Long, Noncoding RNA Dysregulation in Glioblastoma
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supplementary Data
Figure 2S 4 7 A - C 080125 CSCs 080418 CSCs - + IFN-a 48 h + IFN-a 48 h + IFN-a 72 h 6 + IFN-a 72 h 3 5 MRFI 4 2 3 2 1 1 0 0 MHC I MHC II MICA MICB ULBP-1 ULBP-2 ULBP-3 ULBP-4 MHC I MHC II MICA MICB ULBP-1 ULBP-2 ULBP-3 ULBP-4 7 B 13 080125 FBS - D 080418 FBS - + IFN-a 48 h 12 + IFN-a 48 h + IFN-a 72 h + IFN-a 72 h 6 080125 FBS 11 10 5 9 8 4 7 6 3 MRFI 5 4 2 3 2 1 1 0 0 MHC I MHC II MICA MICB ULBP-1 ULBP-2 ULBP-3 ULBP-4 MHC I MHC II MICA MICB ULBP-1 ULBP-2 ULBP-3 ULBP-4 Molecule Molecule FIGURE 4S FIGURE 5S Panel A Panel B FIGURE 6S A B C D Supplemental Results Table 1S. Modulation by IFN-α of APM in GBM CSC and FBS tumor cell lines. Molecule * Cell line IFN-α‡ HLA β2-m# HLA LMP TAP1 TAP2 class II A A HC§ 2 7 10 080125 CSCs - 1∞ (1) 3 (65) 2 (91) 1 (2) 6 (47) 2 (61) 1 (3) 1 (2) 1 (3) + 2 (81) 11 (80) 13 (99) 1 (3) 8 (88) 4 (91) 1 (2) 1 (3) 2 (68) 080125 FBS - 2 (81) 4 (63) 4 (83) 1 (3) 6 (80) 3 (67) 2 (86) 1 (3) 2 (75) + 2 (99) 14 (90) 7 (97) 5 (75) 7 (100) 6 (98) 2 (90) 1 (4) 3 (87) 080418 CSCs - 2 (51) 1 (1) 1 (3) 2 (47) 2 (83) 2 (54) 1 (4) 1 (2) 1 (3) + 2 (81) 3 (76) 5 (75) 2 (50) 2 (83) 3 (71) 1 (3) 2 (87) 1 (2) 080418 FBS - 1 (3) 3 (70) 2 (88) 1 (4) 3 (87) 2 (76) 1 (3) 1 (3) 1 (2) + 2 (78) 7 (98) 5 (99) 2 (94) 5 (100) 3 (100) 1 (4) 2 (100) 1 (2) 070104 CSCs - 1 (2) 1 (3) 1 (3) 2 (78) 1 (3) 1 (2) 1 (3) 1 (3) 1 (2) + 2 (98) 8 (100) 10 (88) 4 (89) 3 (98) 3 (94) 1 (4) 2 (86) 2 (79) * expression of APM molecules was evaluated by intracellular staining and cytofluorimetric analysis; ‡ cells were treatead or not (+/-) for 72 h with 1000 IU/ml of IFN-α; # β-2 microglobulin; § β-2 microglobulin-free HLA-A heavy chain; ∞ values are indicated as ratio between the mean of fluorescence intensity of cells stained with the selected mAb and that of the negative control; bold values indicate significant MRFI (≥ 2). -

Diseasespecific and Inflammationindependent Stromal
Full Length Arthritis & Rheumatism DOI 10.1002/art.37704 Disease-specific and inflammation-independent stromal alterations in spondyloarthritis synovitis Nataliya Yeremenko1,2, Troy Noordenbos1,2, Tineke Cantaert1,3, Melissa van Tok1,2, Marleen van de Sande1, Juan D. Cañete4, Paul P. Tak1,5*, Dominique Baeten1,2 1Department of Clinical Immunology and Rheumatology and 2Department of Experimental Immunology, Academic Medical Center/University of Amsterdam, the Netherlands. 3Department of Immunobiology, Yale University School of Medicine, New Haven, CT, USA. 4Department of Rheumatology, Hospital Clinic de Barcelona and IDIBAPS, Spain. 5Arthrogen B.V., Amsterdam, the Netherlands. *Currently also: GlaxoSmithKline, Stevenage, U.K. Corresponding author: Dominique Baeten, MD, PhD, Department of Clinical Immunology and Rheumatology, F4-105, Academic Medical Center/University of Amsterdam, Meibergdreef 9, 1105 AZ Amsterdam, The Netherlands. E-mail: [email protected] This article has been accepted for publication and undergone full peer review but has not been through the copyediting, typesetting, pagination and proofreading process which may lead to differences between this version and the Version of Record. Please cite this article as an ‘Accepted Article’, doi: 10.1002/art.37704 © 2012 American College of Rheumatology Received: Apr 11, 2012; Revised: Jul 25, 2012; Accepted: Sep 06, 2012 Arthritis & Rheumatism Page 2 of 36 Abstract Objective: The molecular processes driving the distinct patterns of synovial inflammation and tissue remodelling in spondyloarthritis (SpA) versus rheumatoid arthritis (RA) remain largely unknown. Therefore, we aimed to identify novel and unsuspected disease- specific pathways in SpA by a systematic and unbiased synovial gene expression analysis. Methods: Differentially expressed genes were identified by pan-genomic microarray and confirmed by quantitative PCR and immunohistochemistry using synovial tissue biopsies of SpA (n=63), RA (n=28) and gout (n=9) patients. -
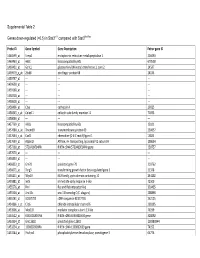
0.5) in Stat3∆/∆ Compared with Stat3flox/Flox
Supplemental Table 2 Genes down-regulated (<0.5) in Stat3∆/∆ compared with Stat3flox/flox Probe ID Gene Symbol Gene Description Entrez gene ID 1460599_at Ermp1 endoplasmic reticulum metallopeptidase 1 226090 1460463_at H60c histocompatibility 60c 670558 1460431_at Gcnt1 glucosaminyl (N-acetyl) transferase 1, core 2 14537 1459979_x_at Zfp68 zinc finger protein 68 24135 1459747_at --- --- --- 1459608_at --- --- --- 1459168_at --- --- --- 1458718_at --- --- --- 1458618_at --- --- --- 1458466_at Ctsa cathepsin A 19025 1458345_s_at Colec11 collectin sub-family member 11 71693 1458046_at --- --- --- 1457769_at H60a histocompatibility 60a 15101 1457680_a_at Tmem69 transmembrane protein 69 230657 1457644_s_at Cxcl1 chemokine (C-X-C motif) ligand 1 14825 1457639_at Atp6v1h ATPase, H+ transporting, lysosomal V1 subunit H 108664 1457260_at 5730409E04Rik RIKEN cDNA 5730409E04Rik gene 230757 1457070_at --- --- --- 1456893_at --- --- --- 1456823_at Gm70 predicted gene 70 210762 1456671_at Tbrg3 transforming growth factor beta regulated gene 3 21378 1456211_at Nlrp10 NLR family, pyrin domain containing 10 244202 1455881_at Ier5l immediate early response 5-like 72500 1455576_at Rinl Ras and Rab interactor-like 320435 1455304_at Unc13c unc-13 homolog C (C. elegans) 208898 1455241_at BC037703 cDNA sequence BC037703 242125 1454866_s_at Clic6 chloride intracellular channel 6 209195 1453906_at Med13l mediator complex subunit 13-like 76199 1453522_at 6530401N04Rik RIKEN cDNA 6530401N04 gene 328092 1453354_at Gm11602 predicted gene 11602 100380944 1453234_at -

Product Description SALSA MLPA Probemix P476-A1 ZNRF3
MRC-Holland ® Product Description version A1-02; Issued 25 September 2018 MLPA Product Description SALSA ® MLPA ® Probemix P476-A1 ZNRF3 To be used with the MLPA General Protocol. Version A1. New Product. Catalogue numbers: • P476-025R: SALSA MLPA Probemix P476 ZNRF3, 25 reactions. • P476-050R: SALSA MLPA Probemix P476 ZNRF3, 50 reactions. • P476-100R: SALSA MLPA Probemix P476 ZNRF3, 100 reactions. To be used in combination with a SALSA MLPA reagent kit, available for various number of reactions. MLPA reagent kits are either provided with FAM or Cy5.0 dye-labelled PCR primer, suitable for Applied Biosystems and Beckman capillary sequencers, respectively (see www.mlpa.com ). Certificate of Analysis: Information regarding storage conditions, quality tests, and a sample electropherogram from the current sales lot is available at www.mlpa.com . Precautions and warnings: For professional use only. Always consult the most recent product description AND the MLPA General Protocol before use: www.mlpa.com . It is the responsibility of the user to be aware of the latest scientific knowledge of the application before drawing any conclusions from findings generated with this product. General information: The SALSA MLPA Probemix P476 ZNRF3 is a research use only (RUO) assay for the detection of deletions and duplications in the ZNRF3 gene. ZNRF3 is an E3 ubiquitin-protein ligase that acts as a negative feedback regulator of Wnt signalling (Hao et al. 2012). Three independent studies show homozygous deletions of the ZNRF3 gene in 10 to 16% of adrenocortical carcinoma cases (Assié et al. 2014; Juhlin et al. 2015; Zheng et al. 2016). Moreover, in 51% of microsatellite stable colorectal cancers deletion events at the ZNRF3 locus are detected (Bond et al. -

Identification of Active Signaling Pathways by Integrating Gene Expression and Protein Interaction Data Md Humayun Kabir1,2,3, Ralph Patrick2,4,5, Joshua W
Kabir et al. BMC Systems Biology 2018, 12(Suppl 9):120 https://doi.org/10.1186/s12918-018-0655-x RESEARCH Open Access Identification of active signaling pathways by integrating gene expression and protein interaction data Md Humayun Kabir1,2,3, Ralph Patrick2,4,5, Joshua W. K. Ho2,4,6* and Michael D. O’Connor1,7* From 29th International Conference on Genome Informatics Yunnan, China. 3-5 December 2018 Abstract Background: Signaling pathways are the key biological mechanisms that transduce extracellular signals to affect transcription factor mediated gene regulation within cells. A number of computational methods have been developed to identify the topological structure of a specific signaling pathway using protein-protein interaction data, but they are not designed for identifying active signaling pathways in an unbiased manner. On the other hand, there are statistical methods based on gene sets or pathway data that can prioritize likely active signaling pathways, but they do not make full use of active pathway structure that link receptor, kinases and downstream transcription factors. Results: Here, we present a method to simultaneously predict the set of active signaling pathways, together with their pathway structure, by integrating protein-protein interaction network and gene expression data. We evaluated the capacity for our method to predict active signaling pathways for dental epithelial cells, ocular lens epithelial cells, human pluripotent stem cell-derived lens epithelial cells, and lens fiber cells. This analysis showed our approach could identify all the known active pathways that are associated with tooth formation and lens development. Conclusions: The results suggest that SPAGI can be a useful approach to identify the potential active signaling pathways given a gene expression profile. -

Oncorequisite Role of an Aldehyde Dehydrogenase in the Pathogenesis of T-Cell Acute Lymphoblastic Leukemia
Acute Lymphoblastic Leukemia SUPPLEMENTARY APPENDIX Oncorequisite role of an aldehyde dehydrogenase in the pathogenesis of T-cell acute lymphoblastic leukemia Chujing Zhang, 1 Stella Amanda, 1 Cheng Wang, 2 Tze King Tan, 1 Muhammad Zulfaqar Ali, 1 Wei Zhong Leong, 1 Ley Moy Ng, 1 Shojiro Kitajima, 3 Zhenhua Li, 4 Allen Eng Juh Yeoh, 1,4 Shi Hao Tan 1 and Takaomi Sanda 1,5 1Cancer Science Institute of Singapore, National University of Singapore, Singapore; 2Department of Anatomy, National University of Singapore, Singapore; 3Institute for Advanced Biosciences, Keio University, Tsuruoka, Japan; 4VIVA-NUS CenTRAL, Department of Pedi - atrics, National University of Singapore, Singapore and 5Department of Medicine, Yong Loo Lin School of Medicine, National University of Singapore, Singapore. ©2021 Ferrata Storti Foundation. This is an open-access paper. doi:10.3324/haematol. 2019.245639 Received: December 18, 2019. Accepted: May 14, 2020. Pre-published: May 15, 2020. Correspondence: TAKAOMI SANDA - [email protected] Zhang et al Roles of ALDH in T-ALL Supplementary Information Supplementary Materials and Methods Cell culture and reagents All human leukemia cell lines were cultured in RPMI-1640 medium (BioWest) supplemented with 10% FBS (BioWest). 293T cells were maintained in DMEM medium (BioWest) supplemented with 10% FBS and penicillin/streptomycin (Life Technologies). All-trans retinaldehyde was purchased from Sigma-Aldrich. Glucose free and Glutamine free RPMI- 1640 media were purchased from ThermoFisher. Patient derived xenograft (PDX) sample T-ALL patient-derived PDX sample (DFCI15) was kindly provided by Alejandro Gutierrez (Boston Children’s Hospital, Boston). Mouse studies were conducted according to the recommendations from the Institutional Animal Care and Use Committee (IACUC) and all protocols were approved by the Committee at the National University of Singapore (NUS). -

Aneuploidy: Using Genetic Instability to Preserve a Haploid Genome?
Health Science Campus FINAL APPROVAL OF DISSERTATION Doctor of Philosophy in Biomedical Science (Cancer Biology) Aneuploidy: Using genetic instability to preserve a haploid genome? Submitted by: Ramona Ramdath In partial fulfillment of the requirements for the degree of Doctor of Philosophy in Biomedical Science Examination Committee Signature/Date Major Advisor: David Allison, M.D., Ph.D. Academic James Trempe, Ph.D. Advisory Committee: David Giovanucci, Ph.D. Randall Ruch, Ph.D. Ronald Mellgren, Ph.D. Senior Associate Dean College of Graduate Studies Michael S. Bisesi, Ph.D. Date of Defense: April 10, 2009 Aneuploidy: Using genetic instability to preserve a haploid genome? Ramona Ramdath University of Toledo, Health Science Campus 2009 Dedication I dedicate this dissertation to my grandfather who died of lung cancer two years ago, but who always instilled in us the value and importance of education. And to my mom and sister, both of whom have been pillars of support and stimulating conversations. To my sister, Rehanna, especially- I hope this inspires you to achieve all that you want to in life, academically and otherwise. ii Acknowledgements As we go through these academic journeys, there are so many along the way that make an impact not only on our work, but on our lives as well, and I would like to say a heartfelt thank you to all of those people: My Committee members- Dr. James Trempe, Dr. David Giovanucchi, Dr. Ronald Mellgren and Dr. Randall Ruch for their guidance, suggestions, support and confidence in me. My major advisor- Dr. David Allison, for his constructive criticism and positive reinforcement. -

Analysis of the Indacaterol-Regulated Transcriptome in Human Airway
Supplemental material to this article can be found at: http://jpet.aspetjournals.org/content/suppl/2018/04/13/jpet.118.249292.DC1 1521-0103/366/1/220–236$35.00 https://doi.org/10.1124/jpet.118.249292 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS J Pharmacol Exp Ther 366:220–236, July 2018 Copyright ª 2018 by The American Society for Pharmacology and Experimental Therapeutics Analysis of the Indacaterol-Regulated Transcriptome in Human Airway Epithelial Cells Implicates Gene Expression Changes in the s Adverse and Therapeutic Effects of b2-Adrenoceptor Agonists Dong Yan, Omar Hamed, Taruna Joshi,1 Mahmoud M. Mostafa, Kyla C. Jamieson, Radhika Joshi, Robert Newton, and Mark A. Giembycz Departments of Physiology and Pharmacology (D.Y., O.H., T.J., K.C.J., R.J., M.A.G.) and Cell Biology and Anatomy (M.M.M., R.N.), Snyder Institute for Chronic Diseases, Cumming School of Medicine, University of Calgary, Calgary, Alberta, Canada Received March 22, 2018; accepted April 11, 2018 Downloaded from ABSTRACT The contribution of gene expression changes to the adverse and activity, and positive regulation of neutrophil chemotaxis. The therapeutic effects of b2-adrenoceptor agonists in asthma was general enriched GO term extracellular space was also associ- investigated using human airway epithelial cells as a therapeu- ated with indacaterol-induced genes, and many of those, in- tically relevant target. Operational model-fitting established that cluding CRISPLD2, DMBT1, GAS1, and SOCS3, have putative jpet.aspetjournals.org the long-acting b2-adrenoceptor agonists (LABA) indacaterol, anti-inflammatory, antibacterial, and/or antiviral activity. Numer- salmeterol, formoterol, and picumeterol were full agonists on ous indacaterol-regulated genes were also induced or repressed BEAS-2B cells transfected with a cAMP-response element in BEAS-2B cells and human primary bronchial epithelial cells by reporter but differed in efficacy (indacaterol $ formoterol . -

Supplementary Table 3: Genes Only Influenced By
Supplementary Table 3: Genes only influenced by X10 Illumina ID Gene ID Entrez Gene Name Fold change compared to vehicle 1810058M03RIK -1.104 2210008F06RIK 1.090 2310005E10RIK -1.175 2610016F04RIK 1.081 2610029K11RIK 1.130 381484 Gm5150 predicted gene 5150 -1.230 4833425P12RIK -1.127 4933412E12RIK -1.333 6030458P06RIK -1.131 6430550H21RIK 1.073 6530401D06RIK 1.229 9030607L17RIK -1.122 A330043C08RIK 1.113 A330043L12 1.054 A530092L01RIK -1.069 A630054D14 1.072 A630097D09RIK -1.102 AA409316 FAM83H family with sequence similarity 83, member H 1.142 AAAS AAAS achalasia, adrenocortical insufficiency, alacrimia 1.144 ACADL ACADL acyl-CoA dehydrogenase, long chain -1.135 ACOT1 ACOT1 acyl-CoA thioesterase 1 -1.191 ADAMTSL5 ADAMTSL5 ADAMTS-like 5 1.210 AFG3L2 AFG3L2 AFG3 ATPase family gene 3-like 2 (S. cerevisiae) 1.212 AI256775 RFESD Rieske (Fe-S) domain containing 1.134 Lipo1 (includes AI747699 others) lipase, member O2 -1.083 AKAP8L AKAP8L A kinase (PRKA) anchor protein 8-like -1.263 AKR7A5 -1.225 AMBP AMBP alpha-1-microglobulin/bikunin precursor 1.074 ANAPC2 ANAPC2 anaphase promoting complex subunit 2 -1.134 ANKRD1 ANKRD1 ankyrin repeat domain 1 (cardiac muscle) 1.314 APOA1 APOA1 apolipoprotein A-I -1.086 ARHGAP26 ARHGAP26 Rho GTPase activating protein 26 -1.083 ARL5A ARL5A ADP-ribosylation factor-like 5A -1.212 ARMC3 ARMC3 armadillo repeat containing 3 -1.077 ARPC5 ARPC5 actin related protein 2/3 complex, subunit 5, 16kDa -1.190 activating transcription factor 4 (tax-responsive enhancer element ATF4 ATF4 B67) 1.481 AU014645 NCBP1 nuclear cap -

Kremen Is Beyond a Subsidiary Co-Receptor of Wnt Signaling: an in Silico Validation
Turkish Journal of Biology Turk J Biol (2015) 39: 501-510 http://journals.tubitak.gov.tr/biology/ © TÜBİTAK Research Article doi:10.3906/biy-1409-1 Kremen is beyond a subsidiary co-receptor of Wnt signaling: an in silico validation 1 2, 3 Hemn MOHAMMADPOUR , Saeed KHALILI *, Zahra Sadat HASHEMI 1 Department of Medical Immunology, Faculty of Medical Science, Tarbiat Modares University, Tehran, Iran 2 Department of Medical Biotechnology, Faculty of Medical Science, Tarbiat Modares University, Tehran, Iran 3 Department of Medical Biotechnology, School of Advanced Medical Technologies, Tehran University of Medical Science, Tehran, Iran Received: 01.09.2014 Accepted/Published Online: 12.02.2015 Printed: 15.06.2015 Abstract: Kremen-1 is a co-receptor of Wnt signaling, interacting with the Dkk-3 protein, which is an antagonist of the Wnt/β catenin pathway. In the present study we attempted to shed some light on the possible orientations of Kremen/Dkk-3 interactions, explaining the mechanisms of Kremen and Dkk-3 functions. Employing state-of-the-art software, a Kremen model was built and subsequently refined. The quality of the final model was evaluated using RMSD calculations. Ultimately, we used the Kremen model for docking analysis between Kremen and Dkk-3 molecules. A model built by the Robetta server showed the best quality scores. Near native coordination of the final model was verified getting < 2 Å RMSD values between our model and an experimentally resolved structure. Docking analysis indicates that one low energy orientation for Kremen/Dkk-3 involves all extracellular domains of Kremen, while another only involves the CUB and WSC domains. -

(Q36.1;Q24) with a Concurrent Submicroscopic Del(4)(Q23q24) in an Adult with Polycythemia Vera
cancers Case Report A Novel Acquired t(2;4)(q36.1;q24) with a Concurrent Submicroscopic del(4)(q23q24) in An Adult with Polycythemia Vera Eigil Kjeldsen Cancer Cytogenetic Section, HemoDiagnostic Laboratory, Department of Hematology, Aarhus University Hospital, Tage-Hansens Gade 2, DK-8000 Aarhus C, Denmark; [email protected]; Tel.: +45-7846-7398; Fax: +45-7846-7399 Received: 6 June 2018; Accepted: 21 June 2018; Published: 25 June 2018 Abstract: Background: Polycythemia vera (PV) is a clonal myeloid stem cell disease characterized by a growth-factor independent erythroid proliferation with an inherent tendency to transform into overt acute myeloid malignancy. Approximately 95% of the PV patients harbor the JAK2V617F mutation while less than 35% of the patients harbor cytogenetic abnormalities at the time of diagnosis. Methods and Results: Here we present a JAK2V617F positive PV patient where G-banding revealed an apparently balanced t(2;4)(q35;q21), which was confirmed by 24-color karyotyping. Oligonucleotide array-based Comparative Genomic Hybridization (aCGH) analysis revealed an interstitial 5.4 Mb large deletion at 4q23q24. Locus-specific fluorescent in situ hybridization (FISH) analyses confirmed the mono-allelic 4q deletion and that it was located on der(4)t(2;4). Additional locus-specific bacterial artificial chromosome (BAC) probes and mBanding refined the breakpoint on chromosome 2. With these methods the karyotype was revised to 46,XX,t(2;4)(q36.1;q24)[18]/46,XX[7]. Conclusions: This is the first report on a PV patient associated with an acquired novel t(2;4)(q36.1;q24) and a concurrent submicroscopic deletion del(4)(q23q24). -

Meta-Analysis Identifies 13 New Loci Associated with Waist-Hip Ratio And
ARTICLES Meta-analysis identifies 13 new loci associated with waist-hip ratio and reveals sexual dimorphism in the genetic basis of fat distribution Waist-hip ratio (WHR) is a measure of body fat distribution and a predictor of metabolic consequences independent of overall adiposity. WHR is heritable, but few genetic variants influencing this trait have been identified. We conducted a meta-analysis of 32 genome-wide association studies for WHR adjusted for body mass index (comprising up to 77,167 participants), following up 16 loci in an additional 29 studies (comprising up to 113,636 subjects). We identified 13 new loci in or near RSPO3, VEGFA, TBX15-WARS2, NFE2L3, GRB14, DNM3-PIGC, ITPR2-SSPN, LY86, HOXC13, ADAMTS9, ZNRF3-KREMEN1, NISCH-STAB1 and CPEB4 (P = 1.9 × 10−9 to P = 1.8 × 10−40) and the known signal at LYPLAL1. Seven of these loci exhibited marked sexual dimorphism, all with a stronger effect on WHR in women than men (P for sex difference = 1.9 × 10−3 to P = 1.2 × 10−13). These findings provide evidence for multiple loci that modulate body fat distribution independent of overall adiposity and reveal strong gene-by-sex interactions. Central obesity and body fat distribution, as measured by waist discovery stage, up to 2,850,269 imputed and genotyped SNPs circumference and WHR, are associated with individual risk of type were examined in 32 GWAS comprising up to 77,167 participants 2 diabetes (T2D)1,2 and coronary heart disease3 and with mortality informative for anthropometric measures of body fat distribution. from all causes4.