Extract....................................................................... 3.0 g Agar.................................................................................15.0 g Demineralized Water...................................................1000.0 ml pH 6.8 ± 0.2 @ 25°C *Adjusted as required to meet performance standards. PRECAUTIONS This product is For Laboratory Use only. It is not intended for use in the diagnosis of disease or other conditions. PREPARATION OF DEHYDRATED CULTURE MEDIUM 1. Suspend 64 g of medium in 1000 ml of demineralized water. -

Evaluating Historical Paradigms of Sterility in Perinatal Microbiology and Ramifications Orf Pregnancy Outcomes
Wayne State University Wayne State University Dissertations January 2020 Evaluating Historical Paradigms Of Sterility In Perinatal Microbiology And Ramifications orF Pregnancy Outcomes Jonathan Greenberg Wayne State University Follow this and additional works at: https://digitalcommons.wayne.edu/oa_dissertations Part of the Microbiology Commons, and the Obstetrics and Gynecology Commons Recommended Citation Greenberg, Jonathan, "Evaluating Historical Paradigms Of Sterility In Perinatal Microbiology And Ramifications orF Pregnancy Outcomes" (2020). Wayne State University Dissertations. 2468. https://digitalcommons.wayne.edu/oa_dissertations/2468 This Open Access Dissertation is brought to you for free and open access by DigitalCommons@WayneState. It has been accepted for inclusion in Wayne State University Dissertations by an authorized administrator of DigitalCommons@WayneState. EVALUATING HISTORICAL PARADIGMS OF STERILITY IN PERINATAL MICROBIOLOGY AND RAMIFICATIONS FOR PREGNANCY OUTCOMES by JONATHAN MURRAY GREENBERG DISSERTATION Submitted to the Graduate School of Wayne State University, Detroit, Michigan in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY 2020 MAJOR: BIOCHEMISTRY, MICROBIOLOGY, & IMMUNOLOGY Approved by: Advisor Date DEDICATION This dissertation is dedicated to my parents and sister, who have all been immeasurably supportive over my graduate career and more importantly my entire life. I could not have done this without you. ii ACKNOWLEDGMENTS There is no way for me to express fully my sincere gratitude to all the individuals who have had an impact on my life and throughout my time here at Wayne State. None of it would have been possible without Dr. Kevin Theis. Rotating in his lab was one of the best decisions of my life and quite literally got me to where I am. -

Bile Esculin Agar Intended Use Bile Esculin Agar Is A
Bile Esculin Agar Intended Use Bile Esculin Agar is a differential medium used for isolation and presumptive identification of group D Streptococci / Enterococci from food samples. Summary Swan formulated Bile Esculin Agar for the isolation and identification of group D Streptococci from foods. Originally, Bile Esculin Test was used for the identification of Enterococci. However, since group D Streptococci share the test with Enterococci, it is advisable that other tests such as salt tolerance be performed while identifying Enterococci. Meyer and Schonfeld showed that majority of Enterococci were able to grow in esculin and split it, while other Streptococci could not. This medium is used to differentiate Enterococci and Streptococcus bovis from other Streptococci. Principle Oxgall inhibits gram-positive bacteria other than group D Streptococci / Enterococci. Ferric citrate is an indicator of esculin hydrolysis and resulting esculetin formation. Enterococci / group D Streptococci hydrolyze the glycoside esculin to esculetin and dextrose. Esculetin reacts with ferric citrate producing brownish black complex. This medium is also shown to aid differentiation of genus Klebsiella-Enterobacter-Serratia from other Enterobacteriaceae on the basis of esculin hydrolysis. Formula* Ingredients g/L Pancreatic Digest of Gelatin 5.0 Beef Extract 3.0 Oxgall 40.0 Ferric Citrate 0.5 Esculin 1.0 Agar 15.0 Final pH (at 25°C) 6.6 ± 0.2 *Adjusted to suit performance parameters Storage and Stability Store dehydrated medium below 30°C in tightly closed container and the prepared medium at 2ºC-8°C. Avoid freezing and overheating. Use before expiry date on the label. Once opened keep powdered medium closed to avoid hydration. -

Bile Esculin Azide Agar (ISO 7899-2:2000) (Dehydrated Culture Media) for Microbiology
PRODUCT CODE: 415523 Bile Esculin Azide Agar (ISO 7899-2:2000) (Dehydrated Culture Media) for microbiology Preparation Suspend 56.6 grams of the medium in one litre of distilled water. Mix well and dissolve by heating with frequent agitation. Boil for one minute until complete dissolution. Dispense into appropriate containers and sterilize in autoclave at 121ºC for 15 minutes. Overheating can cause darkening of the medium. If tubes are used, allow cooling in a slanted position. The prepared medium should be stored at 8-15°C. The colour is tournasol. The dehydrated medium should be homogeneous, free-flowing and toasted in colour. If there are any physical changes, discard the medium. Caution: This medium is toxic if swallowed, inhaled or comes into contact with skin. Wear gloves and eye/face protection. Uses BILE ESCULIN AZIDE AGAR is a modification of Bile Esculin Agar with the addition of sodium azide as an inhibitor and with the reduction of the bile concentration. The resulting medium is more selective but still provides rapid growth and efficient recovery of enterococci. The ability to hydrolyse esculin in the presence of bile is a characteristic of enterococci. Organisms positive for esculin hydrolysis hydrolyse the glycoside esculin to esculetin and dextrose. The esculetin reacts with the Ferric citrate to form a dark brown or black colony. Ox bile does not inhibit enterococci while other Gram-positive bacteria are inhibited. Sodium azide inhibits Gram negative bacteria. Tryptone, Peptone and Yeast extract supply the nutrients essential for growth. Sodium chloride provides the osmotic balance. Bacteriological agar is the solidifying agent. -

Prepared Culture Media
PREPARED CULTURE MEDIA 121517SS PREPARED CULTURE MEDIA Made in the USA AnaeroGRO™ DuoPak A 02 Bovine Blood Agar, 5%, with Esculin 13 AnaeroGRO™ DuoPak B 02 Bovine Blood Agar, 5%, with Esculin/ AnaeroGRO™ BBE Agar 03 MacConkey Biplate 13 AnaeroGRO™ BBE/PEA 03 Bovine Selective Strep Agar 13 AnaeroGRO™ Brucella Agar 03 Brucella Agar with 5% Sheep Blood, Hemin, AnaeroGRO™ Campylobacter and Vitamin K 13 Selective Agar 03 Brucella Broth with 15% Glycerol 13 AnaeroGRO™ CCFA 03 Brucella with H and K/LKV Biplate 14 AnaeroGRO™ Egg Yolk Agar, Modified 03 Buffered Peptone Water 14 AnaeroGRO™ LKV Agar 03 Buffered Peptone Water with 1% AnaeroGRO™ PEA 03 Tween® 20 14 AnaeroGRO™ MultiPak A 04 Buffered NaCl Peptone EP, USP 14 AnaeroGRO™ MultiPak B 04 Butterfield’s Phosphate Buffer 14 AnaeroGRO™ Chopped Meat Broth 05 Campy Cefex Agar, Modified 14 AnaeroGRO™ Chopped Meat Campy CVA Agar 14 Carbohydrate Broth 05 Campy FDA Agar 14 AnaeroGRO™ Chopped Meat Campy, Blood Free, Karmali Agar 14 Glucose Broth 05 Cetrimide Select Agar, USP 14 AnaeroGRO™ Thioglycollate with Hemin and CET/MAC/VJ Triplate 14 Vitamin K (H and K), without Indicator 05 CGB Agar for Cryptococcus 14 Anaerobic PEA 08 Chocolate Agar 15 Baird-Parker Agar 08 Chocolate/Martin Lewis with Barney Miller Medium 08 Lincomycin Biplate 15 BBE Agar 08 CompactDry™ SL 16 BBE Agar/PEA Agar 08 CompactDry™ LS 16 BBE/LKV Biplate 09 CompactDry™ TC 17 BCSA 09 CompactDry™ EC 17 BCYE Agar 09 CompactDry™ YMR 17 BCYE Selective Agar with CAV 09 CompactDry™ ETB 17 BCYE Selective Agar with CCVC 09 CompactDry™ YM 17 BCYE -

APPENDIX a Media and Reagents
APPENDIX A Media and Reagents Pauline K. w. Yu, M.S. The use of appropriate and dependable media is integral to the isolation and identification of microorganisms. Unfortunately, comparative data docu menting the relative efficacy or value of media designed for similar purposes are often lacking. Moreover, one cannot presume identity in composition of a given generic product which is manufactured by several companies because each may supplement the generic products with components, often of a proprietary nature and not specified in the product's labeling. Finally, the actual production of similar products may vary among manufacturers to a sufficient extent to affect their performance. For all of these reasons, therefore, product selection for the laboratory should not be strictly based on cost considerations and should certainly not be based on promotional materials. Evaluations that have been published in the scientific literature should be consulted when available. Alternatively, the prospective buyer should consult a recognized authority in the field. It is seldom necessary for the laboratory to prepare media using basic components since these are usually available combined in dehydrated form from commercial sources; however, knowledge of a medium's basic compo nents is helpful in understanding how the medium works and what might be wrong when it does not work. Hence, the components have been listed for each medium included in this chapter. All dehydrated media must be prepared exactly according to the manu facturers' directions. Any deviation from these directions may adversely affect or significantly alter a medium's performance. Containers of media should be dated on receipt and when opened, and the media should never be used beyond expiration dates specified by the manufacturers or recom mended by quality control programs. -

View Our Full Water Sampling Vials Product Offering
TABLE OF CONTENTS Environmental Monitoring 1 Sample Prep and Dilution 8 Dehydrated Culture Media - Criterion™ 12 Prepared Culture Media 14 Chromogenic Media - HardyCHROM™ 18 CompactDry™ 20 Quality Control 24 Membrane Filtration 25 Rapid Tests 26 Lab Supplies/Sample Collection 27 Made in the USA Headquarters Distribution Centers 1430 West McCoy Lane Santa Maria, California Santa Maria, CA 93455 Olympia, Washington 800.266.2222 : phone Salt Lake City, Utah [email protected] Phoenix, Arizona HardyDiagnostics.com Dallas, Texas Springboro, Ohio Hardy Diagnostics has a Quality Lake City, Florida Management System that is certified Albany, New York to ISO 13485 and is a FDA licensed Copyright © 2020 Hardy Diagnostics Raleigh, North Carolina medical device manufacturer. Environmental Monitoring Hardy Diagnostics offers a wide selection of quality products intended to help keep you up to date with regulatory compliance, ensure the efficacy of your cleaning protocol, and properly monitor your environment. 800.266.2222 [email protected] HardyDiagnostics.com 1 Environmental Monitoring Air Sampling TRIO.BAS™ Impact Air Samplers introduced the newest generation of microbial air sampling. These ergonomically designed instruments combine precise air sampling with modern connectivity to help you properly assess the air quality of your laboratory and simplify your process. MONO DUO Each kit includes: Each kit includes: TRIO.BAS™ MONO air sampler, induction TRIO.BAS™ DUO air sampler, battery battery charger and cable, aspirating charger -

BD Industry Catalog
PRODUCT CATALOG INDUSTRIAL MICROBIOLOGY BD Diagnostics Diagnostic Systems Table of Contents Table of Contents 1. Dehydrated Culture Media and Ingredients 5. Stains & Reagents 1.1 Dehydrated Culture Media and Ingredients .................................................................3 5.1 Gram Stains (Kits) ......................................................................................................75 1.1.1 Dehydrated Culture Media ......................................................................................... 3 5.2 Stains and Indicators ..................................................................................................75 5 1.1.2 Additives ...................................................................................................................31 5.3. Reagents and Enzymes ..............................................................................................75 1.2 Media and Ingredients ...............................................................................................34 1 6. Identification and Quality Control Products 1.2.1 Enrichments and Enzymes .........................................................................................34 6.1 BBL™ Crystal™ Identification Systems ..........................................................................79 1.2.2 Meat Peptones and Media ........................................................................................35 6.2 BBL™ Dryslide™ ..........................................................................................................80 -

Bile Esculin Agar Base M340
Bile Esculin Agar Base M340 Bile Esculin Agar Base is a differential medium recommended for isolation and presumptive identification of group D Streptococci from food and pharmaceutical products. Composition** Ingredients Gms / Litre Peptone 5.000 Meat extract B # 3.000 Bile 40.000 Ferric citrate 0.500 Agar 15.000 Final pH ( at 25°C) 6.6±0.2 **Formula adjusted, standardized to suit performance parameters # Equivalent to Beef extract Directions Suspend 31.75 grams in 500 ml distilled water. Heat to boiling to dissolve the medium completely. Cool to 45-50°C.Add rehydrated contents of 1 vial of Esculin (FD050). Mix and dispense into tubes or flasks as desired. Sterilize by autoclaving at 15lbs pressure (121°C) for 15 minutes. Allow the tubed medium to solidify in slanted position. Principle And Interpretation Group D Streptococci possess the group D lipoteichoic acid antigen in their cell walls. Former Group D species, which are predominant normal inhabitants of the human gastrointestinal tract, are termed as faecal Streptococci or Enterococci (1). The unique ability of Enterococci to split esculin was reported by Meyer and Schonfeld (2). Enterococci and Group D Streptococci hydrolyse esculin to esculetin and dextrose, which reacts with ferric citrate producing brownish black precipitate (3). The use of esculin hydrolysis in identification of Enterococci was first cited by Rochaix (4). Bile Esculin Agar was originally formulated by Swan (6) for the isolation and identification of Group D Streptococci from food. Facklam and Moody (7, 8) further reported that using Bile Esculin Agar, Group D Streptococci could be differentiated from non Group D Streptococci. -

150259-ID-None.Pdf
DIPONEGORO JOURNAL OF MAQUARES Volume 5, Nomor 3, Tahun 2016, Halaman: 157-164 MANAGEMENT OF AQUATIC RESOURCES http://ejournal-s1.undip.ac.id/index.php/maquares ANALISIS TOTAL BAKTERI COLIFORM DI PERAIRAN MUARA KALI WISO JEPARA The Analysis of Total Coliform Bacteria in Kali Wiso Estuary Jepara Wiwid Widyaningsih, Supriharyono *), Niniek Widyorini Program Studi Manajemen Sumberdaya Perairan Fakultas Perikanan dan Ilmu Kelautan, Universitas Diponegoro Jl. Prof. Soedarto, SH, Tembalang, Semarang, Jawa Tengah – 50275, Telp/Fax. +6224 7474698 Email : [email protected] ABSTRAK Kali Wiso merupakan sungai yang berada di tengah kota Jepara. Perairan ini menjadi tempat pembuangan limbah-limbah secara langsung. Limbah tersebut diantaranya limbah domestik, limbah pasar, limbah kapal, serta limbah TPI. Berdasarkan masukan limbah tersebut menjadikan muara ini tercemar. Perairan yang tercemar dapat dilihat dari pengamatan secara fisika, kimia, maupun biologis. Kondisi perairan yang tercemar secara biologis dilihat dari keberadaan bakteri patogen yang ada di perairan. Indikator bakteri yang digunakan yaitu bakteri coliform, karena sifatnya yang berkorelasi positif dengan bakteri patogen lainnya. Pemanfaatan perairan ini digunakan untuk kegiatan pelabuhan, tempat bersandar kapal nelayan, serta kegiatan perikanan yang ada di sekitar perairan Jepara. Oleh karena itu perlu diketahui kepadatan bakteri coliform sehingga dapat bermanfaat sesuai dengan peruntukannya. Tujuan dari penelitian ini adalah untuk mengetahui total bakteri coliform serta mengetahui adanya bakteri Escherichia coli. Penelitain ini dilakukan pada bulan Maret 2016 di Muara Kali Wiso dengan dua kali pengulangan dalam kondisi pasang dan surut. Metode yang digunakan yaitu survei dengan teknik sampling purposive sampling. Metode analisa laboratorium yang digunakan berdasarkan SNI -01-2332- 1991. Kepadatan bakteri coliform pada perairan muara Kali Wiso yaitu >110.000 sel/100ml dan bakteri Escherichia coli sebesar >110.000 sel/100ml. -
DONE Food and Beverage Testing
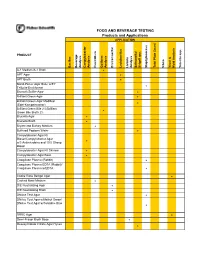
FOOD AND BEVERAGE TESTING Products and Applications APPLICATION PRODUCT Bacillus Beverage Analysis Campylobacter Analysis Clostridium Coliform Analysis Environmental Lactobacillus Listeria Analysis Salmonella/ Shigell spp. Staphylococcus Total Plate Count Vibrio Yeast & Mold Analysis Yersinia spp. A-1 Medium/A-1 Broth ! APT Agar ! APT Broth ! Baird-Parker Agar Base w/EY Tellurite Enrichment ! Bismuth Sulfite Agar ! Brilliant Green Agar ! Brilliant Green Agar Modified (Edel-Kampelmacher) ! Brilliant Green Bile 2%/Brilliant Green Bile Broth 2% ! Brucella Agar ! Brucella Broth ! Bryant and Burkey Medium ! Buffered Peptone Water ! Campylobacter Agar Kit Blaser/Campylobacter Agar w/5 Antimicrobics and 10% Sheep ! Blood Campylobacter Agar Kit Skirrow ! Campylobacter Agar Base ! Coagulase Plasma (Rabbit) ! Coagulase Plasma EDTA (Rabbit)/ Coagulase Plasma w/EDTA ! Cooke Rose Bengal Agar ! Cooked Meat Medium ! D/E Neutralizing Agar ! D/E Neutralizing Broth ! DNAse Test Agar ! DNAse Test Agar w/Methyl Green/ DNAse Test Agar w/Toluidine Blue ! DRBC Agar ! Demi-Fraser Broth Base ! Desoxycholate Citrate Agar Hynes ! FOOD AND BEVERAGE TESTING Products and Applications APPLICATION PRODUCT Bacillus Beverage Analysis Campylobacter Analysis Clostridium Coliform Analysis Environmental Lactobacillus Listeria Analysis Salmonella/ Shigell spp. Staphylococcus Total Plate Count Vibrio Yeast & Mold Analysis Yersinia spp. Differential Reinforced Clostridial Agar ! EC Medium/EC Broth ! EC Medium with MUG/EC Broth w/MUG ! Elliker Broth ! m Endo Agar LES ! m Endo -

Method 1680: Fecal Coliforms in Sewage Sludge (Biosolids) by Multiple-Tube Fermentation Using Lauryl Tryptose Broth (LTB) and EC Medium
United States Office of Water EPA-821-R-14-009 Environmental Protection Agency (4303-T) September 2014 Method 1680: Fecal Coliforms in Sewage Sludge (Biosolids) by Multiple-Tube Fermentation using Lauryl Tryptose Broth (LTB) and EC Medium U.S. Environmental Protection Agency Office of Water (4303T) 1200 Pennsylvania Avenue, NW Washington, DC 20460 Method 1680 Acknowledgments The contributions of the following persons and organizations to this study are gratefully acknowledged: Referee Laboratory • EPA Office of Research and Development, National Risk Management Research Lab: Mark C. Meckes and Karen M. White Volunteer Participant Laboratories • American Interplex: John Overbey and Lizbeth Huggins • BioVir Laboratories: Rick Danielson and Jim Truscott • City of Los Angeles Bureau of Sanitation Environmental Monitoring Division: Farhana Mohamed and Zora Bahariance • County Sanitation Districts of Los Angeles County: Shawn Thompson and Julie Millenbach • Environmental Associates: Susan Boutros and John Chandler • Hampton Roads Sanitation District: Anna Rule and Bob Maunz • King County Environmental Laboratory: Greg Ma and Bobbie Anderson • Hoosier Microbiological Laboratories: Don Hendrickson, Keri Nixon, Katy Bilger, and Lindsey Shelton • Massachusetts Water Resources Authority: Steve Rhode and Mariya Gofshteyn • Milwaukee Metropolitan Sewerage District: Jeff MacDonald and Tim O’Neill • University of Iowa Hygienic Laboratory: Nancy Hall and Cathy Lord • Utah Department of Health: Sanwat Chaudhuri and Devon Cole The following facilities provided