DO WE KNOW WHAT the TEMPERATURE IS? Jiří J
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Physics H190 Spring 2005 Notes 1 Timeline for Nineteenth Century Physics
Physics H190 Spring 2005 Notes 1 Timeline for Nineteenth Century Physics In these notes we give a timeline of major events in 19th century physics, to orient ourselves to the way Planck and other physicists thought about physical problems in the late nineteenth century, and also to understand the open problems that existed at that time. We will start with thermal physics in the nineteenth century, which is most relevant to the story of black body radiation, although toward the end of the century thermal physics becomes mixed with electromagnetic theory. Indeed, the problem of black body radiation was fundamentally one of understanding the thermodynamics of the electromagnetic field. At the beginning of the nineteenth century, the prevailing theory of thermal physics was the caloric theory, in which heat was conceived of as a fluid, called caloric, which presumably permeated all bodies. A given body had more or less caloric, depending on its temperature. The theory gave a satisfactory accounting of a certain range of experimental data, including quantitative issues. For example, one could define a certain quantity of heat (the calorie) as the amount of heat (caloric) needed to heat one gram of water by one degree Centrigrade. If the water later cooled off by contact with colder bodies, one could say that the caloric had flowed out. It is remarkable that Carnot in 1824 gave a formulation of what is now considered the second law of thermodynamics, considering that it was based on the (defective) caloric the- ory. Of course, Carnot did not call it the second law, since the first law of thermodynamics was not known yet. -

International System of Units (SI)
International System of Units (SI) National Institute of Standards and Technology (www.nist.gov) Multiplication Prefix Symbol factor 1018 exa E 1015 peta P 1012 tera T 109 giga G 106 mega M 103 kilo k 102 * hecto h 101 * deka da 10-1 * deci d 10-2 * centi c 10-3 milli m 10-6 micro µ 10-9 nano n 10-12 pico p 10-15 femto f 10-18 atto a * Prefixes of 100, 10, 0.1, and 0.01 are not formal SI units From: Downs, R.J. 1988. HortScience 23:881-812. International System of Units (SI) Seven basic SI units Physical quantity Unit Symbol Length meter m Mass kilogram kg Time second s Electrical current ampere A Thermodynamic temperature kelvin K Amount of substance mole mol Luminous intensity candela cd Supplementary SI units Physical quantity Unit Symbol Plane angle radian rad Solid angle steradian sr From: Downs, R.J. 1988. HortScience 23:881-812. International System of Units (SI) Derived SI units with special names Physical quantity Unit Symbol Derivation Absorbed dose gray Gy J kg-1 Capacitance farad F A s V-1 Conductance siemens S A V-1 Disintegration rate becquerel Bq l s-1 Electrical charge coulomb C A s Electrical potential volt V W A-1 Energy joule J N m Force newton N kg m s-2 Illumination lux lx lm m-2 Inductance henry H V s A-1 Luminous flux lumen lm cd sr Magnetic flux weber Wb V s Magnetic flux density tesla T Wb m-2 Pressure pascal Pa N m-2 Power watt W J s-1 Resistance ohm Ω V A-1 Volume liter L dm3 From: Downs, R.J. -

Chapter IX Atoms, Caloric, and the Kinetic Theory of Heat
Chapter IX Atoms, Caloric, and the Kinetic Theory of Heat It troubled me as once I was, For I was once a child, Concluding how an Atom fell, And yet the Heavens held. The Heavens weighed the most by far, Yet Blue and solid stood, Without a bolt that I could prove. Would Giants understand? - Emily Dickinson The idea that there is an indivisible unit of matter, an atom, dates back to the Greeks. It was advocated by Leucippus and Democritus, and ridiculed by Aristotle. The modern theory got going around 1785-1803 with the experiments and interpretations of Lavoisier, Dalton, and Proust (Joseph the chemist, not Marcel the writer). The most important result was that chemical reactions always involved different substances in definite proportions – which Dalton interpreted as arising from the pairing of elements to form molecules, in definite proportions (1:1, 1:2, etc.) This didn't persuade many physicists, however; it was one fairly complex hypothesis to explain one type of experimental result. In addition, the postulated atoms had a size and mass smaller than anything that can be directly observed. Not observable... this sounded suspicious. Meanwhile, there was a fierce debate raging in physics about the nature of heat. Lying in a hot bath, or at the side of a hot fire, or under the hot Sun, one gets a strong impression that heat is a fluid. The fluid received an official name – caloric – and many books were written exploring the properties of caloric. For one thing, it's self-repellent – that's why heat always spreads to its - 2 - adjacent surroundings. -
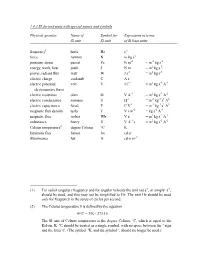
1.4.3 SI Derived Units with Special Names and Symbols
1.4.3 SI derived units with special names and symbols Physical quantity Name of Symbol for Expression in terms SI unit SI unit of SI base units frequency1 hertz Hz s-1 force newton N m kg s-2 pressure, stress pascal Pa N m-2 = m-1 kg s-2 energy, work, heat joule J N m = m2 kg s-2 power, radiant flux watt W J s-1 = m2 kg s-3 electric charge coulomb C A s electric potential, volt V J C-1 = m2 kg s-3 A-1 electromotive force electric resistance ohm Ω V A-1 = m2 kg s-3 A-2 electric conductance siemens S Ω-1 = m-2 kg-1 s3 A2 electric capacitance farad F C V-1 = m-2 kg-1 s4 A2 magnetic flux density tesla T V s m-2 = kg s-2 A-1 magnetic flux weber Wb V s = m2 kg s-2 A-1 inductance henry H V A-1 s = m2 kg s-2 A-2 Celsius temperature2 degree Celsius °C K luminous flux lumen lm cd sr illuminance lux lx cd sr m-2 (1) For radial (angular) frequency and for angular velocity the unit rad s-1, or simply s-1, should be used, and this may not be simplified to Hz. The unit Hz should be used only for frequency in the sense of cycles per second. (2) The Celsius temperature θ is defined by the equation θ/°C = T/K - 273.15 The SI unit of Celsius temperature is the degree Celsius, °C, which is equal to the Kelvin, K. -

Physics, Chapter 15: Heat and Work
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Robert Katz Publications Research Papers in Physics and Astronomy 1-1958 Physics, Chapter 15: Heat and Work Henry Semat City College of New York Robert Katz University of Nebraska-Lincoln, [email protected] Follow this and additional works at: https://digitalcommons.unl.edu/physicskatz Part of the Physics Commons Semat, Henry and Katz, Robert, "Physics, Chapter 15: Heat and Work" (1958). Robert Katz Publications. 167. https://digitalcommons.unl.edu/physicskatz/167 This Article is brought to you for free and open access by the Research Papers in Physics and Astronomy at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Robert Katz Publications by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln. 15 Heat and Work 15-1 The Nature of Heat Until about 1750 the concepts of heat and temperature were not clearly distinguished. The two concepts were thought to be equivalent in the sense that bodies at equal temperatures were thought to "contain" equal amounts of heat. Joseph Black (1728-1799) was the first to make a clear distinction between heat and temperature. Black believed that heat was a form of matter, which subsequently came to be called caloric, and that the change in temperature of a body when caloric was added to it was associated with a property of the body which he called the capacity. Later investigators endowed caloric with additional properties. The caloric fluid was thought to embody a kind of universal repulsive force. When added to a body, the repulsive force of the caloric fluid caused the body to expand. -

EL Light Output Definition.Pdf
GWENT GROUP ADVANCED MATERIAL SYSTEMS Luminance The candela per square metre (cd/m²) is the SI unit of luminance; nit is a non-SI name also used for this unit. It is often used to quote the brightness of computer displays, which typically have luminance’s of 50 to 300 nits (the sRGB spec for monitor’s targets 80 nits). Modern flat-panel (LCD and plasma) displays often exceed 300 cd/m² or 300 nits. The term is believed to come from the Latin "nitere" = to shine. Candela per square metre 1 Kilocandela per square metre 10-3 Candela per square centimetre 10-4 Candela per square foot 0,09 Foot-lambert 0,29 Lambert 3,14×10-4 Nit 1 Stilb 10-4 Luminance is a photometric measure of the density of luminous intensity in a given direction. It describes the amount of light that passes through or is emitted from a particular area, and falls within a given solid angle. The SI unit for luminance is candela per square metre (cd/m2). The CGS unit of luminance is the stilb, which is equal to one candela per square centimetre or 10 kcd/m2 Illuminance The lux (symbol: lx) is the SI unit of illuminance and luminous emittance. It is used in photometry as a measure of the intensity of light, with wavelengths weighted according to the luminosity function, a standardized model of human brightness perception. In English, "lux" is used in both singular and plural. Microlux 1000000 Millilux 1000 Lux 1 Kilolux 10-3 Lumen per square metre 1 Lumen per square centimetre 10-4 Foot-candle 0,09 Phot 10-4 Nox 1000 In photometry, illuminance is the total luminous flux incident on a surface, per unit area. -

Dimensional Analysis and Modeling
cen72367_ch07.qxd 10/29/04 2:27 PM Page 269 CHAPTER DIMENSIONAL ANALYSIS 7 AND MODELING n this chapter, we first review the concepts of dimensions and units. We then review the fundamental principle of dimensional homogeneity, and OBJECTIVES Ishow how it is applied to equations in order to nondimensionalize them When you finish reading this chapter, you and to identify dimensionless groups. We discuss the concept of similarity should be able to between a model and a prototype. We also describe a powerful tool for engi- ■ Develop a better understanding neers and scientists called dimensional analysis, in which the combination of dimensions, units, and of dimensional variables, nondimensional variables, and dimensional con- dimensional homogeneity of equations stants into nondimensional parameters reduces the number of necessary ■ Understand the numerous independent parameters in a problem. We present a step-by-step method for benefits of dimensional analysis obtaining these nondimensional parameters, called the method of repeating ■ Know how to use the method of variables, which is based solely on the dimensions of the variables and con- repeating variables to identify stants. Finally, we apply this technique to several practical problems to illus- nondimensional parameters trate both its utility and its limitations. ■ Understand the concept of dynamic similarity and how to apply it to experimental modeling 269 cen72367_ch07.qxd 10/29/04 2:27 PM Page 270 270 FLUID MECHANICS Length 7–1 ■ DIMENSIONS AND UNITS 3.2 cm A dimension is a measure of a physical quantity (without numerical val- ues), while a unit is a way to assign a number to that dimension. -
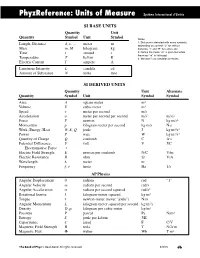
Units & Quantities Reference
PhyzReference: Units of Measure Systéme International d‘Unitès SI BASE UNITS Quantity Unit Quantity Symbol Unit Symbol Notes 1. Distance is denoted with many symbols, Length, Distance d, x, ... meter m depending on context: “y” for vertical Mass m, M kilogram kg distance, “r” and “R” for radius, etc. Time t second s 2. Notice the meter “m” is plain text while the mass “m” is italicized. Temperature T kelvin K 3. We won’t use candelas or moles. Electric Current I ampere A Luminous Intensity L candela cd Amount of Substance N mole mol SI DERIVED UNITS Quantity Unit Alternate Quantity Symbol Unit Symbol Symbol Area A square meter m2 Volume V cubic meter m3 Speed v meter per second m/s Acceleration a meter per second per second m/s2 m/s/s Force F newton N kg·m/s2 Momentum p kilogram-meter per second kg·m/s N·s Work, Energy, Heat W, E, Q joule J kg·m2/s2 Power P watt W kg·m2/s3 Quantity of Charge Q coulomb C A·s Potential Difference, V volt V J/C Electromotive Force ε Electric Field Strength E newton per coulomb N/C V/m Electric Resistance R ohm ΩV/A Wavelength λ meter m Frequency f, ν hertz Hz 1/s AP Physics Angular Displacement θ radians rad “1” Angular Velocity ω radians per second rad/s Angular Acceleration α radians per second squared rad/s2 Rotational Inertia I kilogram-meter squared kg·m2 Torque τ newton-meter (never “joule”) N·m Angular Momentum L kilogram-meter squared per second kg·m2/s Density D, ρ kilogram per cubic meter kg/m3 Pressure P pascal Pa N/m3 Entropy S joule per kelvin J/K Capacitance C farad F C/V Magnetic Field Strength B tesla T N/A·m Magnetic Flux Φ weber Wb T·m2 The Book of Phyz © Dean Baird. -

CHAPTER 1: Units, Physical Quantities, Dimensions
Lecture 1: Math Preliminaries and Introduction to Vectors 1 CHAPTER 1: Units, Physical Quantities, Dimensions 1. PHYSICS is a science of measurement. The things which are measured are called physical quantities which are defined by the describing how they are to be measured. There are three fundamental quantities in mechanics: Length Mass Time All other physical quantities combinations of these three basic quantities. 2. All physical quantities MUST have units attached to them. The standard system of units is called the SI (Systeme Internationale), or equivalently, the METRIC system. This system uses Length in Meters (m) Mass in Kilograms (kg) Time in Seconds (s) With these abbreviations for the fundamental quantities, one can also be said to be using the MKS system. 3. An example of a derived physical quantity is Density which is the mass per unit volume: Density ≡ Mass = Mass Volume (Length)·(Length)·(Length) 4. Physics uses a lot of formulas and equation. A very powerful tool in working out physics problems with these formulas and equations is Dimensional Analysis. The left side of a formula or equation must have the same dimensions as the right side in terms of the fundamental quantities of mass, length and time. 5. A very important skill to acquire is the art of guesstimation, approximating the answer to a problem. Related to that is an appreciation of sizes. Is the answer to a problem orders of magnitude too big or too small. Lecture 1: Math Preliminaries and Introduction to Vectors 2 The Standards of Length, Mass, and Time The three fundamental physical quantities are length, mass, and time. -

When and Why Are the Values of Physical Quantities Expressed with Uncertainties? Aude Caussarieu, Andrée Tiberghien
When and Why Are the Values of Physical Quantities Expressed with Uncertainties? Aude Caussarieu, Andrée Tiberghien To cite this version: Aude Caussarieu, Andrée Tiberghien. When and Why Are the Values of Physical Quantities Expressed with Uncertainties?: A Case Study of a Physics Undergraduate Laboratory Course. International Journal of Science and Mathematics Education, Springer Verlag, 2016, 15 (6), pp.997-1015. halshs- 01355889 HAL Id: halshs-01355889 https://halshs.archives-ouvertes.fr/halshs-01355889 Submitted on 31 Oct 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. 1 When and Why Are the Values of Physical Quantities Expressed with Uncertainties? A Case Study of a Physics Undergraduate Laboratory Course Aude Caussarieu • Andrée Tiberghien International journal of science and mathematics education, vol Published online 10 march 2016 Abstract The understanding of measurement is related to the understanding of the nature of science—one of the main goals of current international science teaching at all levels of education. This case study explores how a first-year university physics course deals with measurement uncertainties in the light of an epistemological analysis of measurement. The data consist of the course documents, interviews with senior instructors, and laboratory instructors’ responses to an online questionnaire. -
Physics Formulas List
Physics Formulas List Home Physics 95 Physics Formulas 23 Listen Physics Formulas List Learning physics is all about applying concepts to solve problems. This article provides a comprehensive physics formulas list, that will act as a ready reference, when you are solving physics problems. You can even use this list, for a quick revision before an exam. Physics is the most fundamental of all sciences. It is also one of the toughest sciences to master. Learning physics is basically studying the fundamental laws that govern our universe. I would say that there is a lot more to ascertain than just remember and mug up the physics formulas. Try to understand what a formula says and means, and what physical relation it expounds. If you understand the physical concepts underlying those formulas, deriving them or remembering them is easy. This Buzzle article lists some physics formulas that you would need in solving basic physics problems. Physics Formulas Mechanics Friction Moment of Inertia Newtonian Gravity Projectile Motion Simple Pendulum Electricity Thermodynamics Electromagnetism Optics Quantum Physics Derive all these formulas once, before you start using them. Study physics and look at it as an opportunity to appreciate the underlying beauty of nature, expressed through natural laws. Physics help is provided here in the form of ready to use formulas. Physics has a reputation for being difficult and to some extent that's http://www.buzzle.com/articles/physics-formulas-list.html[1/10/2014 10:58:12 AM] Physics Formulas List true, due to the mathematics involved. If you don't wish to think on your own and apply basic physics principles, solving physics problems is always going to be tough. -

Measurements of Electrical Quantities - Ján Šaliga
PHYSICAL METHODS, INSTRUMENTS AND MEASUREMENTS - Measurements Of Electrical Quantities - Ján Šaliga MEASUREMENTS OF ELECTRICAL QUANTITIES Ján Šaliga Department of Electronics and Multimedia Telecommunication, Technical University of Košice, Košice, Slovak Republic Keywords: basic electrical quantities, electric voltage, electric current, electric charge, resistance, capacitance, inductance, impedance, electrical power, digital multimeter, digital oscilloscope, spectrum analyzer, vector signal analyzer, impedance bridge. Contents 1. Introduction 2. Basic electrical quantities 2.1. Electric current and charge 2.2. Electric voltage 2.3. Resistance, capacitance, inductance and impedance 2.4 Electrical power and electrical energy 3. Voltage, current and power representation in time and frequency 3. 1. Time domain 3. 2. Frequency domain 4. Parameters of electrical quantities. 5. Measurement methods and instrumentations 5.1. Voltage, current and resistance measuring instruments 5.1.1. Meters 5.1.2. Oscilloscopes 5.1.3. Spectrum and signal analyzers 5.2. Electrical power and energy measuring instruments 5.2.1. Transition power meters 5.2.2. Absorption power meters 5.3. Capacitance, inductance and resistance measuring instruments 6. Conclusions Glossary Bibliography BiographicalUNESCO Sketch – EOLSS Summary In this work, a SAMPLEfundamental overview of measurement CHAPTERS of electrical quantities is given, including units of their measurement. Electrical quantities are of various properties and have different characteristics. They also differ in the frequency range and spectral content from dc up to tens of GHz and the level range from nano and micro units up to Mega and Giga units. No single instrument meets all these requirements even for only one quantity, and therefore the measurement of electrical quantities requires a wide variety of techniques and instrumentations to perform a required measurement.