ARP36414 P050) Data Sheet
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supplementary File 2A Revised
Supplementary file 2A. Differentially expressed genes in aldosteronomas compared to all other samples, ranked according to statistical significance. Missing values were not allowed in aldosteronomas, but to a maximum of five in the other samples. Acc UGCluster Name Symbol log Fold Change P - Value Adj. P-Value B R99527 Hs.8162 Hypothetical protein MGC39372 MGC39372 2,17 6,3E-09 5,1E-05 10,2 AA398335 Hs.10414 Kelch domain containing 8A KLHDC8A 2,26 1,2E-08 5,1E-05 9,56 AA441933 Hs.519075 Leiomodin 1 (smooth muscle) LMOD1 2,33 1,3E-08 5,1E-05 9,54 AA630120 Hs.78781 Vascular endothelial growth factor B VEGFB 1,24 1,1E-07 2,9E-04 7,59 R07846 Data not found 3,71 1,2E-07 2,9E-04 7,49 W92795 Hs.434386 Hypothetical protein LOC201229 LOC201229 1,55 2,0E-07 4,0E-04 7,03 AA454564 Hs.323396 Family with sequence similarity 54, member B FAM54B 1,25 3,0E-07 5,2E-04 6,65 AA775249 Hs.513633 G protein-coupled receptor 56 GPR56 -1,63 4,3E-07 6,4E-04 6,33 AA012822 Hs.713814 Oxysterol bining protein OSBP 1,35 5,3E-07 7,1E-04 6,14 R45592 Hs.655271 Regulating synaptic membrane exocytosis 2 RIMS2 2,51 5,9E-07 7,1E-04 6,04 AA282936 Hs.240 M-phase phosphoprotein 1 MPHOSPH -1,40 8,1E-07 8,9E-04 5,74 N34945 Hs.234898 Acetyl-Coenzyme A carboxylase beta ACACB 0,87 9,7E-07 9,8E-04 5,58 R07322 Hs.464137 Acyl-Coenzyme A oxidase 1, palmitoyl ACOX1 0,82 1,3E-06 1,2E-03 5,35 R77144 Hs.488835 Transmembrane protein 120A TMEM120A 1,55 1,7E-06 1,4E-03 5,07 H68542 Hs.420009 Transcribed locus 1,07 1,7E-06 1,4E-03 5,06 AA410184 Hs.696454 PBX/knotted 1 homeobox 2 PKNOX2 1,78 2,0E-06 -

Aneuploidy: Using Genetic Instability to Preserve a Haploid Genome?
Health Science Campus FINAL APPROVAL OF DISSERTATION Doctor of Philosophy in Biomedical Science (Cancer Biology) Aneuploidy: Using genetic instability to preserve a haploid genome? Submitted by: Ramona Ramdath In partial fulfillment of the requirements for the degree of Doctor of Philosophy in Biomedical Science Examination Committee Signature/Date Major Advisor: David Allison, M.D., Ph.D. Academic James Trempe, Ph.D. Advisory Committee: David Giovanucci, Ph.D. Randall Ruch, Ph.D. Ronald Mellgren, Ph.D. Senior Associate Dean College of Graduate Studies Michael S. Bisesi, Ph.D. Date of Defense: April 10, 2009 Aneuploidy: Using genetic instability to preserve a haploid genome? Ramona Ramdath University of Toledo, Health Science Campus 2009 Dedication I dedicate this dissertation to my grandfather who died of lung cancer two years ago, but who always instilled in us the value and importance of education. And to my mom and sister, both of whom have been pillars of support and stimulating conversations. To my sister, Rehanna, especially- I hope this inspires you to achieve all that you want to in life, academically and otherwise. ii Acknowledgements As we go through these academic journeys, there are so many along the way that make an impact not only on our work, but on our lives as well, and I would like to say a heartfelt thank you to all of those people: My Committee members- Dr. James Trempe, Dr. David Giovanucchi, Dr. Ronald Mellgren and Dr. Randall Ruch for their guidance, suggestions, support and confidence in me. My major advisor- Dr. David Allison, for his constructive criticism and positive reinforcement. -

Lineage-Specific Programming Target Genes Defines Potential for Th1 Temporal Induction Pattern of STAT4
Downloaded from http://www.jimmunol.org/ by guest on October 1, 2021 is online at: average * The Journal of Immunology published online 26 August 2009 from submission to initial decision 4 weeks from acceptance to publication J Immunol http://www.jimmunol.org/content/early/2009/08/26/jimmuno l.0901411 Temporal Induction Pattern of STAT4 Target Genes Defines Potential for Th1 Lineage-Specific Programming Seth R. Good, Vivian T. Thieu, Anubhav N. Mathur, Qing Yu, Gretta L. Stritesky, Norman Yeh, John T. O'Malley, Narayanan B. Perumal and Mark H. Kaplan Submit online. Every submission reviewed by practicing scientists ? is published twice each month by http://jimmunol.org/subscription Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts http://www.jimmunol.org/content/suppl/2009/08/26/jimmunol.090141 1.DC1 Information about subscribing to The JI No Triage! Fast Publication! Rapid Reviews! 30 days* • Why • • Material Permissions Email Alerts Subscription Supplementary The Journal of Immunology The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2009 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. This information is current as of October 1, 2021. Published August 26, 2009, doi:10.4049/jimmunol.0901411 The Journal of Immunology Temporal Induction Pattern of STAT4 Target Genes Defines Potential for Th1 Lineage-Specific Programming1 Seth R. Good,2* Vivian T. Thieu,2† Anubhav N. Mathur,† Qing Yu,† Gretta L. -

Next Generation Sequencing by Multiple Samples Pooling Reveals
Next generation sequencing of pooled samples reveals new SNRNP200 mutations associated with retinitis pigmentosa Paola Benaglio1, Terri L. McGee2, Leonardo P. Capelli1,3, Shyana Harper2, Eliot L. Berson2 and Carlo Rivolta1 1Department of Medical Genetics, University of Lausanne, Lausanne, Switzerland 2The Berman-Gund Laboratory for the Study of Retinal Degenerations, Harvard Medical School, Massachusetts Eye and Ear Infirmary, Boston, Massachusetts, USA 3Department of Genetics and Evolutionary Biology, Institute of Biosciences, University of São Paulo, São Paulo, Brazil Correspondence to: Carlo Rivolta Department of Medical Genetics University of Lausanne Rue du Bugnon 27 1005 Lausanne Switzerland; Phone: +41(21) 692-5451 FAX: +41(21) 692-5455 email: [email protected] 1 ABSTRACT The gene SNRNP200 is composed of 45-exons and encodes a protein essential for pre- mRNA splicing, the 200 kDa helicase hBrr2. Despite the fact that complete lack of this protein is incompatible with cell survival, two independent heterozygous mutations in SNRNP200 have recently been found to be associated with the retinal degenerative disease retinitis pigmentosa (RP) in two families from China. In this work the entire 35-Kb SNRNP200 genomic region was analyzed in a cohort of 96 unrelated North American patients with autosomal dominant RP. To carry out this large-scale sequencing project, we performed ultra high-throughput sequencing of pooled, untagged PCR products and verified the presence of the detected DNA changes by Sanger sequencing of individual samples. One of the two previously known mutations (p.S1087L) was identified in 3 patients, and 4 new missense changes (p.R681C, p.R681H, p.V683L, p.Y689C) affecting highly conserved codons were identified in 6 unrelated individuals. -

Mechanism of Regulation of Spliceosome Activation by Brr2 And
I Mechanism of regulation of spliceosome activation by Brr2 and Prp8 and links to retinal disease Dissertation for the award of the degree “Doctor of Philosophy” (Ph.D.) in the Molecular Biology Program Division of Mathematics and Natural Sciences of the Georg-August-Universität Göttingen submitted by Sina Mozaffari-Jovin born in Sabzevar, Iran Göttingen 2012 II Members of the thesis committee: Prof. Dr. Reinhard Lührmann (Reviewer) Department of Cellular Biochemistry, Max Planck Institute for Biophysical Chemistry, Göttingen Prof. Dr. Reinhard Jahn Department of Neurobiology, Max Planck Institute for Biophysical Chemistry, Göttingen Prof. Dr. Ralf Ficner Department of Molecular Structural Biology, Institute for Microbiology and Genetics, Göttingen Date of submission of Thesis: December, 14th, 2012 III Affidavit I declare that my Ph.D. thesis entitled “Mechanism of regulation of spliceosome activation by Brr2 and Prp8 and links to retinal disease” has been written independently and with no other sources and aids than quoted. Sina Mozaffari-Jovin Göttingen, 2012 IV “The knowledge of anything, since all things have causes, is not acquired or complete unless it is known by its causes” Avicenna (Father of Modern Medicine; c. 980- June 1037) V Table of Contents 1 Abstract ......................................................................................................................... 1 2 Introduction .................................................................................................................. 4 2.1 The chemistry -

Open Data for Differential Network Analysis in Glioma
International Journal of Molecular Sciences Article Open Data for Differential Network Analysis in Glioma , Claire Jean-Quartier * y , Fleur Jeanquartier y and Andreas Holzinger Holzinger Group HCI-KDD, Institute for Medical Informatics, Statistics and Documentation, Medical University Graz, Auenbruggerplatz 2/V, 8036 Graz, Austria; [email protected] (F.J.); [email protected] (A.H.) * Correspondence: [email protected] These authors contributed equally to this work. y Received: 27 October 2019; Accepted: 3 January 2020; Published: 15 January 2020 Abstract: The complexity of cancer diseases demands bioinformatic techniques and translational research based on big data and personalized medicine. Open data enables researchers to accelerate cancer studies, save resources and foster collaboration. Several tools and programming approaches are available for analyzing data, including annotation, clustering, comparison and extrapolation, merging, enrichment, functional association and statistics. We exploit openly available data via cancer gene expression analysis, we apply refinement as well as enrichment analysis via gene ontology and conclude with graph-based visualization of involved protein interaction networks as a basis for signaling. The different databases allowed for the construction of huge networks or specified ones consisting of high-confidence interactions only. Several genes associated to glioma were isolated via a network analysis from top hub nodes as well as from an outlier analysis. The latter approach highlights a mitogen-activated protein kinase next to a member of histondeacetylases and a protein phosphatase as genes uncommonly associated with glioma. Cluster analysis from top hub nodes lists several identified glioma-associated gene products to function within protein complexes, including epidermal growth factors as well as cell cycle proteins or RAS proto-oncogenes. -

Analysis of RP2 and RPGR Mutations in Five X-Linked Chinese Families
www.nature.com/scientificreports OPEN Analysis of RP2 and RPGR Mutations in Five X-Linked Chinese Families with Retinitis Pigmentosa Received: 07 December 2016 Jingjing Jiang1,*, Xiaofei Wu2,*, Di Shen3,*, Lijin Dong4, Xiaodong Jiao5, J. Fielding Hejtmancik5 Accepted: 08 February 2017 & Ningdong Li1,2,5 Published: 15 March 2017 Mutations in RP2 and RPGR genes are responsible for the X-linked retinitis pigmentosa (XLRP). In this study, we analyzed the RP2 and RPGR gene mutations in five Han Chinese families with XLRP. An approximately 17Kb large deletion including the exon 4 and exon 5 of RP2 gene was found in an XLRP family. In addition, four frameshift mutations including three novel mutations of c.1059 + 1 G > T, c.2002dupC and c.2236_2237del CT, as well as a previously reported mutation of c.2899delG were detected in the RPGR gene in the other four families. Our study further expands the mutation spectrum of RP2 and RPGR, and will be helpful for further study molecular pathogenesis of XLRP. Retinitis pigmentosa (RP) is a clinically and genetically heterogeneous group of retinal dystrophies characterized by photoreceptor cell degeneration. The prevalence of RP is about 1/4000 ~1/5000 according to studies in dif- ferent populations1. The clinical features comprise night blindness, progressive constriction of visual field with age, and fundus changes including attenuation of the retinal arterioles, bone spicule-like pigment deposits in the mid-peripheral retinal and waxy pallor of the optic disc. Affected individuals often have severely abnormal or non-detectable rod responses in the electroretinograms (ERG) recordings even in the early stage of the disease2. -

Supplementary Figures and Table
SUPPLEMENTARY DATA Supplementary Figure 1. ©2014 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db141 -0066/-/DC1 SUPPLEMENTARY DATA Supplementary Figure 2. ©2014 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db142 -0066/-/DC1 SUPPLEMENTARY DATA -/- Supplementary Table 1. Fold increase of Ser/Thr/Tyr phosphorylation in livers of MKP-3 male mice versus wild type male mice fed on a high fat diet (n=5 for each group). Symbol Name Phosphorylation KO/WT ratio Q Value sites Apoptosis ACIN1 Acin1 protein S64 11.4 0.02 T66 8.3 0.02 API5 Apoptosis inhibitor 5 S461 2.2 0.03 S462 1.8 0.03 AIFM3 Apoptosis-inducing factor 3 S30 7.4 0.03 TP53BP2 Apoptosis-stimulating of p53 protein 2 S479 3.7 0.02 ACIN1 Apoptotic chromatin condensation inducer S64S70 5.7 0.02 1 S208 7.1 0.02 S210 7.0 0.02 S479S482S491 105.7 0.03 S729 2.8 0.02 PEA15 Astrocytic phosphoprotein PEA-15 S116 10.8 0.02 BAG3 BAG family molecular chaperone regulator S179 3.3 0.02 3 S353S357 2.3 0.03 S360 2.3 0.03 S390 8.4 0.02 BNIP2 BCL2/adenovirus E1B 19 kDa-interacting S114 3.9 0.02 protein 2 alpha BNIP3 BCL2/adenovirus E1B 19 kDa protein- S60 19.8 0.03 interacting protein 3 S85T86 14.5 0.02 S88 6.1 0.02 BCL2L13 Bcl-2-like protein 13 S387 4.0 0.02 T389 3.1 0.02 CAAP1 Caspase activity and apoptosis inhibitor S183 2.3 0.03 CARD6 Card6 caspase recruitment domain family, S809 3.6 0.03 member 6 CASP8 Caspase-8 S188 2.2 0.02 DAP Death-associated protein S51 5.4 0.02 DAPK2 Death-associated protein kinase 2 S299 3.8 0.02 S349 3.5 0.02 FAF1 FAS-associated factor 1 S269 17.1 0.04 GAS2 Growth arrest-specific protein 2 T282 5.3 0.02 S283 7.4 0.02 S287 5.3 0.02 S289 7.4 0.02 GCH1 GTP cyclohydrolase 1 S24 3.9 0.02 HTT Huntingtin S398S409S411 9.7 0.02 KRT18 Keratin, type I cytoskeletal 18 T9 2.7 0.02 S31S32S35 2.8 0.02 S43S45 3.1 0.02 PDCD5 MCG128907 S119 10.7 0.02 Y126 4.0 0.02 BNIP3I MCG2480, isoform CRA_b S61S62 12.9 0.03 S63S64 8.1 0.02 ©2014 American Diabetes Association. -

Vaccine-Increased Seq ID Unigene ID Uniprot ID Gene Names
BMJ Publishing Group Limited (BMJ) disclaims all liability and responsibility arising from any reliance Supplemental material placed on this supplemental material which has been supplied by the author(s) J Immunother Cancer Vaccine-Increased Seq ID Unigene ID Uniprot ID Gene Names 1_HSPA1A_3303 Hs.274402 P0DMV8 HSPA1A HSP72 HSPA1 HSX70 100_AKAP17A_8227 Hs.522572 Q02040 AKAP17A CXYorf3 DXYS155E SFRS17A XE7 1000_H2AFY_9555 Hs.420272 O75367 H2AFY MACROH2A1 1001_ITPK1_3705 Hs.308122 Q13572 ITPK1 1002_PTPN11_5781 Hs.506852 Q06124 PTPN11 PTP2C SHPTP2 1003_EIF3J_8669 Hs.404056 O75822 EIF3J EIF3S1 PRO0391 1004_TRIP12_9320 Hs.591633 Q14669 TRIP12 KIAA0045 ULF 1006_YEATS2_55689 Hs.632575 Q9ULM3 YEATS2 KIAA1197 1007_SEL1L3_23231 Hs.479384 Q68CR1 SEL1L3 KIAA0746 1008_IDH1_3417 Hs.593422 O75874 IDH1 PICD 101_HSPH1_10808 Hs.36927 Q92598 HSPH1 HSP105 HSP110 KIAA0201 1010_LDLR_3949 Hs.213289 P01130 LDLR 1011_FAM129B_64855 Hs.522401 Q96TA1 NIBAN2 C9orf88 FAM129B 1012_MAP3K5_4217 Hs.186486 Q99683 MAP3K5 ASK1 MAPKKK5 MEKK5 1013_NEFH_4744 Hs.198760 P12036 NEFH KIAA0845 NFH 1014_RAP1B_5908 Hs.369920 P61224 RAP1B OK/SW-cl.11 1015_MCCC1_56922 Hs.47649 Q96RQ3 MCCC1 MCCA 1017_MT1E_4493 Hs.534330 P04732 MT1E 1022_TXNDC5_81567 Hs.150837 Q8NBS9 TXNDC5 TLP46 UNQ364/PRO700 1023_STRA13_201254 Hs.37616 O14503 BHLHE40 BHLHB2 DEC1 SHARP2 STRA13 1024_NPEPPS_9520 Hs.443837 P55786 NPEPPS PSA 1025_YIPF6_286451 Hs.82719 Q96EC8 YIPF6 1026_CLIP1_6249 Hs.524809 P30622 CLIP1 CYLN1 RSN 1027_SRSF7_6432 Hs.309090 Q16629 SRSF7 SFRS7 103_RPS25_6230 Hs.512676 P62851 RPS25 1031_SOCS7_30837 -
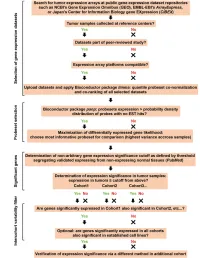
Supplementary Data
SUPPLEMENTAL INFORMATION A study restricted to chemokine receptors as well as a genome-wide transcript analysis uncovered CXCR4 as preferentially expressed in Ewing's sarcoma (Ewing's sarcoma) cells of metastatic origin (Figure 4). Transcriptome analyses showed that in addition to CXCR4, genes known to support cell motility and invasion topped the list of genes preferentially expressed in metastasis-derived cells (Figure 4D). These included kynurenine 3-monooxygenase (KMO), galectin-1 (LGALS1), gastrin-releasing peptide (GRP), procollagen C-endopeptidase enhancer (PCOLCE), and ephrin receptor B (EPHB3). KMO, a key enzyme of tryptophan catabolism, has not been linked to metastasis. Tryptophan and its catabolites, however, are involved in immune evasion by tumors, a process that can assist in tumor progression and metastasis (1). LGALS1, GRP, PCOLCE and EPHB3 have been linked to tumor progression and metastasis of several cancers (2-4). Top genes preferentially expressed in L-EDCL included genes that suppress cell motility and/or potentiate cell adhesion such as plakophilin 1 (PKP1), neuropeptide Y (NPY), or the metastasis suppressor TXNIP (5-7) (Figure 4D). Overall, L-EDCL were enriched in gene sets geared at optimizing nutrient transport and usage (Figure 4D; Supplementary Table 3), a state that may support the early stages of tumor growth. Once tumor growth outpaces nutrient and oxygen supplies, gene expression programs are usually switched to hypoxic response and neoangiogenesis, which ultimately lead to tumor egress and metastasis. Accordingly, gene sets involved in extracellular matrix remodeling, MAPK signaling, and response to hypoxia were up-regulated in M-EDCL (Figure 4D; Supplementary Table 4), consistent with their association to metastasis in other cancers (8, 9). -

Coexpression Networks Based on Natural Variation in Human Gene Expression at Baseline and Under Stress
University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations Fall 2010 Coexpression Networks Based on Natural Variation in Human Gene Expression at Baseline and Under Stress Renuka Nayak University of Pennsylvania, [email protected] Follow this and additional works at: https://repository.upenn.edu/edissertations Part of the Computational Biology Commons, and the Genomics Commons Recommended Citation Nayak, Renuka, "Coexpression Networks Based on Natural Variation in Human Gene Expression at Baseline and Under Stress" (2010). Publicly Accessible Penn Dissertations. 1559. https://repository.upenn.edu/edissertations/1559 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/edissertations/1559 For more information, please contact [email protected]. Coexpression Networks Based on Natural Variation in Human Gene Expression at Baseline and Under Stress Abstract Genes interact in networks to orchestrate cellular processes. Here, we used coexpression networks based on natural variation in gene expression to study the functions and interactions of human genes. We asked how these networks change in response to stress. First, we studied human coexpression networks at baseline. We constructed networks by identifying correlations in expression levels of 8.9 million gene pairs in immortalized B cells from 295 individuals comprising three independent samples. The resulting networks allowed us to infer interactions between biological processes. We used the network to predict the functions of poorly-characterized human genes, and provided some experimental support. Examining genes implicated in disease, we found that IFIH1, a diabetes susceptibility gene, interacts with YES1, which affects glucose transport. Genes predisposing to the same diseases are clustered non-randomly in the network, suggesting that the network may be used to identify candidate genes that influence disease susceptibility. -

Contribution of SNRNP200 Sequence Variations to Retinitis Pigmentosa
Eye (2013) 27, 1204–1213 & 2013 Macmillan Publishers Limited All rights reserved 0950-222X/13 www.nature.com/eye 1 1 1 1 LABORATORY STUDY Contribution X Zhang , TYY Lai , SWY Chiang , POS Tam , DTL Liu1,2, CKM Chan1,3, CP Pang1,2, of SNRNP200 C Zhao4 and LJ Chen1,2 sequence variations to retinitis pigmentosa Abstract mutations in two respective patients affirmed that SNRNP200 contributes to a proportion Purpose Mutations in the SNRNP200 gene of overall RP. have been reported to cause autosomal Eye (2013) 27, 1204–1213; doi:10.1038/eye.2013.137; dominant retinitis pigmentosa (adRP). In this published online 26 July 2013 study, we evaluate the mutation profile of SNRNP200 in a cohort of southern Chinese 1Department of Keywords: retinitis pigmentosa; SNRNP200; RP patients. Ophthalmology and Visual mutation Sciences, The Chinese Methods Twenty adRP patients from University of Hong Kong, 11 families and 165 index patients with Hong Kong, China non-syndromic RP with mixed inheritance patterns were screened for mutations in the Introduction 2Department of mutation hotspots of SNRNP200. These Ophthalmology and Visual Retinitis pigmentosa (RP; MIM268000; included exons 12–16, 22–32, and 38–45, Sciences, Prince of Wales Mendelian Inheritance in Man; National Center Hospital, The Chinese which covered the two helicase ATP-binding for Biotechnology Information, Bethesda, MD, University of Hong Kong, domains in DEAD-box and two sec-63 USA) refers to a clinically and genetically Hong Kong, China domains. The targeted regions were diverse group of diffuse retinal dystrophies. amplified by polymerase chain reaction and 3Hong Kong Eye Hospital, The prevalence of RP is about 1/4000 to 1/6000 analyzed by direct DNA sequencing, Hong Kong, China in certain populations1–3 with no significant followed by in silico analyses.