Role of Proteases in Cancer: a Review
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supplementary Data
Supplementary Data for Quantitative Changes in the Mitochondrial Proteome from Subjects with Mild Cognitive Impairment, Early Stage and Late Stage Alzheimer’s disease Table 1 - 112 unique, non-redundant proteins identified and quantified in at least two of the three analytical replicates for all three disease stages. Table 2 - MCI mitochondrial samples, Protein Summary Table 3 - MCI mitochondrial samples, Experiment 1 Table 4 - MCI mitochondrial samples, Experiment 2 Table 5 - MCI mitochondrial samples, Experiment 3 Table 6 - EAD Mitochondrial Study, Protein Summary Table 7 - EAD Mitochondrial Study, Experiment 1 Table 8 - EAD Mitochondrial Study, Experiment 2 Table 9 - EAD Mitochondrial Study, Experiment 3 Table 10 - LAD Mitochondrial Study, Protein Summary Table 11 - LAD Mitochondrial Study, Experiment 1 Table 12 - LAD Mitochondrial Study, Experiment 2 Table 13 - LAD Mitochondrial Study, Experiment 3 Supplemental Table 1. 112 unique, non-redundant proteins identified and quantified in at least two of the three analytical replicates for all three disease stages. Description Data MCI EAD LAD AATM_HUMAN (P00505) Aspartate aminotransferase, mitochondrial precursor (EC Mean 1.43 1.70 1.31 2.6.1.1) (Transaminase A) (Glutamate oxaloacetate transaminase 2) [MASS=47475] SEM 0.07 0.09 0.09 Count 3.00 3.00 3.00 ACON_HUMAN (Q99798) Aconitate hydratase, mitochondrial precursor (EC 4.2.1.3) Mean 1.24 1.61 1.19 (Citrate hydro-lyase) (Aconitase) [MASS=85425] SEM 0.05 0.17 0.18 Count 3.00 2.00 3.00 ACPM_HUMAN (O14561) Acyl carrier protein, mitochondrial -

Role of Cyclosporine in Gingival Hyperplasia: an in Vitro Study on Gingival Fibroblasts
International Journal of Molecular Sciences Article Role of Cyclosporine in Gingival Hyperplasia: An In Vitro Study on Gingival Fibroblasts 1, , 2, 3 3 Dorina Lauritano * y , Annalisa Palmieri y, Alberta Lucchese , Dario Di Stasio , Giulia Moreo 1 and Francesco Carinci 4 1 Department of Medicine and Surgery, Centre of Neuroscience of Milan, University of Milano-Bicocca, 20126 Milan, Italy; [email protected] 2 Department of Experimental, Diagnostic and Specialty Medicine, University of Bologna, via Belmoro 8, 40126 Bologna, Italy; [email protected] 3 Multidisciplinary Department of Medical and Dental Specialties, University of Campania-Luigi Vanvitelli, 80138 Naples, Italy; [email protected] (A.L.); [email protected] (D.D.S.) 4 Department of Morphology, Surgery and Experimental Medicine, University of Ferrara, 44121 Ferrara, Italy; [email protected] * Correspondence: [email protected]; Tel.: +39-335-679-0163 These authors contributed equally to this work. y Received: 25 November 2019; Accepted: 13 January 2020; Published: 16 January 2020 Abstract: Background: Gingival hyperplasia could occur after the administration of cyclosporine A. Up to 90% of the patients submitted to immunosuppressant drugs have been reported to suffer from this side effect. The role of fibroblasts in gingival hyperplasia has been widely discussed by literature, showing contrasting results. In order to demonstrate the effect of cyclosporine A on the extracellular matrix component of fibroblasts, we investigated the gene expression profile of human fibroblasts after cyclosporine A administration. Materials and methods: Primary gingival fibroblasts were stimulated with 1000 ng/mL cyclosporine A solution for 16 h. Gene expression levels of 57 genes belonging to the “Extracellular Matrix and Adhesion Molecules” pathway were analyzed using real-time PCR in treated cells, compared to untreated cells used as control. -

2335 Roles of Molecules Involved in Epithelial/Mesenchymal Transition
[Frontiers in Bioscience 13, 2335-2355, January 1, 2008] Roles of molecules involved in epithelial/mesenchymal transition during angiogenesis Giulio Ghersi Dipartimento di Biologia Cellulare e dello Sviluppo, Universita di Palermo, Italy TABLE OF CONTENTS 1. Abstract 2. Introduction 3. Extracellular matrix 3.1. ECM and integrins 3.2. Basal lamina components 4. Cadherins. 4.1. Cadherins in angiogenesis 5. Integrins. 5.1. Integrins in angiogenesis 6. Focal adhesion molecules 7. Proteolytic enzymes 7.1. Proteolytic enzymes inhibitors 7.2. Proteolytic enzymes in angiogenesis 8. Perspective 9. Acknowledgements 10. References 1.ABSTRACT 2. INTRODUCTION Formation of vessels requires “epithelial- Growth of new blood vessels (angiogenesis) mesenchymal” transition of endothelial cells, with several plays a key role in several physiological processes, such modifications at the level of endothelial cell plasma as vascular remodeling during embryogenesis and membranes. These processes are associated with wound healing tissue repair in the adult; as well as redistribution of cell-cell and cell-substrate adhesion pathological processes, including rheumatoid arthritis, molecules, cross talk between external ECM and internal diabetic retinopathy, psoriasis, hemangiomas, and cytoskeleton through focal adhesion molecules and the cancer (1). Vessel formation entails the “epithelial- expression of several proteolytic enzymes, including matrix mesenchymal” transition of endothelial cells (ECs) “in metalloproteases and serine proteases. These enzymes with vivo”; a similar phenotypic exchange can be induced “in their degradative action on ECM components, generate vitro” by growing ECs to low cell density, or in “wound molecules acting as activators and/or inhibitors of healing” experiments or perturbing cell adhesion and angiogenesis. The purpose of this review is to provide an associated molecule functions. -

Ubiquitin-Mediated Proteolysis the Nobel Prize in Chemistry for 2004 Is
Advanced information on the Nobel Prize in Chemistry, 6 October 2004 Information Department, P.O. Box 50005, SE-104 05 Stockholm, Sweden Phone: +46 8 673 95 00, Fax: +46 8 15 56 70, E-mail: [email protected], Website: www.kva.se Ubiquitin-mediated proteolysis The Nobel Prize in Chemistry for 2004 is shared between three scientists who have made fundamental discoveries concerning how cells regulate the breakdown of intracellular proteins with extreme specificity as to target, time and space. Aaron Ciechanover, Avram Hershko and Irwin Rose together discovered ubiquitin- mediated proteolysis, a process where an enzyme system tags unwanted proteins with many molecules of the 76-amino acid residue protein ubiquitin. The tagged proteins are then transported to the proteasome, a large multisubunit protease complex, where they are degraded. Numerous cellular processes regulated by ubiquitin-mediated proteolysis include the cell cycle, DNA repair and transcription, protein quality control and the immune response. Defects in this proteolysis have a causal role in many human diseases, including a variety of cancers. Fig. 1 Ubiquitin-mediated proteolysis and its many biological functions 2 Introduction Eukaryotic cells, from yeast to human, contain some 6000 to 30000 protein-encoding genes and at least as many proteins. While much attention and research had been devoted to how proteins are synthesized, the reverse process, i.e. how proteins are degraded, long received little attention. A pioneer in this field was Schoenheimer, who in 1942 published results from isotope tracer techniques indicating that proteins in animals are continuously synthesized and degraded and therefore are in a dynamic state (Schoenheimer, 1942). -

Secreted Metalloproteinase ADAMTS-3 Inactivates Reelin
The Journal of Neuroscience, March 22, 2017 • 37(12):3181–3191 • 3181 Cellular/Molecular Secreted Metalloproteinase ADAMTS-3 Inactivates Reelin Himari Ogino,1* Arisa Hisanaga,1* XTakao Kohno,1 Yuta Kondo,1 Kyoko Okumura,1 Takana Kamei,1 Tempei Sato,2 Hiroshi Asahara,2 Hitomi Tsuiji,1 Masaki Fukata,3 and Mitsuharu Hattori1 1Department of Biomedical Science, Graduate School of Pharmaceutical Sciences, Nagoya City University, Nagoya, Aichi 467-8603, Japan, 2Department of Systems BioMedicine, Graduate School of Medical and Dental Sciences, Tokyo Medical and Dental University, Tokyo 113-8510, Japan, and 3Division of Membrane Physiology, Department of Molecular and Cellular Physiology, National Institute for Physiological Sciences, National Institutes of Natural Sciences, Okazaki, Aichi 444-8787, Japan The secreted glycoprotein Reelin regulates embryonic brain development and adult brain functions. It has been suggested that reduced Reelin activity contributes to the pathogenesis of several neuropsychiatric and neurodegenerative disorders, such as schizophrenia and Alzheimer’s disease; however, noninvasive methods that can upregulate Reelin activity in vivo have yet to be developed. We previously found that the proteolytic cleavage of Reelin within Reelin repeat 3 (N-t site) abolishes Reelin activity in vitro, but it remains controversial as to whether this effect occurs in vivo. Here we partially purified the enzyme that mediates the N-t cleavage of Reelin from the culture supernatant of cerebral cortical neurons. This enzyme was identified as a disintegrin and metalloproteinase with thrombospondin motifs-3 (ADAMTS-3). Recombinant ADAMTS-3 cleaved Reelin at the N-t site. ADAMTS-3 was expressed in excitatory neurons in the cerebral cortex and hippocampus. -

Data Sheet Cathepsin V Inhibitor Screening Assay Kit Catalog #79589 Size: 96 Reactions
6405 Mira Mesa Blvd Ste. 100 San Diego, CA 92121 Tel: 1.858.202.1401 Fax: 1.858.481.8694 Email: [email protected] Data Sheet Cathepsin V Inhibitor Screening Assay Kit Catalog #79589 Size: 96 reactions BACKGROUND: Cathepsin V, also called Cathepsin L2, is a lysosomal cysteine endopeptidase with high sequence homology to cathepsin L and other members of the papain superfamily of cysteine proteinases. Its expression is regulated in a tissue-specific manner and is high in thymus, testis and cornea. Expression analysis of cathepsin V in human tumors revealed widespread expression in colorectal and breast carcinomas, suggesting a possible role in tumor processes. DESCRIPTION: The Cathepsin V Inhibitor Screening Assay Kit is designed to measure the protease activity of Cathepsin V for screening and profiling applications. The Cathepsin V assay kit comes in a convenient 96-well format, with purified Cathepsin V, its fluorogenic substrate, and Cathepsin buffer for 100 enzyme reactions. In addition, the kit includes the cathepsin inhibitor E- 64 for use as a control inhibitor. COMPONENTS: Catalog # Component Amount Storage 80009 Cathepsin V 10 µg -80 °C Fluorogenic Cathepsin Avoid 80349 Substrate 1 (5 mM) 10 µl -20 °C multiple *4X Cathepsin buffer 2 ml -20 °C freeze/thaw E-64 (1 mM) 10 µl -20 °C cycles! 96-well black microplate * Add 120 µl of 0.5 M DTT before use. MATERIALS OR INSTRUMENTS REQUIRED BUT NOT SUPPLIED: 0.5 M DTT in aqueous solution Adjustable micropipettor and sterile tips Fluorescent microplate reader APPLICATIONS: Great for studying enzyme kinetics and screening small molecular inhibitors for drug discovery and HTS applications. -
What Is a Protease?
What is a Protease? Proteases (or peptidases) are enzymes secreted by animals for a number of physiological processes, among which is the Dr. Rolando A. Valientes digestion of feed protein. Regional Category Manager- Animals normally secrete suffi - Eubiotics / RONOZYME ProAct cient amount of enzymes to ade - Asia Pacific, DSM quately digest enough of their [email protected] feed so that they grow and remain healthy under normal conditions, such as those found in the wild. Any increased needs for protein (amino acids) for more rapid growth due to improved genetics has been traditionally met by adding more protein (or synthetic amino acids) into the feed. This was facilitated by a relative low cost for most protein-rich ingredi - Dr. Katrine Pontoppidan ents, such as soybean meal, and Research Scientist synthetic amino acids, such as Novozymes L-lysine HCL. Thus, an exogenous [email protected] protease (as a feed supplement) was not considered essential; that is, until recently. size, the rate of passage of feed Today we face not only the prob - through the digestive tract, the lem of feeding animals of continu - age of the animal, and its physio - ously increasing genetic potential logical/health condition. All of (this requires diets increasingly these variables are rather difficult richer in amino acids), but also an to control, but supplementing unprecedented rise in ingredient animal feeds with extra enzymes prices, leaving very small (if any) is rather easy if it can be done in a margin for profitability. Thus, it profitable way. has been deemed essential to seek ways to improve the nutritive Up until recently, any protease value of existing ingredients activity in commercial enzyme reducing feed cost. -

Felipe Jun Fuzita Molecular Physiology of Digestion In
FELIPE JUN FUZITA MOLECULAR PHYSIOLOGY OF DIGESTION IN ARACHNIDA: FUNCTIONAL AND COMPARATIVE-EVOLUTIONARY APPROACHES Thesis presented to the Programa de Pós-Graduação Interunidades em Biotecnologia USP/Instituto Butantan/IPT, to obtain the Title of Doctor in Biotechnology. São Paulo 2014 FELIPE JUN FUZITA MOLECULAR PHYSIOLOGY OF DIGESTION IN ARACHNIDA: FUNCTIONAL AND COMPARATIVE-EVOLUTIONARY APPROACHES Thesis presented to the Programa de Pós-Graduação Interunidades em Biotecnologia USP/Instituto Butantan/IPT, to obtain the Title of Doctor in Biotechnology. Concentration area: Biotechnology Advisor: Dr. Adriana Rios Lopes Rocha Corrected version. The original electronic version is available either in the library of the Institute of Biomedical Sciences and in the Digital Library of Theses and Dissertations of the University of Sao Paulo (BDTD). São Paulo 2014 DADOS DE CATALOGAÇÃO NA PUBLICAÇÃO (CIP) Serviço de Biblioteca e Informação Biomédica do Instituto de Ciências Biomédicas da Universidade de São Paulo © reprodução total Fuzita, Felipe Jun. Molecular physiology of digestion in Arachnida: functional and comparative-evolutionary approaches / Felipe Jun Fuzita. -- São Paulo, 2014. Orientador: Profa. Dra. Adriana Rios Lopes Rocha. Tese (Doutorado) – Universidade de São Paulo. Instituto de Ciências Biomédicas. Programa de Pós-Graduação Interunidades em Biotecnologia USP/IPT/Instituto Butantan. Área de concentração: Biotecnologia. Linha de pesquisa: Bioquímica, biologia molecular, espectrometria de massa. Versão do título para o português: Fisiologia molecular da digestão em Arachnida: abordagens funcional e comparativo-evolutiva. 1. Digestão 2. Aranha 3. Escorpião 4. Enzimologia 5. Proteoma 6. Transcriptoma I. Rocha, Profa. Dra. Adriana Rios Lopes I. Universidade de São Paulo. Instituto de Ciências Biomédicas. Programa de Pós-Graduação Interunidades em Biotecnologia USP/IPT/Instituto Butantan III. -

Effect of Nanoparticles on the Expression and Activity of Matrix Metalloproteinases
Nanotechnol Rev 2018; 7(6): 541–553 Review Magdalena Matysiak-Kucharek*, Magdalena Czajka, Krzysztof Sawicki, Marcin Kruszewski and Lucyna Kapka-Skrzypczak Effect of nanoparticles on the expression and activity of matrix metalloproteinases https://doi.org/10.1515/ntrev-2018-0110 Received September 14, 2018; accepted October 11, 2018; previously 1 Introduction published online November 15, 2018 Matrix metallopeptidases, commonly known as matrix Abstract: Matrix metallopeptidases, commonly known metalloproteinases (MMPs), are zinc-dependent proteo- as matrix metalloproteinases (MMPs), are a group of pro- lytic enzymes whose primary function is the degradation teolytic enzymes whose main function is the remodeling and remodeling of extracellular matrix (ECM) compo- of the extracellular matrix. Changes in the activity of nents. ECM is a complex, dynamic structure that condi- these enzymes are observed in many pathological states, tions the proper tissue architecture. MMPs by digesting including cancer metastases. An increasing body of evi- ECM proteins eliminate structural barriers and allow dence indicates that nanoparticles (NPs) can lead to the cell migration. Moreover, by hydrolyzing extracellularly deregulation of MMP expression and/or activity both in released proteins, MMPs can change the activity of many vitro and in vivo. In this work, we summarized the current signal peptides, such as growth factors, cytokines, and state of knowledge on the impact of NPs on MMPs. The chemokines. MMPs are involved in many physiological literature analysis showed that the impact of NPs on MMP processes, such as embryogenesis, reproduction cycle, or expression and/or activity is inconclusive. NPs exhibit wound healing; however, their increased activity is also both stimulating and inhibitory effects, which might be associated with a number of pathological conditions, such dependent on multiple factors, such as NP size and coat- as diabetes, cardiovascular diseases and neurodegenera- ing or a cellular model used in the research. -

The Involvement of Cysteine Proteases and Protease Inhibitor Genes in the Regulation of Programmed Cell Death in Plants
The Plant Cell, Vol. 11, 431–443, March 1999, www.plantcell.org © 1999 American Society of Plant Physiologists The Involvement of Cysteine Proteases and Protease Inhibitor Genes in the Regulation of Programmed Cell Death in Plants Mazal Solomon,a,1 Beatrice Belenghi,a,1 Massimo Delledonne,b Ester Menachem,a and Alex Levine a,2 a Department of Plant Sciences, Hebrew University of Jerusalem, Givat-Ram, Jerusalem 91904, Israel b Istituto di Genetica, Università Cattolica S.C., Piacenza, Italy Programmed cell death (PCD) is a process by which cells in many organisms die. The basic morphological and bio- chemical features of PCD are conserved between the animal and plant kingdoms. Cysteine proteases have emerged as key enzymes in the regulation of animal PCD. Here, we show that in soybean cells, PCD-activating oxidative stress in- duced a set of cysteine proteases. The activation of one or more of the cysteine proteases was instrumental in the PCD of soybean cells. Inhibition of the cysteine proteases by ectopic expression of cystatin, an endogenous cysteine pro- tease inhibitor gene, inhibited induced cysteine protease activity and blocked PCD triggered either by an avirulent strain of Pseudomonas syringae pv glycinea or directly by oxidative stress. Similar expression of serine protease inhib- itors was ineffective. A glutathione S-transferase–cystatin fusion protein was used to purify and characterize the in- duced proteases. Taken together, our results suggest that plant PCD can be regulated by activity poised between the cysteine proteases and the cysteine protease inhibitors. We also propose a new role for proteinase inhibitor genes as modulators of PCD in plants. -
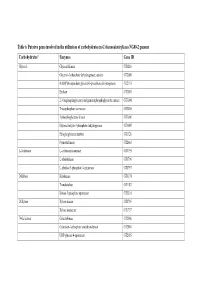
Table 6. Putative Genes Involved in the Utilization of Carbohydrates in G
Table 6. Putative genes involved in the utilization of carbohydrates in G. thermodenitrificans NG80-2 genome Carbohydrates* Enzymes Gene ID Glycerol Glycerol Kinase GT1216 Glycerol-3-phosphate dehydrogenase, aerobic GT2089 NAD(P)H-dependent glycerol-3-phosphate dehydrogenase GT2153 Enolase GT3003 2,3-bisphosphoglycerate-independentphosphoglycerate mutase GT3004 Triosephosphate isomerase GT3005 3-phosphoglycerate kinase GT3006 Glyceraldehyde-3-phosphate dehydrogenase GT3007 Phosphoglycerate mutase GT1326 Pyruvate kinase GT2663 L-Arabinose L-arabinose isomerase GT1795 L-ribulokinase GT1796 L-ribulose 5-phosphate 4-epimerase GT1797 D-Ribose Ribokinase GT3174 Transketolase GT1187 Ribose 5-phosphate epimerase GT3316 D-Xylose Xylose kinase GT1756 Xylose isomerase GT1757 D-Galactose Galactokinase GT2086 Galactose-1-phosphate uridyltransferase GT2084 UDP-glucose 4-epimerase GT2085 Carbohydrates* Enzymes Gene ID D-Fructose 1-phosphofructokinase GT1727 Fructose-1,6-bisphosphate aldolase GT1805 Fructose-1,6-bisphosphate aldolase type II GT3331 Triosephosphate isomerase GT3005 D-Mannose Mannnose-6 phospate isomelase GT3398 6-phospho-1-fructokinase GT2664 D-Mannitol Mannitol-1-phosphate dehydrogenase GT1844 N-Acetylglucosamine N-acetylglucosamine-6-phosphate deacetylase GT2205 N-acetylglucosamine-6-phosphate isomerase GT2204 D-Maltose Alpha-1,4-glucosidase GT0528, GT1643 Sucrose Sucrose phosphorylase GT3215 D-Trehalose Alpha-glucosidase GT1643 Glucose kinase GT2381 Inositol Myo-inositol catabolism protein iolC;5-dehydro-2- GT1807 deoxygluconokinase -

Insights Into Clpxp Proteolysis: Heterooligomerization and Partial Deactivation Cite This: Chem
Chemical Science View Article Online EDGE ARTICLE View Journal | View Issue Insights into ClpXP proteolysis: heterooligomerization and partial deactivation Cite this: Chem. Sci.,2017,8,1592 enhance chaperone affinity and substrate turnover in Listeria monocytogenes† a a a b a Dora´ Balogh,‡ Maria Dahmen,‡ Matthias Stahl, Marcin Poreba, Malte Gersch,§ Marcin Dragb and Stephan A. Sieber*a Caseinolytic proteases (ClpP) are important for recognition and controlled degradation of damaged proteins. While the majority of bacterial organisms utilize only a single ClpP, Listeria monocytogenes expresses two isoforms (LmClpP1 and LmClpP2). LmClpPs assemble into either a LmClpP2 homocomplex or a LmClpP1/2 heterooligomeric complex. The heterocomplex in association with the chaperone ClpX, exhibits a boost in proteolytic activity for unknown reasons. Here, we use a combined chemical and biochemical strategy to unravel two activation principles of LmClpPs. First, determination Creative Commons Attribution 3.0 Unported Licence. of apparent affinity constants revealed a 7-fold elevated binding affinity between the LmClpP1/2 heterocomplex and ClpX, compared to homooligomeric LmClpP2. This tighter interaction favors the formation of the proteolytically active complex between LmClpX and LmClpP1/2 and thereby accelerating the overall turnover. Second, screening a diverse library of fluorescent labeled peptides and proteins with various ClpP mutants allowed the individual analysis of substrate preferences for both isoforms within the heterocomplex. In addition to Leu and Met, LmClpP2 preferred a long aliphatic chain (2-Aoc) in the P1 position for cleavage. Strikingly, design and synthesis of a corresponding 2-Aoc chloromethyl ketone inhibitor resulted in stimulation of proteolysis by 160% when LmClpP2 was partially Received 2nd August 2016 This article is licensed under a alkylated on 20% of the active sites.