Lidocaine Hcl) Solution
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Evaluation of Liposomal Delivery System for Topical Anesthesia
THERAPEUTICS FOR THE CLINICIAN Evaluation of Liposomal Delivery System for Topical Anesthesia Mohamed L. Elsaie, MD, MBA; Leslie S. Baumann, MD Local anesthesia is an integral aspect of cutane- infiltrative anesthetics, now can be accomplished ous surgery. Its effects provide a reversible loss safely and comfortably with the use of topi- of sensation in a limited area of skin, allowing cal anesthetics.1 Topical anesthetics originated in dermatologists to perform diagnostic and thera- South America; native Peruvians noted perioral peutic procedures safely, with minimal discomfort numbness when chewing the leaf of the cocoa plant and risk to the patient. Moreover, the skin acts (Erythroxylon coca). The active alkaloid, cocaine, was as a major target as well as principle barrier for isolated by Niemann in 1860 and applied to con- topical/transdermal (TT) drug delivery. The stra- junctival mucosa for topical anesthesia by Koller in tum corneum (SC) plays a crucial role in barrier 1884. The development of similar benzoic acid esters function for TT drug delivery. Despite the major continued until 1943 when Loefgren synthesized lido- research and development efforts in TT systems caine hydrochloride, the first amide anesthetic.2 and their implementation for use of topical anes- We review the administration of local anes- thetics, low SC permeability limits the useful- thetics, specifically the liposomal delivery system ness of topical delivery, which has led to other for topical anesthesia, based on a review of the delivery system developments, including vesicu- literature and clinical experience. lar systems such as liposomes, niosomes, and proniosomes, with effectiveness relying on their Anatomy physiochemical properties. -

Topical Therapy As a Treatmentfor Brachioradial
Journal of Case Reports: Open Access Case report Open Access Topical Therapy as a Treatmentfor Brachioradial Pruritis: a Case Report Brianna De Souza M.D, Amy McMichael M.D* Department of Dermatology, Wake Forest University School of Medicine, Winston-Salem, North Carolina *Corresponding author: Amy McMichael, MD Department of Dermatology, Wake Forest Baptist Medical Center,1 Medical Center Blvd, Winston-Salem, NC 27157, Phone: 336-716-7882, Email: [email protected] Received Date: April 12, 2019 Accepted Date: May 06, 2019 Published Date: May 08, 2019 Citation: Brianna De Souza (2019) Topical Therapy as a Treatmentfor Brachioradial Pruritis: a Case Report. Case Reports: Open Access 4: 1-5. Abstract Management of brachioradial pruritus (BRP) presents a formidable challenge to dermatologists and neurologists. BRP is a rare, neurocutaneous condition characterized by sharply localized, chronic pain with associated itching, burning, stinging, and or tingling sensation. Effective care of this patient population is confounded by limitations within the litera- ture, comprised of case series and case reports. We present a case of one middle-aged female with a chronic history of BRP recalcitrant to the following oral therapies: pregabalin, gabapentin, mirtazapine, prednisone, and amitriptyline, as well as topical triamcinolone. After being evaluated in the clinic, the patient was started on combination therapy withKetamine 10%, Amitriptyline 5%, and Lidocaine 5% topical cream to which she responded. Keywords: Brachioradial pruritus, Brachioradial, Pruritus, Neurocutaneous ©2019 The Authors. Published by the JScholar under the terms of the Crea- tive Commons Attribution License http://creativecommons.org/licenses/ by/3.0/, which permits unrestricted use, provided the original author and source are credited. -

Prolonged Duration Topical Corneal Anesthesia with the Cationic Lidocaine Derivative QX-314
https://doi.org/10.1167/tvst.8.5.28 Article Prolonged Duration Topical Corneal Anesthesia With the Cationic Lidocaine Derivative QX-314 Alan G. Woodruff1,2, Claudia M. Santamaria1, Manisha Mehta1,2, Grant L. Pemberton1, Kathleen Cullion1,2,4, and Daniel S. Kohane1,2,3 1 Kohane Lab for Biomaterials and Drug Delivery, Department of Anesthesia, Perioperative and Pain Medicine, Division of Critical Care, Boston Children’s Hospital, Boston, MA, USA 2 Harvard Medical School, Boston, MA, USA 3 David H. Koch Institute, Massachusetts Institute of Technology, Cambridge, MA, USA 4 Department of Medicine, Division of Medicine Critical Care, Boston Children’s Hospital, Harvard Medical School, Boston, MA, USA Correspondence: Daniel S. Kohane, Purpose: Topical corneal local anesthetics are short acting and may impair corneal Laboratory for Biomaterials and healing. In this study we compared corneal anesthesia and toxicity of topically applied Drug Delivery, Department of Anes- N-ethyl lidocaine (QX-314) versus the conventional local anesthetic, proparacaine thesia, Division of Critical Care (PPC). Medicine, Boston Children’s Hospi- tal, Harvard Medical School, 61 Methods: Various concentrations of QX-314 and 15 mM (0.5%) PPC were topically Binney Street, Room 361, Boston, applied to rat corneas. Corneal anesthesia was assessed with a Cochet-Bonnet MA 02115, USA. e-mail: Daniel. esthesiometer at predetermined time points. PC12 cells were exposed to the same [email protected] solutions to assess cytotoxicity. Repeated topical corneal administration in rats was then used to assess for histologic evidence of toxicity. Finally, we created uniform Received: 6 May 2019 corneal epithelial defects in rats and assessed the effect of repeated administration of Accepted: 15 August 2019 these compounds on the defect healing rate. -

Antagonism of Lidocaine Inhibition by Open-Channel Blockers That Generate Resurgent Na Current
4976 • The Journal of Neuroscience, March 13, 2013 • 33(11):4976–4987 Cellular/Molecular Antagonism of Lidocaine Inhibition by Open-Channel Blockers That Generate Resurgent Na Current Jason S. Bant,1,3 Teresa K. Aman,2,3 and Indira M. Raman1,2,3 1Interdepartmental Biological Sciences Program, 2Northwestern University Interdepartmental Neuroscience Program, and 3Department of Neurobiology, Northwestern University, Evanston, Illinois 60208 Na channels that generate resurgent current express an intracellular endogenous open-channel blocking protein, whose rapid binding upon depolarization and unbinding upon repolarization minimizes fast and slow inactivation. Na channels also bind exogenous com- pounds, such as lidocaine, which functionally stabilize inactivation. Like the endogenous blocking protein, these use-dependent inhibi- tors bind most effectively at depolarized potentials, raising the question of how lidocaine-like compounds affect neurons with resurgent Na current. We therefore recorded lidocaine inhibition of voltage-clamped, tetrodotoxin-sensitive Na currents in mouse Purkinje neu- rons, which express a native blocking protein, and in mouse hippocampal CA3 pyramidal neurons with and without a peptide from the   cytoplasmic tail of NaV 4 (the 4 peptide), which mimics endogenous open-channel block. To control channel states during drug exposure, lidocaine was applied with rapid-solution exchange techniques during steps to specific voltages. Inhibition of Na currents by lidocaine was diminished by either the 4 peptide or the native blocking protein. In peptide-free CA3 cells, prolonging channel opening with a site-3 toxin, anemone toxin II, reduced lidocaine inhibition; this effect was largely occluded by open-channel blockers, suggesting that lidocaine binding is favored by inactivation but prevented by open-channel block. -
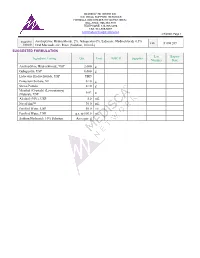
Amitriptyline Hydrochloride 2%, Gabapentin 6%, Lidocaine Hydrochloride 0.5% FIN F 008 269 Formula Oral Mucoadhesive Rinse (Solution, 100 Ml)
MEDISCA® NETWORK INC. TECHNICAL SUPPORT SERVICES FORMULATION CHEMISTRY DEPARTMENT TOLL-FREE: 866-333-7811 TELEPHONE: 514-905-5096 FAX: 514-905-5097 [email protected] 4/7/2020; Page 1 Suggested Amitriptyline Hydrochloride 2%, Gabapentin 6%, Lidocaine Hydrochloride 0.5% FIN F 008 269 Formula Oral Mucoadhesive Rinse (Solution, 100 mL) SUGGESTED FORMULATION Lot Expiry Ingredient Listing Qty. Unit NDC # Supplier Number Date Amitriptyline Hydrochloride, USP 2.000 g Gabapentin, USP 6.000 g Lidocaine Hydrochloride, USP TBD Potassium Sorbate, NF 0.10 g Stevia Powder 0.10 g Menthol (Crystals) (Levorotatory) 0.02 g (Natural), USP Alcohol (95%), USP 5.0 mL NovaFilm™ 30.0 mL Purified Water, USP 50.0 mL Purified Water, USP q.s. to 100.0 mL Sodium Hydroxide 10% Solution As required MEDISCA® NETWORK INC. TECHNICAL SUPPORT SERVICES FORMULATION CHEMISTRY DEPARTMENT TOLL-FREE: 866-333-7811 TELEPHONE: 514-905-5096 FAX: 514-905-5097 [email protected] 4/7/2020; Page 2 Suggested Amitriptyline Hydrochloride 2%, Gabapentin 6%, Lidocaine Hydrochloride 0.5% FIN F 008 269 Formula Oral Mucoadhesive Rinse (Solution, 100 mL) SPECIAL PREPARATORY CONSIDERATIONS Ingredient-Specific Information Light Sensitive (protect from light whenever possible): Amitriptyline Hydrochloride, Gabapentin Hygroscopic (protect from moisture whenever possible): Stevia Powder Narrow Therapeutic Index Lidocaine Hydrochloride Suggested Preparatory Guidelines ■ Non-Sterile Preparation □ Sterile Preparation Processing Error / To account for processing error and pH testing considerations during preparation, it is Testing Considerations: suggested to measure an additional 3 to 5% of the required quantities of ingredients. Special Instruction: This formula may contain one or more Active Pharmaceutical Ingredients (APIs) that may be classified as hazardous, please refer & verify the current NIOSH list of Antineoplastic and Other Hazardous Drugs in Healthcare Settings, 2016. -

The Effects of Lidocaine and Mefenamic Acid on Post-Episiotomy
Shiraz E-Med J. 2016 March; 17(3):e36286. doi: 10.17795/semj36286. Published online 2016 March 27. Research Article The Effects of Lidocaine and Mefenamic Acid on Post-Episiotomy Pain: A Comparative Study Masoumeh Delaram,1,* Lobat Jafar Zadeh,2 and Sahand Shams3 1Faculty of Nursing and Midwifery, Shahrekord University of Medical Sciences, Shahrekord, IR Iran 2Faculty of Medicine, Shahrekord University of Medical Sciences, Shahrekord, IR Iran 3Faculty of Veterinary Medicine, Shahrekord University, Shahrekord, IR Iran *Corresponding author: Masoumeh Delaram, Faculty of Nursing and Midwifery, Shahrekord University of Medical Sciences, Shahrekord, IR Iran. Tel: +98-3813335648, Fax: +98-3813346714, E-mail: [email protected] Received 2016 January 13; Revised 2016 February 29; Accepted 2016 March 04. Abstract Background: Most women suffer pain following an episiotomy and oral non-steroidal anti-inflammatory drugs are commonly used for pain relief. Due to the gastrointestinal side effects of oral drugs, it seems that women are more accepting of topical medications for pain relief. Objectives: Therefore, the aim of this study was to compare the effects of lidocaine and mefenamic acid on post-episiotomy pain. Patients and Methods: This clinical trial was carried out in 2011. It involved sixty women with singleton pregnancy who were given an episiotomy at 38 to 42 weeks of gestation. The participants were randomly divided into two groups. One group received 2% lido- caine cream (n = 30), while the other group received 250 mg of mefenamic acid (n = 30). The data were collected via a questionnaire and a visual analog scale. Pain intensity was compared from the first complaint by the mother and at 6, 12, and 24 hours after the delivery in both groups. -

Safety Alert: Risks Associated with Ophthalmic Anesthetics
WRHA Pharmacy Program Health Sciences Centre MS-189 820 Sherbrook St. Winnipeg, Manitoba R3A 1R9 CANADA TEL: 204-787-7183 Fax: 204-787-3195 FAX: 204-787-3195 Safety Alert: Risks associated with Ophthalmic Anesthetics The self-administration of ophthalmic anesthetics by patients for the relief of eye pain should be avoided and they should not be given to patients to take home for pain relief. Vision threatening complications of topical anesthetic abuse are common. There is no indication for the use of ophthalmic anesthetics except for diagnostic and short term therapeutic purposes (the removal of a foreign body or ocular surgery) and therefore, these products should only be used under a physician’s supervision. Eye trauma resulting in a corneal abrasion (epithelial injury) is a common complaint in the Emergency department (1). A superficial corneal injury can cause intense pain causing a patient to seek medical help or immediate relief from available over the counter remedies. In Canada, only two topical ophthalmic anesthetic drugs are available commercially as single entities, proparacaine (proxymetacaine) and tetracaine (available in bottle and minim forms). Benoxinate (oxybuprocaine) is only available in combination with fluorescein (3). Lidocaine is also used in ophthalmic surgical procedures however, it is not available in the Canadian market as an ophthalmic preparation. Topical ophthalmic anesthetics function by blocking nerve conduction when applied to the cornea and conjunctiva. The ocular surface is innervated by the multiple branches of the trigeminal nerve. The cornea is supplied by the long and short ciliary nerves, the nasociliary nerve and the lacrimal nerve (4). Topical anesthetics reduce sodium permeability preventing generation and conduction of nerve impulses, increasing excitation threshold, and slowing the nerve impulse propagation. -

Fenestrations Control Resting-State Block of a Voltage-Gated Sodium
Fenestrations control resting-state block of a voltage- gated sodium channel Tamer M. Gamal El-Dina,1, Michael J. Lenaeusa,b,1, Ning Zhenga,c,2,3, and William A. Catteralla,2,3 aDepartment of Pharmacology, University of Washington, Seattle, WA 98195; bDivision of General Internal Medicine, Department of Medicine, University of Washington, Seattle, WA 98195; and cHoward Hughes Medical Institute, University of Washington, Seattle, WA 98195 Contributed by William A. Catterall, October 31, 2018 (sent for review September 4, 2018; reviewed by Ryan Hibbs, Michael C. Sanguinetti, and Joerg Striessnig) Potency of drug action is usually determined by binding to a specific Receptor Hypothesis posits that resting-state block is mediated receptor site on target proteins. In contrast to this conventional by drug entry from the lipid phase of the membrane into the drug paradigm, we show here that potency of local anesthetics (LAs) and receptor site in the pore, and rapid open-state block occurs as the antiarrhythmic drugs (AADs) that block sodium channels is con- drug enters the open pore from the cytoplasm (15). Both of these trolled by fenestrations that allow drug access to the receptor site forms of block are enhanced when the channel enters the inac- directly from the membrane phase. Voltage-gated sodium channels tivated state, which has high affinity for bound drug (15). It is initiate action potentials in nerve and cardiac muscle, where their well-established that LAs and AADs can reach their binding site hyperactivity causes pain and cardiac arrhythmia, respectively. LAs from the intracellular side if the activation gate is open, which and AADs selectively block sodium channels in rapidly firing nerve gives open-state block, and that inactivation increases the affinity and muscle cells to relieve these conditions. -

Anaesthetic Implications of Calcium Channel Blockers
436 Anaesthetic implications of calcium channel Leonard C. Jenkins aA MD CM FRCPC blockers Peter J. Scoates a sc MD FRCPC CONTENTS The object of this review is to emphasize the anaesthetic implications of calcium channel block- Physiology - calcium/calcium channel blockers Uses of calcium channel blockers ers for the practising anaesthetist. These drugs have Traditional played an expanding role in therapeutics since their Angina pectoris introduction and thus anaesthetists can expect to see Arrhythmias increasing numbers of patients presenting for anaes- Hypertension thesia who are being treated with calcium channel Newer and investigational Cardiac blockers. Other reviews have emphasized the basic - Hypertrophic cardiomyopathy pharmacology of calcium channel blockers. 1-7 - Cold cardioplegia - Pulmonary hypertension Physiology - calcium/calcium channel blockers Actions on platelets Calcium plays an important role in many physio- Asthma Obstetrics logical processes, such as blood coagulation, en- - Premature labor zyme systems, muscle contraction, bone metabo- - Pre-eclampsia lism, synaptic transmission, and cell membrane Achalasia and oesophageal spasm excitability. Especially important is the role of Increased intraocular pressure therapy calcium in myocardial contractility and conduction Protective effect on kidney after radiocontrast Cerebral vasospasm as well as in vascular smooth muscle reactivity. 7 Induced hypotensive anaesthesia Thus, it can be anticipated that any drug interfering Drag interactions with calcium channel blockers with the action of calcium could have widespread With anaesthetic agents effects. Inhalation agents In order to understand the importance of calcium - Effect on haemodynamics - Effect on MAC in cellular excitation, it is necessary to review some Neuromuscular blockers membrane physiology. Cell membranes are pri- Effects on epinephrine-induced arrhythmias marily phospholipids arranged in a bilayer. -

Conlaining Lidocaine Or Benzocaine Mark E
Anesth Prog 35:9-13 1988 Antimicrobial Properties of Topical Anestletc Liquids Conlaining Lidocaine or Benzocaine Mark E. Morrow, DDS, MSD,* and Charles W. Berry, PhDt *Department of Pediatric Dentistry, tDepartment of Microbiology, Baylor College of Dentistry, Dallas, Texas Six species of microorganisms commonly found ganisms have been isolated from the oral cavity,3 it is within the oral cavity were exposed for either one logical that an antiseptic agent should be wiped on the minute or two hours to 5% lidocaine liquid mucosa before the administration of an injectable local topical anesthetic and benzocaine liquid topical anesthetic in the dental setting. However, few dentists anesthetic. Mixtures of microorganisms and currently follow this regimen.4 Instead, the use of a anesthetics were diluted and plated onto a brain topical anesthetic just before administration of a local heart infusion medium. Reduction in cell viability anesthetic to block the pain of the needle puncture is very was 73-100% after exposure to the anesthetic common.5 agents when compared with the saline/buffer The possible antimicrobial properties of local anesthe- controls. A significant reduction (p < .005) in cell tic agents were originally described by Jonnesco.6 He growth by Streptococcus mutans, S. sanguis, S. indicated that there was no need to sterilize local anesthe- mitis, S. salivarius, Actinomyces viscosus, and tics used for spinal anesthesia because of their antiseptic Candida albicans was associated with a one- characteristics. The earliest work documenting the in vitro minute and two-hour exposure to lidocaine, antimicrobial activity of anesthetic agents was reported by benzocaine, 5% lidocaine, and the benzocaine Schlegal and Swan,7 who described the inhibition of vehicle control. -

Pharmacological Treatments Protocols of Alcohol and Drugs Abuse
Pharmacological Treatments Protocols of Alcohol and Drugs Abuse 1 Purpose of the protocols: Use and abuse of drugs and alcohol is becoming common and can have serious and harmful consequences on individuals, families, and society. Care with a tailored treatment program and follow-up options can be crucial to success. Treatment should include both medical and mental health services as needed in managing withdrawal symptoms, prevent relapse, and treat co- occurring conditions. Follow-up care may include community- or family-based recovery support systems. These protocols have been developed to guide medical practitioners and nurses in the use of the most effective available treatments of alcohol and drug abuse in the in-patient and out- patient settings and serve as a framework for clinical decisions and supporting best practices. Targeted end users: • Psychiatry and Addiction Medicine Consultants, Specialists and Residents • Nurses • Psychiatry clinical pharmacists • Pharmacists 2 TABLE OF CONTENTS 1. Chapter (1) Alcohol 4 3.1 Introduction 4 3.2 Intoxication 4 3.3 Withdrawal 7 2. Chapter (2) Benzodiazepines 21 2.1 Introduction 21 2.2 Intoxication 21 2.3 Withdrawal 22 3. Chapter (3) Opioids 27 3.1 Introduction 27 3.2 Intoxication 28 3.3 Withdrawal 29 4. Chapter (4) Psychostimulants 38 2.1 Introduction 38 2.2 Intoxication 39 2.3 Withdrawal 40 5. Chapter (5) Cannabis 41 3.1 Introduction 41 3.2 Intoxication 41 3.3 Withdrawal 43 6. References 46 3 CHAPTER 1 ALCOHOL INTRODUCTION Alcohol is a Central Nervous System (CNS) depressant. Its’ psychoactive properties contribute to changes in mood, cognition and behavior. -

(Lidocaine Hcl Injection, USP) 3 Xylocaine
1 721566-04 451175A/Revised February 2010 ® 2 Xylocaine (lidocaine HCl Injection, USP) ® 3 Xylocaine (lidocaine HCl and epinephrine Injection, USP) 4 For Infiltration and Nerve Block 5 Rx only 6 DESCRIPTION : 7 Xylocaine (lidocaine HCl) Injections are sterile, nonpyrogenic, aqueous solutions that contain a 8 local anesthetic agent with or without epinephrine and are administered parenterally by injection. 9 See INDICATIONS for specific uses. 10 Xylocaine solutions contain lidocaine HCl, which is chemically designated as acetamide, 11 2-(diethylamino)-N-(2,6-dimethylphenyl)-, monohydrochloride and has the molecular wt. 270.8. 12 Lidocaine HCl (C14H22N2O • HCl) has the following structural formula: 13 14 15 Epinephrine is (-) -3, 4-Dihydroxy-α-[(methylamino) methyl] benzyl alcohol and has the 16 molecular wt. 183.21. Epinephrine (C9H13NO3) has the following structural formula: 17 18 Dosage forms listed as Xylocaine-MPF indicate single dose solutions that are Methyl 19 Paraben Free (MPF). 20 Xylocaine MPF is a sterile, nonpyrogenic, isotonic solution containing sodium chloride. 21 Xylocaine in multiple dose vials: Each mL also contains 1 mg methylparaben as antiseptic 22 preservative. The pH of these solutions is adjusted to approximately 6.5 (5.0–7.0) with sodium 23 hydroxide and/or hydrochloric acid. 1 1 Xylocaine MPF with Epinephrine is a sterile, nonpyrogenic, isotonic solution containing 2 sodium chloride. Each mL contains lidocaine hydrochloride and epinephrine, with 0.5 mg 3 sodium metabisulfite as an antioxidant and 0.2 mg citric acid as a stabilizer. Xylocaine with 4 Epinephrine in multiple dose vials: Each mL also contains 1 mg methylparaben as antiseptic 5 preservative.