Head and Neck Embryology: an Overview of Development, Growth and Defect in the Human Fetus Allison Baylis University of Connecticut - Storrs, [email protected]
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Te2, Part Iii
TERMINOLOGIA EMBRYOLOGICA Second Edition International Embryological Terminology FIPAT The Federative International Programme for Anatomical Terminology A programme of the International Federation of Associations of Anatomists (IFAA) TE2, PART III Contents Caput V: Organogenesis Chapter 5: Organogenesis (continued) Systema respiratorium Respiratory system Systema urinarium Urinary system Systemata genitalia Genital systems Coeloma Coelom Glandulae endocrinae Endocrine glands Systema cardiovasculare Cardiovascular system Systema lymphoideum Lymphoid system Bibliographic Reference Citation: FIPAT. Terminologia Embryologica. 2nd ed. FIPAT.library.dal.ca. Federative International Programme for Anatomical Terminology, February 2017 Published pending approval by the General Assembly at the next Congress of IFAA (2019) Creative Commons License: The publication of Terminologia Embryologica is under a Creative Commons Attribution-NoDerivatives 4.0 International (CC BY-ND 4.0) license The individual terms in this terminology are within the public domain. Statements about terms being part of this international standard terminology should use the above bibliographic reference to cite this terminology. The unaltered PDF files of this terminology may be freely copied and distributed by users. IFAA member societies are authorized to publish translations of this terminology. Authors of other works that might be considered derivative should write to the Chair of FIPAT for permission to publish a derivative work. Caput V: ORGANOGENESIS Chapter 5: ORGANOGENESIS -

Induction and Specification of Cranial Placodes ⁎ Gerhard Schlosser
Developmental Biology 294 (2006) 303–351 www.elsevier.com/locate/ydbio Review Induction and specification of cranial placodes ⁎ Gerhard Schlosser Brain Research Institute, AG Roth, University of Bremen, FB2, PO Box 330440, 28334 Bremen, Germany Received for publication 6 October 2005; revised 22 December 2005; accepted 23 December 2005 Available online 3 May 2006 Abstract Cranial placodes are specialized regions of the ectoderm, which give rise to various sensory ganglia and contribute to the pituitary gland and sensory organs of the vertebrate head. They include the adenohypophyseal, olfactory, lens, trigeminal, and profundal placodes, a series of epibranchial placodes, an otic placode, and a series of lateral line placodes. After a long period of neglect, recent years have seen a resurgence of interest in placode induction and specification. There is increasing evidence that all placodes despite their different developmental fates originate from a common panplacodal primordium around the neural plate. This common primordium is defined by the expression of transcription factors of the Six1/2, Six4/5, and Eya families, which later continue to be expressed in all placodes and appear to promote generic placodal properties such as proliferation, the capacity for morphogenetic movements, and neuronal differentiation. A large number of other transcription factors are expressed in subdomains of the panplacodal primordium and appear to contribute to the specification of particular subsets of placodes. This review first provides a brief overview of different cranial placodes and then synthesizes evidence for the common origin of all placodes from a panplacodal primordium. The role of various transcription factors for the development of the different placodes is addressed next, and it is discussed how individual placodes may be specified and compartmentalized within the panplacodal primordium. -

Endothelium in the Pharyngeal Arches 3, 4 and 6 Is Derived from the Second Heart Field
Thomas Jefferson University Jefferson Digital Commons Center for Translational Medicine Faculty Papers Center for Translational Medicine 1-15-2017 Endothelium in the pharyngeal arches 3, 4 and 6 is derived from the second heart field. Xia Wang Thomas Jefferson University Dongying Chen Thomas Jefferson University Kelley Chen Thomas Jefferson University Ali Jubran Thomas Jefferson University AnnJosette Ramirez Thomas Jefferson University Follow this and additional works at: https://jdc.jefferson.edu/transmedfp Part of the Translational Medical Research Commons LetSee next us page know for additional how authors access to this document benefits ouy Recommended Citation Wang, Xia; Chen, Dongying; Chen, Kelley; Jubran, Ali; Ramirez, AnnJosette; and Astrof, Sophie, "Endothelium in the pharyngeal arches 3, 4 and 6 is derived from the second heart field." (2017). Center for Translational Medicine Faculty Papers. Paper 45. https://jdc.jefferson.edu/transmedfp/45 This Article is brought to you for free and open access by the Jefferson Digital Commons. The Jefferson Digital Commons is a service of Thomas Jefferson University's Center for Teaching and Learning (CTL). The Commons is a showcase for Jefferson books and journals, peer-reviewed scholarly publications, unique historical collections from the University archives, and teaching tools. The Jefferson Digital Commons allows researchers and interested readers anywhere in the world to learn about and keep up to date with Jefferson scholarship. This article has been accepted for inclusion in Center for Translational Medicine Faculty Papers by an authorized administrator of the Jefferson Digital Commons. For more information, please contact: [email protected]. Authors Xia Wang, Dongying Chen, Kelley Chen, Ali Jubran, AnnJosette Ramirez, and Sophie Astrof This article is available at Jefferson Digital Commons: https://jdc.jefferson.edu/transmedfp/45 HHS Public Access Author manuscript Author ManuscriptAuthor Manuscript Author Dev Biol Manuscript Author . -
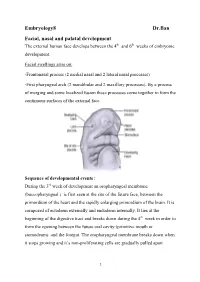
Embryology8 Dr.Ban Facial, Nasal and Palatal Development the External Human Face Develops Between the 4Th and 6Th Weeks of Embryonic Development
Embryology8 Dr.Ban Facial, nasal and palatal development The external human face develops between the 4th and 6th weeks of embryonic development. Facial swellings arise on: -Frontonasal process (2 medial nasal and 2 lateral nasal processes) -First pharyngeal arch (2 mandibular and 2 maxillary processes). By a process of merging and some localized fusion these processes come together to form the continuous surfaces of the external face. Sequence of developmental events : During the 3rd week of development an oropharyngeal membrane (buccopharyngeal ) is first seen at the site of the future face, between the primordium of the heart and the rapidly enlarging primordium of the brain. It is composed of ectoderm externally and endoderm internally. It lies at the beginning of the digestive tract and breaks down during the 4th week in order to form the opening between the future oral cavity (primitive mouth or stomodeum) and the foregut. The oropharyngeal membrane breaks down when it stops growing and it’s non-proliferating cells are gradually pulled apart 1 because they cannot fill the expanding area.The tissues around it expand very rapidly. The face develops from five primordia that appear in the fourth week: the frontonasal prominence, the two maxillary swellings, and the two mandibular swellings. The external face forms from two sources that surround the oropharyngeal membrane 1-Tissues of the frontonasal process that cover the forebrain, predominantly of neural crest origin. 2-The tissues of the first (or mandibular) pharyngeal arch, of mixed mesoderm and neural crest origin Face initially formed by 5 mesenchymal swellings ( prominences): Two mandibular prominences Two maxillary prominences Frontonasal prominence (midline structure is a single structure that is ventral to the forebrain. -

Vocabulario De Morfoloxía, Anatomía E Citoloxía Veterinaria
Vocabulario de Morfoloxía, anatomía e citoloxía veterinaria (galego-español-inglés) Servizo de Normalización Lingüística Universidade de Santiago de Compostela COLECCIÓN VOCABULARIOS TEMÁTICOS N.º 4 SERVIZO DE NORMALIZACIÓN LINGÜÍSTICA Vocabulario de Morfoloxía, anatomía e citoloxía veterinaria (galego-español-inglés) 2008 UNIVERSIDADE DE SANTIAGO DE COMPOSTELA VOCABULARIO de morfoloxía, anatomía e citoloxía veterinaria : (galego-español- inglés) / coordinador Xusto A. Rodríguez Río, Servizo de Normalización Lingüística ; autores Matilde Lombardero Fernández ... [et al.]. – Santiago de Compostela : Universidade de Santiago de Compostela, Servizo de Publicacións e Intercambio Científico, 2008. – 369 p. ; 21 cm. – (Vocabularios temáticos ; 4). - D.L. C 2458-2008. – ISBN 978-84-9887-018-3 1.Medicina �������������������������������������������������������������������������veterinaria-Diccionarios�������������������������������������������������. 2.Galego (Lingua)-Glosarios, vocabularios, etc. políglotas. I.Lombardero Fernández, Matilde. II.Rodríguez Rio, Xusto A. coord. III. Universidade de Santiago de Compostela. Servizo de Normalización Lingüística, coord. IV.Universidade de Santiago de Compostela. Servizo de Publicacións e Intercambio Científico, ed. V.Serie. 591.4(038)=699=60=20 Coordinador Xusto A. Rodríguez Río (Área de Terminoloxía. Servizo de Normalización Lingüística. Universidade de Santiago de Compostela) Autoras/res Matilde Lombardero Fernández (doutora en Veterinaria e profesora do Departamento de Anatomía e Produción Animal. -

6 Development of the Teeth: Root and Supporting Structures Nagat M
AVERY Chap.06 27-11-2002 10:09 Pagina 108 108 II Development of the Teeth and Supporting Structures 6 Development of the Teeth: Root and Supporting Structures Nagat M. ElNesr and James K. Avery Chapter Outline Introduction Introduction... 108 Objectives... 108 Root development is initiated through the contributions Root Sheath Development... 109 of the cells originating from the enamel organ, dental Single-Root Formation... 110 papilla, and dental follicle. The cells of the outer enamel Multiple-Root Formation... 111 epithelium contact the inner enamel epithelium at the Root Formation Anomalies... 112 base of the enamel organ, the cervical loop (Figs. 6.1 and Fate of the Epithelial Root Sheath (Hertwig's Sheath)... 113 6.2A). Later, with crown completion, the cells of the cer- Dental Follicle... 114 vical loop continue to grow away from the crown and Development of (Intermediate) Cementum... 116 become root sheath cells (Figs. 6.2B and 6.3). The inner Cellular and Acellular Cementum... 116 root sheath cells cause root formation by inducing the Development of the Periodontal Ligament... 117 adjacent cells of the dental papilla to become odonto- Development of the Alveolar Process... 119 blasts, which in turn will form root dentin. The root Summary... 121 sheath will further dictate whether the tooth will have Self-Evaluation Review... 122 single or multiple roots. The remainder of the cells of the dental papilla will then become the cells of the root pulp.The third compo- nent in root formation, the dental follicle, is the tissue that surrounds the enamel organ, the dental papilla, and the root. -

Is the Skeleton Male Or Female? the Pelvis Tells the Story
Activity: Is the Skeleton Male or Female? The pelvis tells the story. Distinct features adapted for childbearing distinguish adult females from males. Other bones and the skull also have features that can indicate sex, though less reliably. In young children, these sex-related features are less obvious and more difficult to interpret. Subtle sex differences are detectable in younger skeletons, but they become more defined following puberty and sexual maturation. What are the differences? Compare the two illustrations below in Figure 1. Female Pelvic Bones Male Pelvic Bones Broader sciatic notch Narrower sciatic notch Raised auricular surface Flat auricular surface Figure 1. Female and male pelvic bones. (Source: Smithsonian Institution, illustrated by Diana Marques) Figure 2. Pelvic bone of the skeleton in the cellar. (Source: Smithsonian Institution) Skull (Cranium and Mandible) Male Skulls Generally larger than female Larger projections behind the Larger brow ridges, with sloping, ears (mastoid processes) less rounded forehead Square chin with a more vertical Greater definition of muscle (acute) angle of the jaw attachment areas on the back of the head Figure 3. Male skulls. (Source: Smithsonian Institution, illustrated by Diana Marques) Female Skulls Smoother bone surfaces where Smaller projections behind the muscles attach ears (mastoid processes) Less pronounced brow ridges, Chin more pointed, with a larger, with more vertical forehead obtuse angle of the jaw Sharp upper margins of the eye orbits Figure 4. Female skulls. (Source: Smithsonian Institution, illustrated by Diana Marques) What Do You Think? Comparing the skull from the cellar in Figure 5 (below) with the illustrated male and female skulls in Figures 3 and 4, write Male or Female to note the sex depicted by each feature. -

Eagle's Syndrome, Elongated Styloid Process and New
Musculoskeletal Science and Practice 50 (2020) 102219 Contents lists available at ScienceDirect Musculoskeletal Science and Practice journal homepage: www.elsevier.com/locate/msksp Masterclass Eagle’s syndrome, elongated styloid process and new evidence for pre-manipulative precautions for potential cervical arterial dysfunction Andrea M. Westbrook a,*, Vincent J. Kabbaz b, Christopher R. Showalter c a Method Manual Physical Therapy & Wellness, Raleigh, NC, 27617, USA b HEAL Physical Therapy, Monona, WI, USA c Program Director, MAPS Accredited Fellowship in Orthopedic Manual Therapy, Cutchogue, NY, 11935, USA ARTICLE INFO ABSTRACT Keywords: Introduction: Safety with upper cervical interventions is a frequently discussed and updated concern for physical Eagle’s syndrome therapists, chiropractors and osteopaths. IFOMPT developed the framework for safety assessment of the cervical Styloid spine, and this topic has been discussed in-depth with past masterclasses characterizing carotid artery dissection CAD and cervical arterial dysfunction. Our masterclass will expand on this information with knowledge of specific Carotid anatomical anomalies found to produce Eagle’s syndrome, and cause carotid artery dissection, stroke and even Autonomic Manipulation death. Eagle’s syndrome is an underdiagnosed, multi-mechanism symptom assortment produced by provocation of the sensitive carotid space structures by styloid process anomalies. As the styloid traverses between the internal and external carotid arteries, provocation of the vessels and periarterial sympathetic nerve fiberscan lead to various neural, vascular and autonomic symptoms. Eagle’s syndrome commonly presents as neck, facial and jaw pain, headache and arm paresthesias; problems physical therapists frequently evaluate and treat. Purpose: This masterclass aims to outline the safety concerns, assessment and management of patients with Eagle’s syndrome and styloid anomalies. -

Embryology of Branchial Region
TRANSCRIPTIONS OF NARRATIONS FOR EMBRYOLOGY OF THE BRANCHIAL REGION Branchial Arch Development, slide 2 This is a very familiar picture - a median sagittal section of a four week embryo. I have actually done one thing correctly, I have eliminated the oropharyngeal membrane, which does disappear sometime during the fourth week of development. The cloacal membrane, as you know, doesn't disappear until the seventh week, and therefore it is still intact here, but unlabeled. But, I've labeled a couple of things not mentioned before. First of all, the most cranial part of the foregut, that is, the part that is cranial to the chest region, is called the pharynx. The part of the foregut in the chest region is called the esophagus; you probably knew that. And then, leading to the pharynx from the outside, is an ectodermal inpocketing, which is called the stomodeum. That originally led to the oropharyngeal membrane, but now that the oropharyngeal membrane is ruptured, the stomodeum is a pathway between the amniotic cavity and the lumen of the foregut. The stomodeum is going to become your oral cavity. Branchial Arch Development, slide 3 This is an actual picture of a four-week embryo. It's about 5mm crown-rump length. The stomodeum is labeled - that is the future oral cavity that leads to the pharynx through the ruptured oropharyngeal membrane. And I've also indicated these ridges separated by grooves that lie caudal to the stomodeum and cranial to the heart, which are called branchial arches. Now, if this is a four- week old embryo, clearly these things have developed during the fourth week, and I've never mentioned them before. -

The Cat Mandible (II): Manipulation of the Jaw, with a New Prosthesis Proposal, to Avoid Iatrogenic Complications
animals Review The Cat Mandible (II): Manipulation of the Jaw, with a New Prosthesis Proposal, to Avoid Iatrogenic Complications Matilde Lombardero 1,*,† , Mario López-Lombardero 2,†, Diana Alonso-Peñarando 3,4 and María del Mar Yllera 1 1 Unit of Veterinary Anatomy and Embryology, Department of Anatomy, Animal Production and Clinical Veterinary Sciences, Faculty of Veterinary Sciences, Campus of Lugo—University of Santiago de Compostela, 27002 Lugo, Spain; [email protected] 2 Engineering Polytechnic School of Gijón, University of Oviedo, 33203 Gijón, Spain; [email protected] 3 Department of Animal Pathology, Faculty of Veterinary Sciences, Campus of Lugo—University of Santiago de Compostela, 27002 Lugo, Spain; [email protected] 4 Veterinary Clinic Villaluenga, calle Centro n◦ 2, Villaluenga de la Sagra, 45520 Toledo, Spain * Correspondence: [email protected]; Tel.: +34-982-822-333 † Both authors contributed equally to this manuscript. Simple Summary: The small size of the feline mandible makes its manipulation difficult when fixing dislocations of the temporomandibular joint or mandibular fractures. In both cases, non-invasive techniques should be considered first. When not possible, fracture repair with internal fixation using bone plates would be the best option. Simple jaw fractures should be repaired first, and caudal to rostral. In addition, a ventral approach makes the bone fragments exposure and its manipulation easier. However, the cat mandible has little space to safely place the bone plate screws without damaging the tooth roots and/or the mandibular blood and nervous supply. As a consequence, we propose a conceptual model of a mandibular prosthesis that would provide biomechanical Citation: Lombardero, M.; stabilization, avoiding any unintended (iatrogenic) damage to those structures. -

Pharyngeal Arch Cheat Sheet V3
Pharyngeal Arches v3 (updates in red) Each pharyngeal arch contains: (1) a skeletal component (early on, a cartilaginous rod); (2) a muscular component; (3) an aortic arch; and (4) a nerve. Pharyngeal Arch Skeletal Component Muscles Aortic Arch Nerve First P. Arch Meckel's cartilage (malleus, incus) mm. of mastication, ant. belly 1st aortic arch Trigeminal N “mandibular maxillary prominence (maxilla*) of digastric, mylohyoid, tensor (maxillary AA) V2 and V3 arch” mandibular prominence (mandible*) tympani, tensor veli palatini Second P. Arch Reichert's cartilage (stapes, styloid mm. of facial expression, 2nd aortic arch Facial N “hyoid arch” process, stylohyoid lig.) buccinator, platysma, stapedius, (stapedial AA) VII hyoid, lesser cornua & superior body stylohyoid, post. belly of digastric Third P. Arch hyoid, greater cornua & inferior body stylopharyngeus 3rd aortic arch Glossophar. N (common carotids) IX Fourth P. Arch thyroid cartilage, epiglottic cartilage† cricothyroid, all intrinsic mm. 4th aortic arch Vagus: SLN of soft palate except tensor (aorta, R subclav.‡) & pharyn. plex. veli palatini X Fifth P. Arch - no derivatives - Sixth P. Arch cricoid, arytenoid, and corniculate all intrinsic mm. of larynx 6th aortic arch Vagus: RLN cartilages† except cricothyroid (pulmonary AA, X ductus arteriosus) * The maxillary and mandibular prominences are modeled on the first arch, but the bones develop separately from the first arch cartilage. † According to some sources the corniculate and cuneiform cartilages are derived from the 4th arch, not the 6th; I may revise this later on. ‡ Right subclavian A is derived from 4th aortic arch, right dorsal aorta, and right 7th intersegmental A; left subclavian A is derived from left 7th intersegmental A only, with no aortic arch component. -

Results Description of the SKULLS. the Overall Size of Both Skulls Was Considered to Be Within Normal Limits for Their Ethnic
Ossification Defects and Craniofacial Morphology In Incomplete Forms of Mandibulofacial Dysostosis A Description of Two Dry Skulls ERIK DAHL, D.D.S., DR. ODONT. ARNE BJORK, D.D.S., ODONT. DR. Copenhagen, Denmark The morphology of two East Indian dry skulls exhibiting anomalies which were suggested to represent incomplete forms of mandibulofacial dysostosis is described. Obvious although minor ossification anomalies were found localized to the temporal, sphenoid, the zygomatic, the maxillary and the mandibular bones. The observations substantiate the concept of the regional and bilateral nature of this malformation syndrome. Bilateral orbital deviations, hypoplasia of the malar bones, and incomplete zygomatic arches appear to be hard tissue aberrations which may be helpful in exami- nation for subclinical carrier status. Changes in mandibular morphology seem to be less distinguishing features in incomplete or abortive types of mandibulofacial dysostosis. KEY WORDS craniofacial problems, mandible, mandibulofacial dysostosis, maxilla, sphenoid bone, temporal bone, zygomatic bone Mandibulofacial dysostosis (MFD) often roentgencephalometric examinations were results in the development of a characteristic made of the skulls, and tomograms were ob- facial disfigurement with considerable simi- tained of the internal and middle ear. Com- larity between affected individuals. However, parisons were made with normal adult skulls the symptoms may vary highly in respect to and with an adult skull exhibiting the char- type and degree, and both incomplete and acteristics of MFD. All of the skulls were from abortive forms of the syndrome have been the same ethnic group. ' reported in the literature (Franceschetti and Klein, 1949; Moss et al., 1964; Rogers, 1964). Results In previous papers, we have shown the DEsCRIPTION OF THE SKULLS.