The Effects of Lidocaine and Mefenamic Acid on Post-Episiotomy
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

4% Citanest® Plain Dental (Prilocaine Hydrochloride Injection, USP) for Local Anesthesia in Dentistry
4% Citanest® Plain Dental (Prilocaine Hydrochloride Injection, USP) For Local Anesthesia in Dentistry Rx only DESCRIPTION 4% Citanest Plain Dental (prilocaine HCI Injection, USP), is a sterile, non-pyrogenic isotonic solution that contains a local anesthetic agent and is administered parenterally by injection. See INDICATIONS AND USAGE for specific uses. The quantitative composition is shown in Table 1. 4% Citanest Plain Dental contains prilocaine HCl, which is chemically designated as propanamide, N-(2- methyl-phenyl) -2- (propylamino)-, monohydrochloride and has the following structural formula: Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. The specific quantitative composition is shown in Table 1. TABLE 1. COMPOSITION Product Identification Formula (mg/mL) Prilocaine HCl pH 4% Citanest® Plain Dental 40.0 6.0 to 7.0 Note: Sodium hydroxide or hydrochloric acid may be used to adjust the pH of 4% Citanest Plain Dental Injection. CLINICAL PHARMACOLOGY Mechanism of Action: Prilocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses, thereby effecting local anesthetic action. Onset and Duration of Action: When used for infiltration injection in dental patients, the time of onset of anesthesia averages less than 2 minutes with an average duration of soft tissue anesthesia of approximately 2 hours. Based on electrical stimulation studies, 4% Citanest Plain Dental Injection provides a duration of pulpal anesthesia of approximately 10 minutes in maxillary infiltration injections. In clinical studies, this has been found to provide complete anesthesia for procedures lasting an average of 20 minutes. When used for inferior alveolar nerve block, the time of onset of 4% Citanest Plain Dental Injection averages less than three minutes with an average duration of soft tissue anesthesia of approximately 2 ½hours. -

Topical Therapy As a Treatmentfor Brachioradial
Journal of Case Reports: Open Access Case report Open Access Topical Therapy as a Treatmentfor Brachioradial Pruritis: a Case Report Brianna De Souza M.D, Amy McMichael M.D* Department of Dermatology, Wake Forest University School of Medicine, Winston-Salem, North Carolina *Corresponding author: Amy McMichael, MD Department of Dermatology, Wake Forest Baptist Medical Center,1 Medical Center Blvd, Winston-Salem, NC 27157, Phone: 336-716-7882, Email: [email protected] Received Date: April 12, 2019 Accepted Date: May 06, 2019 Published Date: May 08, 2019 Citation: Brianna De Souza (2019) Topical Therapy as a Treatmentfor Brachioradial Pruritis: a Case Report. Case Reports: Open Access 4: 1-5. Abstract Management of brachioradial pruritus (BRP) presents a formidable challenge to dermatologists and neurologists. BRP is a rare, neurocutaneous condition characterized by sharply localized, chronic pain with associated itching, burning, stinging, and or tingling sensation. Effective care of this patient population is confounded by limitations within the litera- ture, comprised of case series and case reports. We present a case of one middle-aged female with a chronic history of BRP recalcitrant to the following oral therapies: pregabalin, gabapentin, mirtazapine, prednisone, and amitriptyline, as well as topical triamcinolone. After being evaluated in the clinic, the patient was started on combination therapy withKetamine 10%, Amitriptyline 5%, and Lidocaine 5% topical cream to which she responded. Keywords: Brachioradial pruritus, Brachioradial, Pruritus, Neurocutaneous ©2019 The Authors. Published by the JScholar under the terms of the Crea- tive Commons Attribution License http://creativecommons.org/licenses/ by/3.0/, which permits unrestricted use, provided the original author and source are credited. -

Local Anesthetics
Local Anesthetics Introduction and History Cocaine is a naturally occurring compound indigenous to the Andes Mountains, West Indies, and Java. It was the first anesthetic to be discovered and is the only naturally occurring local anesthetic; all others are synthetically derived. Cocaine was introduced into Europe in the 1800s following its isolation from coca beans. Sigmund Freud, the noted Austrian psychoanalyst, used cocaine on his patients and became addicted through self-experimentation. In the latter half of the 1800s, interest in the drug became widespread, and many of cocaine's pharmacologic actions and adverse effects were elucidated during this time. In the 1880s, Koller introduced cocaine to the field of ophthalmology, and Hall introduced it to dentistry Overwiev Local anesthetics (LAs) are drugs that block the sensation of pain in the region where they are administered. LAs act by reversibly blocking the sodium channels of nerve fibers, thereby inhibiting the conduction of nerve impulses. Nerve fibers which carry pain sensation have the smallest diameter and are the first to be blocked by LAs. Loss of motor function and sensation of touch and pressure follow, depending on the duration of action and dose of the LA used. LAs can be infiltrated into skin/subcutaneous tissues to achieve local anesthesia or into the epidural/subarachnoid space to achieve regional anesthesia (e.g., spinal anesthesia, epidural anesthesia, etc.). Some LAs (lidocaine, prilocaine, tetracaine) are effective on topical application and are used before minor invasive procedures (venipuncture, bladder catheterization, endoscopy/laryngoscopy). LAs are divided into two groups based on their chemical structure. The amide group (lidocaine, prilocaine, mepivacaine, etc.) is safer and, hence, more commonly used in clinical practice. -

Antagonism of Lidocaine Inhibition by Open-Channel Blockers That Generate Resurgent Na Current
4976 • The Journal of Neuroscience, March 13, 2013 • 33(11):4976–4987 Cellular/Molecular Antagonism of Lidocaine Inhibition by Open-Channel Blockers That Generate Resurgent Na Current Jason S. Bant,1,3 Teresa K. Aman,2,3 and Indira M. Raman1,2,3 1Interdepartmental Biological Sciences Program, 2Northwestern University Interdepartmental Neuroscience Program, and 3Department of Neurobiology, Northwestern University, Evanston, Illinois 60208 Na channels that generate resurgent current express an intracellular endogenous open-channel blocking protein, whose rapid binding upon depolarization and unbinding upon repolarization minimizes fast and slow inactivation. Na channels also bind exogenous com- pounds, such as lidocaine, which functionally stabilize inactivation. Like the endogenous blocking protein, these use-dependent inhibi- tors bind most effectively at depolarized potentials, raising the question of how lidocaine-like compounds affect neurons with resurgent Na current. We therefore recorded lidocaine inhibition of voltage-clamped, tetrodotoxin-sensitive Na currents in mouse Purkinje neu- rons, which express a native blocking protein, and in mouse hippocampal CA3 pyramidal neurons with and without a peptide from the   cytoplasmic tail of NaV 4 (the 4 peptide), which mimics endogenous open-channel block. To control channel states during drug exposure, lidocaine was applied with rapid-solution exchange techniques during steps to specific voltages. Inhibition of Na currents by lidocaine was diminished by either the 4 peptide or the native blocking protein. In peptide-free CA3 cells, prolonging channel opening with a site-3 toxin, anemone toxin II, reduced lidocaine inhibition; this effect was largely occluded by open-channel blockers, suggesting that lidocaine binding is favored by inactivation but prevented by open-channel block. -
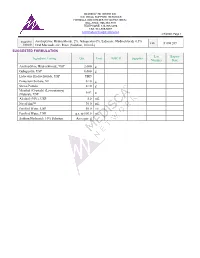
Amitriptyline Hydrochloride 2%, Gabapentin 6%, Lidocaine Hydrochloride 0.5% FIN F 008 269 Formula Oral Mucoadhesive Rinse (Solution, 100 Ml)
MEDISCA® NETWORK INC. TECHNICAL SUPPORT SERVICES FORMULATION CHEMISTRY DEPARTMENT TOLL-FREE: 866-333-7811 TELEPHONE: 514-905-5096 FAX: 514-905-5097 [email protected] 4/7/2020; Page 1 Suggested Amitriptyline Hydrochloride 2%, Gabapentin 6%, Lidocaine Hydrochloride 0.5% FIN F 008 269 Formula Oral Mucoadhesive Rinse (Solution, 100 mL) SUGGESTED FORMULATION Lot Expiry Ingredient Listing Qty. Unit NDC # Supplier Number Date Amitriptyline Hydrochloride, USP 2.000 g Gabapentin, USP 6.000 g Lidocaine Hydrochloride, USP TBD Potassium Sorbate, NF 0.10 g Stevia Powder 0.10 g Menthol (Crystals) (Levorotatory) 0.02 g (Natural), USP Alcohol (95%), USP 5.0 mL NovaFilm™ 30.0 mL Purified Water, USP 50.0 mL Purified Water, USP q.s. to 100.0 mL Sodium Hydroxide 10% Solution As required MEDISCA® NETWORK INC. TECHNICAL SUPPORT SERVICES FORMULATION CHEMISTRY DEPARTMENT TOLL-FREE: 866-333-7811 TELEPHONE: 514-905-5096 FAX: 514-905-5097 [email protected] 4/7/2020; Page 2 Suggested Amitriptyline Hydrochloride 2%, Gabapentin 6%, Lidocaine Hydrochloride 0.5% FIN F 008 269 Formula Oral Mucoadhesive Rinse (Solution, 100 mL) SPECIAL PREPARATORY CONSIDERATIONS Ingredient-Specific Information Light Sensitive (protect from light whenever possible): Amitriptyline Hydrochloride, Gabapentin Hygroscopic (protect from moisture whenever possible): Stevia Powder Narrow Therapeutic Index Lidocaine Hydrochloride Suggested Preparatory Guidelines ■ Non-Sterile Preparation □ Sterile Preparation Processing Error / To account for processing error and pH testing considerations during preparation, it is Testing Considerations: suggested to measure an additional 3 to 5% of the required quantities of ingredients. Special Instruction: This formula may contain one or more Active Pharmaceutical Ingredients (APIs) that may be classified as hazardous, please refer & verify the current NIOSH list of Antineoplastic and Other Hazardous Drugs in Healthcare Settings, 2016. -

Document Detail
DENTSPLY INTERNATIONAL Document Detail 1 Type: 0090-MSDS Document No.: 0090-SDS-008[00] Title: 4% Citanest Plain Dental - US and CA Comment Status: CURRENT Effective Date: 12-Jan-2018 Approvals Role Sign-off By Sign-off Date Pharma Marketing Approver Joe Santos 12-Jan-2018 2:40 pm GMT Pharma Regulatory Affairs Approver Deb Crouse 10-Jan-2018 6:43 pm GMT Pharma Regulatory Affairs Approver Lakeisha R Williams 10-Jan-2018 4:21 pm GMT Pharma Marketing Approver Richard Tootchen 10-Jan-2018 6:57 pm GMT Pharma Quality Assurance Approver Candace Walker 10-Jan-2018 4:05 pm GMT DPD Research and Development Tom Simonton 10-Jan-2018 8:48 pm GMT DPD Document Control Lucy Brenneman 12-Jan-2018 7:47 pm GMT Pharma Document Control Beth Bodnar 12-Jan-2018 4:18 pm GMT SOP DOC.022-F01 Rev.1 Safety Data Sheet Safety Data Sheet conforms to Regulation (EC) 1907/2006, Regulation (EC) 1272/2008 and Regulation (EC) 2015/830, Date Issued: 01November 2017 US 29CFR1910.1200, Canada Hazardous Products Document Number: SDS-008 Regulation. Date Revised: New SDS Revision Number: 00 1. IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY/UNDERTAKING 1.1 Product Identifier: Trade Name (as labeled): 4% Citanest® Plain DENTAL (prilocaine HCl Injection, USP) Part/Item Numbers: 46616 71115 1.2 Relevant Identified Uses of the Substance or Mixture and Uses Advised Against: Recommended Use: Local anesthetic solution for use in peripheral nerve blocks Restrictions on Use: For Professional Use Only 1.3 Details of the Supplier of the Safety Data Sheet: Manufacturer/Supplier Name: DENTSPLY Pharmaceutical Manufacturer/Supplier Address: 1301 Smile Way York, PA 17404 Manufacturer/Supplier Telephone Number: 800-989-8826 or 717-767-8502 (Product Information) Email address: [email protected] 1.4 Emergency Telephone Number: Transportation Emergency Contact Number: 800-424-9300 Chemtrec 2. -

Comparison of Articaine and Prilocaine Anestiesia by Infiltration in Maxillary and Mandibular Archles Daniel A
Anesth Prog 37:230-237 1990 SCIENTIFIC REPORTS Comparison of Articaine and Prilocaine Anestiesia by Infiltration in Maxillary and Mandibular Archles Daniel A. Haas, DDS, PhD, David G. Harper, DDS, Michael A. Saso, DDS, and Earle R. Young, DDS, MSc Department of Anaesthesia, Faculty of Dentistry, University of Toronto Claims that labial infiltration of the local anesthetic It is commonly accepted that in order to achieve local articaine HCl (Ultracaine DS"M) results in anesthesia for procedures on teeth or buccal soft tissue anesthesia of mandibular pulpal as well as in the maxillary arch, administration of local anesthetics maxillary and mandibular lingual soft tissue have by buccal infiltration, also known as a paraperiosteal field never been scientifically substantiated. The aim of block, is routinely successful. Palatal soft tissue anesthesia this investigation was to evaluate these claims, by requires a separate palatal injection, a technique that is comparing articaine to a standard anesthetic, often painful for the patient. Any local anesthetic that prilocaine HCI (Citanest ForteTM). To investigate would permit use of buccal infiltration to gain palatal anes- this, a double blind, randomized study was thesia would be of great advantage in dentistry. For proce- conducted in healthy adult volunteers. In each dures in the mandibular arch, in adults, the thickness of volunteer, the ability to induce maxillary and buccal cortical bone precludes buccal infiltration ap- mandibular anesthesia following labial infiltration proaches producing pulpal or lingual soft tissue anesthe- with articaine was compared to prilocaine given sia, necessitating administration of local anesthetic by contralaterally. Anesthesia was determined by nerve block techniques. -

Local Anesthetics – Substances with Multiple Application in Medicine
ISSN 2411-958X (Print) European Journal of January-April 2016 ISSN 2411-4138 (Online) Interdisciplinary Studies Volume 2, Issue 1 Local Anesthetics – Substances with Multiple Application in Medicine Rodica SÎRBU Ovidius University of Constanta, Faculty of Pharmacy, Campus Corp B, University Alley No. 1, Constanta, Romania Emin CADAR UMF Carol Davila Bucharest, Faculty of Pharmacy, Str. Traian Vuia No. 6, Sector 2, Bucharest, Romania Cezar Laurențiu TOMESCU Ovidius University of Constanta, Faculty of Medicine, Campus Corp B, University Alley No. 1, Constanta, Romania Cristina-Luiza ERIMIA Corresponding author, [email protected] Ovidius University of Constanta, Faculty of Pharmacy, Campus Corp B, University Alley No. 1, Constanta, Romania Stelian PARIS Ovidius University of Constanta, Faculty of Pharmacy, Campus Corp B, University Alley No. 1, Constanta, Romania Aneta TOMESCU Ovidius University of Constanta, Faculty of Medicine, Campus Corp B, University Alley No. 1, Constanta, Romania Abstract Local anesthetics are substances which, by local action groups on the runners, cause loss of reversible a painful sensation, delimited corresponding to the application. They allow small surgery, short in duration and the endoscopic maneuvers. May be useful in soothe teething pain of short duration and in the locking of the nervous disorders in medical care. Local anesthesia is a process useful for the carrying out of surgery and of endoscopic maneuvers, to soothe teething pain in certain conditions, for depriving the temporary structures peripheral nervous control. Reversible locking of the transmission nociceptive, the set of the vegetative and with a local anesthetic at the level of the innervations peripheral nerve, roots and runners, a trunk nervous, around the components of a ganglion or coolant is cefalorahidian practice anesthesia loco-regional. -

Fenestrations Control Resting-State Block of a Voltage-Gated Sodium
Fenestrations control resting-state block of a voltage- gated sodium channel Tamer M. Gamal El-Dina,1, Michael J. Lenaeusa,b,1, Ning Zhenga,c,2,3, and William A. Catteralla,2,3 aDepartment of Pharmacology, University of Washington, Seattle, WA 98195; bDivision of General Internal Medicine, Department of Medicine, University of Washington, Seattle, WA 98195; and cHoward Hughes Medical Institute, University of Washington, Seattle, WA 98195 Contributed by William A. Catterall, October 31, 2018 (sent for review September 4, 2018; reviewed by Ryan Hibbs, Michael C. Sanguinetti, and Joerg Striessnig) Potency of drug action is usually determined by binding to a specific Receptor Hypothesis posits that resting-state block is mediated receptor site on target proteins. In contrast to this conventional by drug entry from the lipid phase of the membrane into the drug paradigm, we show here that potency of local anesthetics (LAs) and receptor site in the pore, and rapid open-state block occurs as the antiarrhythmic drugs (AADs) that block sodium channels is con- drug enters the open pore from the cytoplasm (15). Both of these trolled by fenestrations that allow drug access to the receptor site forms of block are enhanced when the channel enters the inac- directly from the membrane phase. Voltage-gated sodium channels tivated state, which has high affinity for bound drug (15). It is initiate action potentials in nerve and cardiac muscle, where their well-established that LAs and AADs can reach their binding site hyperactivity causes pain and cardiac arrhythmia, respectively. LAs from the intracellular side if the activation gate is open, which and AADs selectively block sodium channels in rapidly firing nerve gives open-state block, and that inactivation increases the affinity and muscle cells to relieve these conditions. -

Hypersensitivity to Local Anaesthetics – 6 Facts and 7 Myths
HYPERSENSITIVITY TO LOCAL ANAESTHETICS – 6 FACTS AND 7 MYTHS Joanna Lukawska, MRCP The pharmacological differences between LAs make M Rosario Caballero, PhD certain LAs more favourable than others for specific procedures, i.e. lignocaine is widely used for subcuta- Sophia Tsabouri, MD, PhD neous injection, and bupivacaine is preferred for Pierre Dugué, FRCP, MD, APH epidural or gum injections. Drug Allergy Clinic, Guy’s & St Thomas’ Hospitals, NHS Foundation Trust, London, UK Amino-ester LA chemical structure ABSTRACT Local anaesthetics (LAs) are commonly used drugs. In spite of their widespread use, true hypersensitiv- ity appears to be very infrequent. In fact most of the adverse reactions are due to pharmacological, toxic or vasovagal effects of LAs. Our review of the literature has shown that true allergy to LA is in fact exceptional. Skin tests for LA allergy, including skin-prick tests (SPT) and intrader- Aromatic Ester Amine group mal (ID) tests, have poor sensitivity and specificity. group True LA allergy, when appropriate, has to be con- firmed by challenge. Provocation challenge is safe and well tolerated. Since the discovery of the anaesthetic effect of cocaine in 1884, local anaesthetics (LAs) have been widely used. It has been estimated that 6 million peo- ple are injected with LAs each day around the world. In spite of their widespread use, true hypersensitivity appears to be infrequent. However the perception of allergy to LA among the general public is high. In our Aromatic Amide Amine drug allergy clinic, referrals for LA allergy are in the group group same range as penicillin or aspirin allergy referrals. -

Anaesthetic Implications of Calcium Channel Blockers
436 Anaesthetic implications of calcium channel Leonard C. Jenkins aA MD CM FRCPC blockers Peter J. Scoates a sc MD FRCPC CONTENTS The object of this review is to emphasize the anaesthetic implications of calcium channel block- Physiology - calcium/calcium channel blockers Uses of calcium channel blockers ers for the practising anaesthetist. These drugs have Traditional played an expanding role in therapeutics since their Angina pectoris introduction and thus anaesthetists can expect to see Arrhythmias increasing numbers of patients presenting for anaes- Hypertension thesia who are being treated with calcium channel Newer and investigational Cardiac blockers. Other reviews have emphasized the basic - Hypertrophic cardiomyopathy pharmacology of calcium channel blockers. 1-7 - Cold cardioplegia - Pulmonary hypertension Physiology - calcium/calcium channel blockers Actions on platelets Calcium plays an important role in many physio- Asthma Obstetrics logical processes, such as blood coagulation, en- - Premature labor zyme systems, muscle contraction, bone metabo- - Pre-eclampsia lism, synaptic transmission, and cell membrane Achalasia and oesophageal spasm excitability. Especially important is the role of Increased intraocular pressure therapy calcium in myocardial contractility and conduction Protective effect on kidney after radiocontrast Cerebral vasospasm as well as in vascular smooth muscle reactivity. 7 Induced hypotensive anaesthesia Thus, it can be anticipated that any drug interfering Drag interactions with calcium channel blockers with the action of calcium could have widespread With anaesthetic agents effects. Inhalation agents In order to understand the importance of calcium - Effect on haemodynamics - Effect on MAC in cellular excitation, it is necessary to review some Neuromuscular blockers membrane physiology. Cell membranes are pri- Effects on epinephrine-induced arrhythmias marily phospholipids arranged in a bilayer. -

Estonian Statistics on Medicines 2016 1/41
Estonian Statistics on Medicines 2016 ATC code ATC group / Active substance (rout of admin.) Quantity sold Unit DDD Unit DDD/1000/ day A ALIMENTARY TRACT AND METABOLISM 167,8985 A01 STOMATOLOGICAL PREPARATIONS 0,0738 A01A STOMATOLOGICAL PREPARATIONS 0,0738 A01AB Antiinfectives and antiseptics for local oral treatment 0,0738 A01AB09 Miconazole (O) 7088 g 0,2 g 0,0738 A01AB12 Hexetidine (O) 1951200 ml A01AB81 Neomycin+ Benzocaine (dental) 30200 pieces A01AB82 Demeclocycline+ Triamcinolone (dental) 680 g A01AC Corticosteroids for local oral treatment A01AC81 Dexamethasone+ Thymol (dental) 3094 ml A01AD Other agents for local oral treatment A01AD80 Lidocaine+ Cetylpyridinium chloride (gingival) 227150 g A01AD81 Lidocaine+ Cetrimide (O) 30900 g A01AD82 Choline salicylate (O) 864720 pieces A01AD83 Lidocaine+ Chamomille extract (O) 370080 g A01AD90 Lidocaine+ Paraformaldehyde (dental) 405 g A02 DRUGS FOR ACID RELATED DISORDERS 47,1312 A02A ANTACIDS 1,0133 Combinations and complexes of aluminium, calcium and A02AD 1,0133 magnesium compounds A02AD81 Aluminium hydroxide+ Magnesium hydroxide (O) 811120 pieces 10 pieces 0,1689 A02AD81 Aluminium hydroxide+ Magnesium hydroxide (O) 3101974 ml 50 ml 0,1292 A02AD83 Calcium carbonate+ Magnesium carbonate (O) 3434232 pieces 10 pieces 0,7152 DRUGS FOR PEPTIC ULCER AND GASTRO- A02B 46,1179 OESOPHAGEAL REFLUX DISEASE (GORD) A02BA H2-receptor antagonists 2,3855 A02BA02 Ranitidine (O) 340327,5 g 0,3 g 2,3624 A02BA02 Ranitidine (P) 3318,25 g 0,3 g 0,0230 A02BC Proton pump inhibitors 43,7324 A02BC01 Omeprazole