Therapeutic Targeting of Nuclear Transport
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

KETCH1 Imports HYL1 to Nucleus for Mirna Biogenesis in Arabidopsis
KETCH1 imports HYL1 to nucleus for miRNA biogenesis in Arabidopsis Zhonghui Zhanga,b,1, Xinwei Guoa,c,1, Chunxiao Gea,1, Zeyang Maa, Mengqiu Jianga, Tianhong Lic, Hisashi Koiwad, Seong Wook Yange, and Xiuren Zhanga,2 aDepartment of Biochemistry and Biophysics, Institute for Plant Genomics and Biotechnology, Texas A&M University, College Station, TX 77843; bGuangdong Provincial Key Laboratory of Biotechnology for Plant Development, School of Life Science, South China Normal University, Guangzhou 510631, China; cCollege of Horticulture, China Agricultural University, Beijing 100193, China; dDepartment of Horticultural Sciences, Texas A&M University, College Station, TX 77843; and eDepartment of Systems Biology, College of Life Science and Biotechnology, Yonsei University, Seoul 120-749, Republic of Korea Edited by Xuemei Chen, University of California, Riverside, CA, and approved March 9, 2017 (received for review December 2, 2016) MicroRNA (miRNA) is processed from primary transcripts with hairpin premiRNAs in mammalians (11, 12). Importin-8 facilitates the structures (pri-miRNAs) by microprocessors in the nucleus. How recruitment of AGO2-containing RISC to target mRNAs to pro- cytoplasmic-borne microprocessor components are transported into mote efficient and specific gene silencing in the cytoplasm, whereas the nucleus to fulfill their functions remains poorly understood. Here, the protein can also transport AGO2 and AGO2 partners, GW we report KETCH1 (karyopherin enabling the transport of the proteins and miRNAs, into the nucleus to balance levels of cyto- cytoplasmic HYL1) as a partner of hyponastic leaves 1 (HYL1) protein, plasmic gene-silencing effectors (13–15). Arabidopsis encodes a core component of microprocessor in Arabidopsis and functional 18 importin β-proteins, among which few have also been reported counterpart of DGCR8/Pasha in animals. -

Karyopherin Beta 3 (IPO5) (BD027479) Human Untagged Clone Product Data
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for SC105419 Karyopherin beta 3 (IPO5) (BD027479) Human Untagged Clone Product data: Product Type: Expression Plasmids Product Name: Karyopherin beta 3 (IPO5) (BD027479) Human Untagged Clone Tag: Tag Free Symbol: IPO5 Vector: pCMV6-XL4 E. coli Selection: Ampicillin (100 ug/mL) Cell Selection: None Fully Sequenced ORF: >NCBI ORF sequence for BD027479, the custom clone sequence may differ by one or more nucleotides CTTCTCTCTCACGCCTAGCGCAATGGCGGCGGCCGCGGCGGASCAGCAACAGTTCTACCTGCTCCTGGGA AACCTGCTCAGCCCCGACAATGTGGTCCGGAAACAGGCAGAGGAAACCTATGAGAATANTCCCAGGCCAG TCAAAGATCACATTCCTCTTACAAGCCATCAGAAATACAACAGCTGCTGAAGAGGCTAGACAAATGGCCG CCGTTCTCCTAAGACGTCTCTTGTCCTCTGCATTTGATGAAGTCTATCCAGCACTTCCCTCTGATGTTCA GACTGCCATCAAGAGTGAGCTACTCATGATTATTCAGATGGAAACACAATCTAGCATGAGGAAAAAAGTT TGTGATATTGCGGSAGAACTGGCCAGGAATTTAATAGATGAGGATGGCAATAACCAGTGGCCCGAAGTTT GAAGTTCCTTTTTGATTCAGTCAG 5' Read Nucleotide >OriGene 5' read for BD027479 unedited Sequence: TGTATACGACTCATATAGGGCGGCCGCGAATCGGCACGAGGCGCCGGCGCCGGCGGCCGC GGCGGGGTGAGAGGCCGCGAGGCCCCGCCCCGTCCTCCCCTTTCCCCTTTGCCCCGCCCT TCCCGCGCGGCCCCCCGCAAGCCCCGCGCCGCCGCTGGTGCCGGTCCCCGCGCTGGGCCC GCCCCCGCCCCTCCCGCGGCCCGCGAGCGCGCCTCACGGCTCCTGTCTCCCCTCCCTCCT TCTCTCTCACGCCTAGCGCAATGGCGGCGGCCGCGGCGGAGCAGCAACAGTTCTACCTGC TCCTGGGAAACCTGCTCAGCCCCGACAATGTGGTCCGGAAACAGGCAGAGGAAACCTATG AGAATATCCCAGGCCAGTCAAAGATCACATTCCTCTTACAAGCCATCAGAAATACAACAG -

Genetic and Genomic Analysis of Hyperlipidemia, Obesity and Diabetes Using (C57BL/6J × TALLYHO/Jngj) F2 Mice
University of Tennessee, Knoxville TRACE: Tennessee Research and Creative Exchange Nutrition Publications and Other Works Nutrition 12-19-2010 Genetic and genomic analysis of hyperlipidemia, obesity and diabetes using (C57BL/6J × TALLYHO/JngJ) F2 mice Taryn P. Stewart Marshall University Hyoung Y. Kim University of Tennessee - Knoxville, [email protected] Arnold M. Saxton University of Tennessee - Knoxville, [email protected] Jung H. Kim Marshall University Follow this and additional works at: https://trace.tennessee.edu/utk_nutrpubs Part of the Animal Sciences Commons, and the Nutrition Commons Recommended Citation BMC Genomics 2010, 11:713 doi:10.1186/1471-2164-11-713 This Article is brought to you for free and open access by the Nutrition at TRACE: Tennessee Research and Creative Exchange. It has been accepted for inclusion in Nutrition Publications and Other Works by an authorized administrator of TRACE: Tennessee Research and Creative Exchange. For more information, please contact [email protected]. Stewart et al. BMC Genomics 2010, 11:713 http://www.biomedcentral.com/1471-2164/11/713 RESEARCH ARTICLE Open Access Genetic and genomic analysis of hyperlipidemia, obesity and diabetes using (C57BL/6J × TALLYHO/JngJ) F2 mice Taryn P Stewart1, Hyoung Yon Kim2, Arnold M Saxton3, Jung Han Kim1* Abstract Background: Type 2 diabetes (T2D) is the most common form of diabetes in humans and is closely associated with dyslipidemia and obesity that magnifies the mortality and morbidity related to T2D. The genetic contribution to human T2D and related metabolic disorders is evident, and mostly follows polygenic inheritance. The TALLYHO/ JngJ (TH) mice are a polygenic model for T2D characterized by obesity, hyperinsulinemia, impaired glucose uptake and tolerance, hyperlipidemia, and hyperglycemia. -

An Evaluation of Host Factors As Novel Therapeutic Targets During Influenza Infection Using RNA Technologies
California State University, San Bernardino CSUSB ScholarWorks Electronic Theses, Projects, and Dissertations Office of aduateGr Studies 6-2018 An Evaluation of Host Factors as Novel Therapeutic Targets During Influenza Infection Using RNA Technologies Michael Ryan Haden Thompson California State University - San Bernardino Follow this and additional works at: https://scholarworks.lib.csusb.edu/etd Part of the Virology Commons Recommended Citation Thompson, Michael Ryan Haden, "An Evaluation of Host Factors as Novel Therapeutic Targets During Influenza Infection Using RNA Technologies" (2018). Electronic Theses, Projects, and Dissertations. 721. https://scholarworks.lib.csusb.edu/etd/721 This Thesis is brought to you for free and open access by the Office of aduateGr Studies at CSUSB ScholarWorks. It has been accepted for inclusion in Electronic Theses, Projects, and Dissertations by an authorized administrator of CSUSB ScholarWorks. For more information, please contact [email protected]. AN EVALUATION OF HOST FACTORS AS NOVEL THERAPEUTIC TARGETS DURING INFLUENZA INFECTION USING RNA TECHNOLOGIES _____________________ A Thesis Presented to the Faculty of California State University, San Bernardino _____________________ In Partial Fulfillment of the Requirements for the Degree Master of Science in Biology _____________________ by Michael Ryan Haden Thompson June 2018 AN EVALUATION OF HOST FACTORS AS NOVEL THERAPEUTIC TARGETS DURING INFLUENZA INFECTION USING RNA TECHNOLOGIES _____________________ A Thesis Presented to the Faculty of California State University, San Bernardino _____________________ By Michael Ryan Haden Thompson June 2018 Approved by: Dr. Laura L Newcomb, Committee Chair, Biology Dr. Nicole Bournias-Vardiabasis, Committee Member Dr. Daniel Nickerson, Committee Member © 2018 Michael Ryan Haden Thompson ABSTRACT Influenza A is a single-stranded, multi-segmented, negative sense RNA virus of the family Orthomyxoviridae and is the causative agent of seasonal Influenza. -

Genes with 5' Terminal Oligopyrimidine Tracts Preferentially Escape Global Suppression of Translation by the SARS-Cov-2 NSP1 Protein
Downloaded from rnajournal.cshlp.org on September 28, 2021 - Published by Cold Spring Harbor Laboratory Press Genes with 5′ terminal oligopyrimidine tracts preferentially escape global suppression of translation by the SARS-CoV-2 Nsp1 protein Shilpa Raoa, Ian Hoskinsa, Tori Tonna, P. Daniela Garciaa, Hakan Ozadama, Elif Sarinay Cenika, Can Cenika,1 a Department of Molecular Biosciences, University of Texas at Austin, Austin, TX 78712, USA 1Corresponding author: [email protected] Key words: SARS-CoV-2, Nsp1, MeTAFlow, translation, ribosome profiling, RNA-Seq, 5′ TOP, Ribo-Seq, gene expression 1 Downloaded from rnajournal.cshlp.org on September 28, 2021 - Published by Cold Spring Harbor Laboratory Press Abstract Viruses rely on the host translation machinery to synthesize their own proteins. Consequently, they have evolved varied mechanisms to co-opt host translation for their survival. SARS-CoV-2 relies on a non-structural protein, Nsp1, for shutting down host translation. However, it is currently unknown how viral proteins and host factors critical for viral replication can escape a global shutdown of host translation. Here, using a novel FACS-based assay called MeTAFlow, we report a dose-dependent reduction in both nascent protein synthesis and mRNA abundance in cells expressing Nsp1. We perform RNA-Seq and matched ribosome profiling experiments to identify gene-specific changes both at the mRNA expression and translation level. We discover that a functionally-coherent subset of human genes are preferentially translated in the context of Nsp1 expression. These genes include the translation machinery components, RNA binding proteins, and others important for viral pathogenicity. Importantly, we uncovered a remarkable enrichment of 5′ terminal oligo-pyrimidine (TOP) tracts among preferentially translated genes. -

Novel Mutation and Three Other Sequence Variants Segregating with Phenotype at Keratoconus 13Q32 Susceptibility Locus
European Journal of Human Genetics (2012) 20, 389–397 & 2012 Macmillan Publishers Limited All rights reserved 1018-4813/12 www.nature.com/ejhg ARTICLE Novel mutation and three other sequence variants segregating with phenotype at keratoconus 13q32 susceptibility locus Marta Czugala1,6, Justyna A Karolak1,6, Dorota M Nowak1, Piotr Polakowski2, Jose Pitarque3, Andrea Molinari3, Malgorzata Rydzanicz1, Bassem A Bejjani4, Beatrice YJT Yue5, Jacek P Szaflik2 and Marzena Gajecka*,1 Keratoconus (KTCN), a non-inflammatory corneal disorder characterized by stromal thinning, represents a major cause of corneal transplantations. Genetic and environmental factors have a role in the etiology of this complex disease. Previously reported linkage analysis revealed that chromosomal region 13q32 is likely to contain causative gene(s) for familial KTCN. Consequently, we have chosen eight positional candidate genes in this region: MBNL1, IPO5, FARP1, RNF113B, STK24, DOCK9, ZIC5 and ZIC2, and sequenced all of them in 51 individuals from Ecuadorian KTCN families and 105 matching controls. The mutation screening identified one mutation and three sequence variants showing 100% segregation under a dominant model with KTCN phenotype in one large Ecuadorian family. These substitutions were found in three different genes: c.2262A4C (p.Gln754His) and c.720+43A4GinDOCK9; c.2377-132A4CinIPO5 and c.1053+29G4CinSTK24. PolyPhen analyses predicted that c.2262A4C (Gln754His) is possibly damaging for the protein function and structure. Our results suggest that c.2262A4C (p.Gln754His) -
![Karyopherin Beta 3 (IPO5) Mouse Monoclonal Antibody [Clone ID: OTI1C5] Product Data](https://docslib.b-cdn.net/cover/8149/karyopherin-beta-3-ipo5-mouse-monoclonal-antibody-clone-id-oti1c5-product-data-488149.webp)
Karyopherin Beta 3 (IPO5) Mouse Monoclonal Antibody [Clone ID: OTI1C5] Product Data
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for TA811923 Karyopherin beta 3 (IPO5) Mouse Monoclonal Antibody [Clone ID: OTI1C5] Product data: Product Type: Primary Antibodies Clone Name: OTI1C5 Applications: WB Recommended Dilution: WB 1:2000 Reactivity: Human, Mouse, Rat Host: Mouse Isotype: IgG1 Clonality: Monoclonal Immunogen: Human recombinant protein fragment corresponding to amino acids 1-220 of human IPO5 (NP_002262) produced in E.coli. Formulation: PBS (PH 7.3) containing 1% BSA, 50% glycerol and 0.02% sodium azide. Concentration: 1 mg/ml Purification: Purified from mouse ascites fluids or tissue culture supernatant by affinity chromatography (protein A/G) Conjugation: Unconjugated Storage: Store at -20°C as received. Stability: Stable for 12 months from date of receipt. Predicted Protein Size: 125.4 kDa Gene Name: importin 5 Database Link: NP_002262 Entrez Gene 70572 MouseEntrez Gene 306182 RatEntrez Gene 3843 Human O00410 This product is to be used for laboratory only. Not for diagnostic or therapeutic use. View online » ©2021 OriGene Technologies, Inc., 9620 Medical Center Drive, Ste 200, Rockville, MD 20850, US 1 / 3 Karyopherin beta 3 (IPO5) Mouse Monoclonal Antibody [Clone ID: OTI1C5] – TA811923 Background: Nucleocytoplasmic transport, a signal- and energy-dependent process, takes place through nuclear pore complexes embedded in the nuclear envelope. The import of proteins containing a nuclear localization signal (NLS) requires the NLS import receptor, a heterodimer of importin alpha and beta subunits also known as karyopherins. Importin alpha binds the NLS-containing cargo in the cytoplasm and importin beta docks the complex at the cytoplasmic side of the nuclear pore complex. -

Mir-17-92 Fine-Tunes MYC Expression and Function to Ensure
ARTICLE Received 31 Mar 2015 | Accepted 22 Sep 2015 | Published 10 Nov 2015 DOI: 10.1038/ncomms9725 OPEN miR-17-92 fine-tunes MYC expression and function to ensure optimal B cell lymphoma growth Marija Mihailovich1, Michael Bremang1, Valeria Spadotto1, Daniele Musiani1, Elena Vitale1, Gabriele Varano2,w, Federico Zambelli3, Francesco M. Mancuso1,w, David A. Cairns1,w, Giulio Pavesi3, Stefano Casola2 & Tiziana Bonaldi1 The synergism between c-MYC and miR-17-19b, a truncated version of the miR-17-92 cluster, is well-documented during tumor initiation. However, little is known about miR-17-19b function in established cancers. Here we investigate the role of miR-17-19b in c-MYC-driven lymphomas by integrating SILAC-based quantitative proteomics, transcriptomics and 30 untranslated region (UTR) analysis upon miR-17-19b overexpression. We identify over one hundred miR-17-19b targets, of which 40% are co-regulated by c-MYC. Downregulation of a new miR-17/20 target, checkpoint kinase 2 (Chek2), increases the recruitment of HuR to c- MYC transcripts, resulting in the inhibition of c-MYC translation and thus interfering with in vivo tumor growth. Hence, in established lymphomas, miR-17-19b fine-tunes c-MYC activity through a tight control of its function and expression, ultimately ensuring cancer cell homeostasis. Our data highlight the plasticity of miRNA function, reflecting changes in the mRNA landscape and 30 UTR shortening at different stages of tumorigenesis. 1 Department of Experimental Oncology, European Institute of Oncology, Via Adamello 16, Milan 20139, Italy. 2 Units of Genetics of B cells and lymphomas, IFOM, FIRC Institute of Molecular Oncology Foundation, Milan 20139, Italy. -

Aneuploidy: Using Genetic Instability to Preserve a Haploid Genome?
Health Science Campus FINAL APPROVAL OF DISSERTATION Doctor of Philosophy in Biomedical Science (Cancer Biology) Aneuploidy: Using genetic instability to preserve a haploid genome? Submitted by: Ramona Ramdath In partial fulfillment of the requirements for the degree of Doctor of Philosophy in Biomedical Science Examination Committee Signature/Date Major Advisor: David Allison, M.D., Ph.D. Academic James Trempe, Ph.D. Advisory Committee: David Giovanucci, Ph.D. Randall Ruch, Ph.D. Ronald Mellgren, Ph.D. Senior Associate Dean College of Graduate Studies Michael S. Bisesi, Ph.D. Date of Defense: April 10, 2009 Aneuploidy: Using genetic instability to preserve a haploid genome? Ramona Ramdath University of Toledo, Health Science Campus 2009 Dedication I dedicate this dissertation to my grandfather who died of lung cancer two years ago, but who always instilled in us the value and importance of education. And to my mom and sister, both of whom have been pillars of support and stimulating conversations. To my sister, Rehanna, especially- I hope this inspires you to achieve all that you want to in life, academically and otherwise. ii Acknowledgements As we go through these academic journeys, there are so many along the way that make an impact not only on our work, but on our lives as well, and I would like to say a heartfelt thank you to all of those people: My Committee members- Dr. James Trempe, Dr. David Giovanucchi, Dr. Ronald Mellgren and Dr. Randall Ruch for their guidance, suggestions, support and confidence in me. My major advisor- Dr. David Allison, for his constructive criticism and positive reinforcement. -

Downloaded the “Top Edge” Version
bioRxiv preprint doi: https://doi.org/10.1101/855338; this version posted December 6, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 1 Drosophila models of pathogenic copy-number variant genes show global and 2 non-neuronal defects during development 3 Short title: Non-neuronal defects of fly homologs of CNV genes 4 Tanzeen Yusuff1,4, Matthew Jensen1,4, Sneha Yennawar1,4, Lucilla Pizzo1, Siddharth 5 Karthikeyan1, Dagny J. Gould1, Avik Sarker1, Yurika Matsui1,2, Janani Iyer1, Zhi-Chun Lai1,2, 6 and Santhosh Girirajan1,3* 7 8 1. Department of Biochemistry and Molecular Biology, Pennsylvania State University, 9 University Park, PA 16802 10 2. Department of Biology, Pennsylvania State University, University Park, PA 16802 11 3. Department of Anthropology, Pennsylvania State University, University Park, PA 16802 12 4 contributed equally to work 13 14 *Correspondence: 15 Santhosh Girirajan, MBBS, PhD 16 205A Life Sciences Building 17 Pennsylvania State University 18 University Park, PA 16802 19 E-mail: [email protected] 20 Phone: 814-865-0674 21 1 bioRxiv preprint doi: https://doi.org/10.1101/855338; this version posted December 6, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 22 ABSTRACT 23 While rare pathogenic copy-number variants (CNVs) are associated with both neuronal and non- 24 neuronal phenotypes, functional studies evaluating these regions have focused on the molecular 25 basis of neuronal defects. -

Case Report 12Q14 Microdeletions: Additional Case Series with Confirmation of a Macrocephaly Region
Hindawi Publishing Corporation Case Reports in Genetics Volume 2015, Article ID 192071, 7 pages http://dx.doi.org/10.1155/2015/192071 Case Report 12q14 Microdeletions: Additional Case Series with Confirmation of a Macrocephaly Region Adrian Mc Cormack,1 Cynthia Sharpe,2 Nerine Gregersen,3 Warwick Smith,4 Ian Hayes,3 Alice M. George,1 and Donald R. Love1 1 Diagnostic Genetics, LabPLUS, Auckland City Hospital, P.O. Box 110031, Auckland 1148, New Zealand 2Department of Neuroservices, Starship Children’s Health, Private Bag 92024, Auckland 1142, New Zealand 3Genetic Health Service New Zealand-Northern Hub, Auckland City Hospital, Private Bag 92024, Auckland 1142, New Zealand 4Middlemore Hospital, Private Bag 93311, Otahuhu, Auckland 1640, New Zealand Correspondence should be addressed to Donald R. Love; [email protected] Received 16 April 2015; Revised 7 July 2015; Accepted 7 July 2015 Academic Editor: Patrick Morrison Copyright © 2015 Adrian Mc Cormack et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. To date, there have been only a few reports of patients carrying a microdeletion in chromosome 12q14. These patients usually present with pre- and postnatal growth retardation, and developmental delay. Here we report on two additional patients with both genotype and phenotype differences. Similar to previously published cases, one patient has haploinsufficiency ofthe HMGA2 gene and shows severe short stature and developmental delay. The second patient is only one of a handful without the loss of the HMGA2 gene and shows a much better growth profile, but with absolute macrocephaly. -
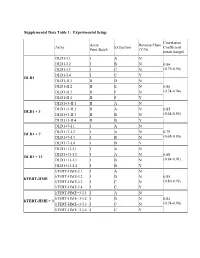
Table 3: Average Gene Expression Profiles by Chromosome
Supplemental Data Table 1: Experimental Setup Correlation Array Reverse Fluor Array Extraction Coefficient Print Batch (Y/N) mean (range) DLD1-I.1 I A N DLD1-I.2 I B N 0.86 DLD1-I.3 I C N (0.79-0.90) DLD1-I.4 I C Y DLD1 DLD1-II.1 II D N DLD1-II.2 II E N 0.86 DLD1-II.3 II F N (0.74-0.94) DLD1-II.4 II F Y DLD1+3-II.1 II A N DLD1+3-II.2 II A N 0.85 DLD1 + 3 DLD1+3-II.3 II B N (0.64-0.95) DLD1+3-II.4 II B Y DLD1+7-I.1 I A N DLD1+7-I.2 I A N 0.79 DLD1 + 7 DLD1+7-I.3 I B N (0.68-0.90) DLD1+7-I.4 I B Y DLD1+13-I.1 I A N DLD1+13-I.2 I A N 0.88 DLD1 + 13 DLD1+13-I.3 I B N (0.84-0.91) DLD1+13-I.4 I B Y hTERT-HME-I.1 I A N hTERT-HME-I.2 I B N 0.85 hTERT-HME hTERT-HME-I.3 I C N (0.80-0.92) hTERT-HME-I.4 I C Y hTERT-HME+3-I.1 I A N hTERT-HME+3-I.2 I B N 0.84 hTERT-HME + 3 hTERT-HME+3-I.3 I C N (0.74-0.90) hTERT-HME+3-I.4 I C Y Supplemental Data Table 2: Average gene expression profiles by chromosome arm DLD1 hTERT-HME Ratio.7 Ratio.1 Ratio.3 Ratio.3 Chrom.