Cefepime Combined with Amoxicillin/Clavulanic Acid: a New Choice For
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 200327 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 200327 Priority or Standard Standard Submit Date(s) December 29, 2009 Received Date(s) December 30, 2009 PDUFA Goal Date October 30, 2010 Division / Office Division of Anti-Infective and Ophthalmology Products Office of Antimicrobial Products Reviewer Name(s) Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD Review Completion October 29, 2010 Date Established Name Ceftaroline fosamil for injection (Proposed) Trade Name Teflaro Therapeutic Class Cephalosporin; ß-lactams Applicant Cerexa, Inc. Forest Laboratories, Inc. Formulation(s) 400 mg/vial and 600 mg/vial Intravenous Dosing Regimen 600 mg every 12 hours by IV infusion Indication(s) Acute Bacterial Skin and Skin Structure Infection (ABSSSI); Community-acquired Bacterial Pneumonia (CABP) Intended Population(s) Adults ≥ 18 years of age Template Version: March 6, 2009 Reference ID: 2857265 Clinical Review Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD NDA 200327: Teflaro (ceftaroline fosamil) Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 9 1.1 Recommendation on Regulatory Action ........................................................... 10 1.2 Risk Benefit Assessment.................................................................................. 10 1.3 Recommendations for Postmarketing Risk Evaluation and Mitigation Strategies ........................................................................................................................ -

“Ceftriaxone– Sulbactam–EDTA” and Various Antibiotics Against Gram
ORIGINAL ARTICLE A Comparative In Vitro Sensitivity Study of “Ceftriaxone– Sulbactam–EDTA” and Various Antibiotics against Gram- negative Bacterial Isolates from Intensive Care Unit Sweta Singh1, Chinmoy Sahu2, Sangram Singh Patel3, Abhay Singh4, Nidhi Yaduvanshi5 ABSTRACT Introduction: A rapid increase in multidrug-resistant (MDR) strains is being seen across the globe especially in the Southeast Asian region, including India. Carbapenems and colistin form the mainstay of treatment against gram-negative pathogens, especially extended-spectrum beta-lactamase (ESBL)- and metallo-beta-lactamse (MBL)-producing isolates. However, due to increased resistance to carbapenems and toxicity of colistin, especially in intensive care units (ICUs), carbapenem-sparing antibiotics like ceftriaxone–sulbactam–EDTA (CSE) combination needs to be evaluated. Materials and methods: Bacterial isolates cultured from various clinical samples from all ICUs for a period of 9 months were evaluated. Bacterial identification was performed by matrix assisted laser desorption ionization time of flight mass spectrometry (MALDI-TOF MS) and antibiotic susceptibility testing were performed by disk diffusion and E test method. Antibiogram of various antibiotics was noted. Extended-spectrum beta-lactamase- and MBL-producing bacteria were identified by phenotypic methods. Antibiotic sensitivity results of CSE were compared with the comparator drugs like colistin, carbapenems, and tigecycline in Enterobacteriaceae, Acinetobacter spp., and Pseudomonas spp. along with ESBL and MBL producers. Results: A total of 2,760 samples of blood, cerebrospinal fluid (CSF), respiratory samples, tissue, and pus were collected from ICUs with maximum isolates from pus (37%) followed by respiratory samples (31%) and blood (27%). Escherichia coli and Klebsiella pneumoniae were the predominant gram-negative pathogens accounting for 56% of the isolates followed by Acinetobacter spp. -

Cefoperazone Sodium
Disclaimer While these labels are the most current Drug Control Authority (DCA) approved versions of content, these are not necessarily reflective of final printed labeling currently in distribution LPD Title : Cefoperazone Sodium LPD Date : 07 February 2017 Country : Malaysia Reference : CDS version 5.0 dated 07 January 2017 Reason : Addition of adverse drug reactions (ADRs) Haemorrhage and Coagulopathy to Section 4.8 Undesirable effects, and also, addition of serious hemorrhage and coagulopathy to the warning about vitamin K deficiency in Section 4.4 Special warnings and precautions for use Addition of ADRs Anaphylactic reaction and Anaphylactic shock to Section 4.8 Undesirable effects Addition of ADR Dermatitis exfoliative to Section 4.8 Undesirable Effects and a warning statement on severe cutaneous adverse reactions (SCARs) to Section 4.4, Special warnings and precautions for use 1. NAME OF THE MEDICINAL PRODUCT CEFOBID 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Cefoperazone sodium is a semisynthetic broad-spectrum cephalosporin antibiotic for parenteral use only. Cefoperazone contains 34 mg sodium (1.5 mEq) per gram. Cefoperazone is a white crystalline powder which is freely soluble in water. The pH of a 25% aqueous solution is 5.0 to 6.5 and the solution is colorless to straw-yellow, depending on the concentration. The empirical formula is C25H26N9NaO8S2. 3. PHARMACEUTICAL FORM Cefoperazone sodium is available in vials containing 1 and 2 grams. Not For Distribution PFIZER CONFIDENTIAL Not all product strengths or pack sizes -

Pharmacodynamic Evaluation of Suppression of in Vitro Resistance In
www.nature.com/scientificreports OPEN Pharmacodynamic evaluation of suppression of in vitro resistance in Acinetobacter baumannii strains using polymyxin B‑based combination therapy Nayara Helisandra Fedrigo1, Danielle Rosani Shinohara1, Josmar Mazucheli2, Sheila Alexandra Belini Nishiyama1, Floristher Elaine Carrara‑Marroni3, Frederico Severino Martins4, Peijuan Zhu5, Mingming Yu6, Sherwin Kenneth B. Sy2 & Maria Cristina Bronharo Tognim1* The emergence of polymyxin resistance in Gram‑negative bacteria infections has motivated the use of combination therapy. This study determined the mutant selection window (MSW) of polymyxin B alone and in combination with meropenem and fosfomycin against A. baumannii strains belonging to clonal lineages I and III. To evaluate the inhibition of in vitro drug resistance, we investigate the MSW‑derived pharmacodynamic indices associated with resistance to polymyxin B administrated regimens as monotherapy and combination therapy, such as the percentage of each dosage interval that free plasma concentration was within the MSW (%TMSW) and the percentage of each dosage interval that free plasma concentration exceeded the mutant prevention concentration (%T>MPC). The MSW of polymyxin B varied between 1 and 16 µg/mL for polymyxin B‑susceptible strains. The triple combination of polymyxin B with meropenem and fosfomycin inhibited the polymyxin B‑resistant subpopulation in meropenem‑resistant isolates and polymyxin B plus meropenem as a double combination sufciently inhibited meropenem‑intermediate, and susceptible strains. T>MPC 90% was reached for polymyxin B in these combinations, while %TMSW was 0 against all strains. TMSW for meropenem and fosfomycin were also reduced. Efective antimicrobial combinations signifcantly reduced MSW. The MSW‑derived pharmacodynamic indices can be used for the selection of efective combination regimen to combat the polymyxin B‑resistant strain. -

General Items
Essential Medicines List (EML) 2019 Application for the inclusion of imipenem/cilastatin, meropenem and amoxicillin/clavulanic acid in the WHO Model List of Essential Medicines, as reserve second-line drugs for the treatment of multidrug-resistant tuberculosis (complementary lists of anti-tuberculosis drugs for use in adults and children) General items 1. Summary statement of the proposal for inclusion, change or deletion This application concerns the updating of the forthcoming WHO Model List of Essential Medicines (EML) and WHO Model List of Essential Medicines for Children (EMLc) to include the following medicines: 1) Imipenem/cilastatin (Imp-Cln) to the main list but NOT the children’s list (it is already mentioned on both lists as an option in section 6.2.1 Beta Lactam medicines) 2) Meropenem (Mpm) to both the main and the children’s lists (it is already on the list as treatment for meningitis in section 6.2.1 Beta Lactam medicines) 3) Clavulanic acid to both the main and the children’s lists (it is already listed as amoxicillin/clavulanic acid (Amx-Clv), the only commercially available preparation of clavulanic acid, in section 6.2.1 Beta Lactam medicines) This application makes reference to amendments recommended in particular to section 6.2.4 Antituberculosis medicines in the latest editions of both the main EML (20th list) and the EMLc (6th list) released in 2017 (1),(2). On the basis of the most recent Guideline Development Group advising WHO on the revision of its guidelines for the treatment of multidrug- or rifampicin-resistant (MDR/RR-TB)(3), the applicant considers that the three agents concerned be viewed as essential medicines for these forms of TB in countries. -

Efficacy of Fosfomycin Trometamol and Cefuroxime Axetil in the Treatment of Kashmir, India Urinary Tract Infections During Pregnancy
International Journal of Surgery Science 2019; 3(2): 33-35 E-ISSN: 2616-3470 P-ISSN: 2616-3462 © Surgery Science Efficacy of fosfomycin trometamol and cefuroxime axetil www.surgeryscience.com 2019; 3(2): 33-35 in the treatment of UTI during pregnancy: A Received: 18-11-2018 Accepted: 22-12-2018 comparative study Dr. Robindera Kour In Charge Consultant Surgery, Dr. Robindera Kour, Dr. Gurpreet Kour, Dr. Iqbal Singh, Dr. KK Gupta Govt. Hospital Sarwal, Jammu, and Dr. Rajiv Sharma Jammu and Kashmir, India Dr. Gurpreet Kour DOI: https://doi.org/10.33545/surgery.2019.v3.i2a.373 Medical Officer, Acharya Shri Chander College of Medical Abstract Sciences, Jammu, Jammu and Objective: To compare the efficacy of fosfomycin trometamol and cefuroxime axetil in the treatment of Kashmir, India urinary tract infections during pregnancy. Methods: Prospective clinical study was conducted among pregnant women who were followed as part of Dr. Iqbal Singh routine prenatal care at Government Hospital Sarwal, Jammu, India with complaints of lower UTI were Assistant Professor, Public Health assessed. 60 patients were enrolled, divided into 2 equal groups for treatment with single-dose fosfomycin Dentistry, Indira Gandhi Government Dental College, trometamol and 5-day courses of cefuroxime axetil. Follow-up was carried out after 5 weeks. Jammu, Jammu and Kashmir, Results: The treatment groups did not differ significantly in terms of demographics, clinical compliance India rate and adverse effects. But the patients who were taking 5-day courses of cefuroxime axetil showed statistically significant higher cure rate as well as lower recurrence rate as compared to fosfomycin Dr. -

Severe Sepsis and Septic Shock Antibiotic Guide
Stanford Health Issue Date: 05/2017 Stanford Antimicrobial Safety and Sustainability Program Severe Sepsis and Septic Shock Antibiotic Guide Table 1: Antibiotic selection options for healthcare associated and/or immunocompromised patients • Healthcare associated: intravenous therapy, wound care, or intravenous chemotherapy within the prior 30 days, residence in a nursing home or other long-term care facility, hospitalization in an acute care hospital for two or more days within the prior 90 days, attendance at a hospital or hemodialysis clinic within the prior 30 days • Immunocompromised: Receiving chemotherapy, known systemic cancer not in remission, ANC <500, severe cell-mediated immune deficiency Table 2: Antibiotic selection options for community acquired, immunocompetent patients Table 3: Antibiotic selection options for patients with simple sepsis, community acquired, immunocompetent patients requiring hospitalization. Risk Factors for Select Organisms P. aeruginosa MRSA Invasive Candidiasis VRE (and other resistant GNR) Community acquired: • Known colonization with MDROs • Central venous catheter • Liver transplant • Prior IV antibiotics within 90 day • Recent MRSA infection • Broad-spectrum antibiotics • Known colonization • Known colonization with MDROs • Known MRSA colonization • + 1 of the following risk factors: • Prolonged broad antibacterial • Skin & Skin Structure and/or IV access site: ♦ Parenteral nutrition therapy Hospital acquired: ♦ Purulence ♦ Dialysis • Prolonged profound • Prior IV antibiotics within 90 days ♦ Abscess -

In Vitro Susceptibility of Multi-Drug Resistant Klebsiella Pneumoniae Strains Causing Nosocomial Infections to Fosfomycin. a Comparison of Determination Methods
pathogens Article In Vitro Susceptibility of Multi-Drug Resistant Klebsiella pneumoniae Strains Causing Nosocomial Infections to Fosfomycin. A Comparison of Determination Methods Beata M ˛aczy´nska 1,* , Justyna Paleczny 1 , Monika Oleksy-Wawrzyniak 1 , Irena Choroszy-Król 2 and Marzenna Bartoszewicz 1 1 Department of Pharmaceutical Microbiology and Parasitology, Faculty of Pharmacy, Medical University, 50-367 Wroclaw, Poland; [email protected] (J.P.); [email protected] (M.O.-W.); [email protected] (M.B.) 2 Department of Basic Sciences, Faculty of Health Sciences, Medical University, 50-367 Wroclaw, Poland; [email protected] * Correspondence: [email protected] Abstract: Introduction: Over the past few decades, Klebsiella pneumoniae strains increased their pathogenicity and antibiotic resistance, thereby becoming a major therapeutic challenge. One of the few available therapeutic options seems to be intravenous fosfomycin. Unfortunately, the determination of sensitivity to fosfomycin performed in hospital laboratories can pose a significant problem. Therefore, the aim of the present research was to evaluate the activity of fosfomycin against clinical, multidrug-resistant Klebsiella pneumoniae strains isolated from nosocomial infections between Citation: M ˛aczy´nska,B.; Paleczny, J.; 2011 and 2020, as well as to evaluate the methods routinely used in hospital laboratories to assess Oleksy-Wawrzyniak, M.; bacterial susceptibility to this antibiotic. Materials and Methods: 43 multidrug-resistant Klebsiella Choroszy-Król, I.; Bartoszewicz, M. strains isolates from various infections were tested. All the strains had ESBL enzymes, and 20 In Vitro Susceptibility of Multi-Drug also showed the presence of carbapenemases. Susceptibility was determined using the diffusion Resistant Klebsiella pneumoniae Strains method (E-test) and the automated system (Phoenix), which were compared with the reference Causing Nosocomial Infections to method (agar dilution). -
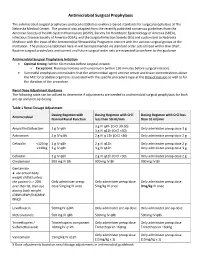
Antimicrobial Surgical Prophylaxis
Antimicrobial Surgical Prophylaxis The antimicrobial surgical prophylaxis protocol establishes evidence-based standards for surgical prophylaxis at The Nebraska Medical Center. The protocol was adapted from the recently published consensus guidelines from the American Society of Health-System Pharmacists (ASHP), Society for Healthcare Epidemiology of America (SHEA), Infectious Disease Society of America (IDSA), and the Surgical Infection Society (SIS) and customized to Nebraska Medicine with the input of the Antimicrobial Stewardship Program in concert with the various surgical groups at the institution. The protocol established here-in will be implemented via standard order sets utilized within One Chart. Routine surgical prophylaxis and current and future surgical order sets are expected to conform to this guidance. Antimicrobial Surgical Prophylaxis Initiation Optimal timing: Within 60 minutes before surgical incision o Exceptions: Fluoroquinolones and vancomycin (within 120 minutes before surgical incision) Successful prophylaxis necessitates that the antimicrobial agent achieve serum and tissue concentrations above the MIC for probable organisms associated with the specific procedure type at the time of incision as well as for the duration of the procedure. Renal Dose Adjustment Guidance The following table can be utilized to determine if adjustments are needed to antimicrobial surgical prophylaxis for both pre-op and post-op dosing. Table 1 Renal Dosage Adjustment Dosing Regimen with Dosing Regimen with CrCl Dosing Regimen with -

In Vitro Susceptibilities of Escherichia Coli and Klebsiella Spp. To
Jpn. J. Infect. Dis., 60, 227-229, 2007 Short Communication In Vitro Susceptibilities of Escherichia coli and Klebsiella Spp. to Ampicillin-Sulbactam and Amoxicillin-Clavulanic Acid Birgul Kacmaz* and Nedim Sultan1 Department of Central Microbiology and 1Department of Microbiology, Faculty of Medicine, Gazi University, Ankara, Turkey (Received January 30, 2007. Accepted April 13, 2007) SUMMARY: Ampicillin-sulbactam (A/S) and amoxicillin-clavulanic acid (AUG) are thought to be equally efficacious clinically against the Enterobacteriaceae family. In this study, the in vitro activities of the A/S and AUG were evaluated and compared against Escherichia coli and Klebsiella spp. Antimicrobial susceptibility tests were performed by standard agar dilution and disc diffusion techniques according to the Clinical and Laboratory Standards Institute (CLSI). During the study period, 973 strains were isolated. Of the 973 bacteria isolated, 823 were E. coli and 150 Klebsiella spp. More organisms were found to be susceptible to AUG than A/S, regardless of the susceptibility testing methodology. The agar dilution results of the isolates that were found to be sensitive or resistant were also compatible with the disc diffusion results. However, some differences were seen in the agar dilution results of some isolates that were found to be intermediately resistant with disc diffusion. In E. coli isolates, 17 of the 76 AUG intermediately resistant isolates (by disc diffusion), and 17 of the 63 A/S intermediately resistant isolates (by disc diffusion) showed different resistant patterns by agar dilution. When the CLSI breakpoint criteria are applied it should be considered that AUG and A/S sensitivity in E. coli and Klebsiella spp. -

Antibiotic Use Guidelines for Companion Animal Practice (2Nd Edition) Iii
ii Antibiotic Use Guidelines for Companion Animal Practice (2nd edition) iii Antibiotic Use Guidelines for Companion Animal Practice, 2nd edition Publisher: Companion Animal Group, Danish Veterinary Association, Peter Bangs Vej 30, 2000 Frederiksberg Authors of the guidelines: Lisbeth Rem Jessen (University of Copenhagen) Peter Damborg (University of Copenhagen) Anette Spohr (Evidensia Faxe Animal Hospital) Sandra Goericke-Pesch (University of Veterinary Medicine, Hannover) Rebecca Langhorn (University of Copenhagen) Geoffrey Houser (University of Copenhagen) Jakob Willesen (University of Copenhagen) Mette Schjærff (University of Copenhagen) Thomas Eriksen (University of Copenhagen) Tina Møller Sørensen (University of Copenhagen) Vibeke Frøkjær Jensen (DTU-VET) Flemming Obling (Greve) Luca Guardabassi (University of Copenhagen) Reproduction of extracts from these guidelines is only permitted in accordance with the agreement between the Ministry of Education and Copy-Dan. Danish copyright law restricts all other use without written permission of the publisher. Exception is granted for short excerpts for review purposes. iv Foreword The first edition of the Antibiotic Use Guidelines for Companion Animal Practice was published in autumn of 2012. The aim of the guidelines was to prevent increased antibiotic resistance. A questionnaire circulated to Danish veterinarians in 2015 (Jessen et al., DVT 10, 2016) indicated that the guidelines were well received, and particularly that active users had followed the recommendations. Despite a positive reception and the results of this survey, the actual quantity of antibiotics used is probably a better indicator of the effect of the first guidelines. Chapter two of these updated guidelines therefore details the pattern of developments in antibiotic use, as reported in DANMAP 2016 (www.danmap.org). -

Swedres-Svarm 2010
SVARM|2010 Swedish Veterinary Antimicrobial Resistance Monitoring Content Swedish Veterinary Antimicrobial Resistance Monitoring 2010 Preface .............................................................................................3 Guidance for readers ........................................................................4 Editors Summary ..........................................................................................5 Björn Bengtsson, Helle Ericsson Unnerstad, Sammanfattning...............................................................................7 Christina Greko, Ulrika Grönlund Andersson and Annica Landén Use of antimicrobials .......................................................................9 Department of Animal Health and Zoonotic bacteria ...........................................................................14 Antimicrobial Strategies, National Veterinary Salmonella ...................................................................................14 Institute (SVA) SE-751 89 Uppsala, Sweden Campylobacter .............................................................................18 Methicillin resistant Staphylococcus aureus (MRSA) ....................19 Authors Highlight: Escherichia coli with ESBL - or transferrable Björn Bengtsson, Helle Ericsson Unnerstad, AmpC-type resistance in broilers .............................................22 Christina Greko, Ulrika Grönlund Andersson and Annica Landén Indicator bacteria ...........................................................................24