SAFETY DATA SHEET Revision Date 01/14/2020 Print Date 09/25/2021
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Polymorphism, Halogen Bonding, and Chalcogen Bonding in the Diiodine Adducts of 1,3- and 1,4-Dithiane
molecules Article Polymorphism, Halogen Bonding, and Chalcogen Bonding in the Diiodine Adducts of 1,3- and 1,4-Dithiane Andrew J. Peloquin 1, Srikar Alapati 2, Colin D. McMillen 1, Timothy W. Hanks 2 and William T. Pennington 1,* 1 Department of Chemistry, Clemson University, Clemson, SC 29634, USA; [email protected] (A.J.P.); [email protected] (C.D.M.) 2 Department of Chemistry, Furman University, Greenville, SC 29613, USA; [email protected] (S.A.); [email protected] (T.W.H.) * Correspondence: [email protected] Abstract: Through variations in reaction solvent and stoichiometry, a series of S-diiodine adducts of 1,3- and 1,4-dithiane were isolated by direct reaction of the dithianes with molecular diiodine in solution. In the case of 1,3-dithiane, variations in reaction solvent yielded both the equatorial and the axial isomers of S-diiodo-1,3-dithiane, and their solution thermodynamics were further studied via DFT. Additionally, S,S’-bis(diiodo)-1,3-dithiane was also isolated. The 1:1 cocrystal, (1,4-dithiane)·(I2) was further isolated, as well as a new polymorph of S,S’-bis(diiodo)-1,4-dithiane. Each structure showed significant S···I halogen and chalcogen bonding interactions. Further, the product of the diiodine-promoted oxidative addition of acetone to 1,4-dithiane, as well as two new cocrystals of 1,4-dithiane-1,4-dioxide involving hydronium, bromide, and tribromide ions, was isolated. Keywords: crystal engineering; chalcogen bonding; halogen bonding; polymorphism; X-ray diffraction Citation: Peloquin, A.J.; Alapati, S.; McMillen, C.D.; Hanks, T.W.; Pennington, W.T. -
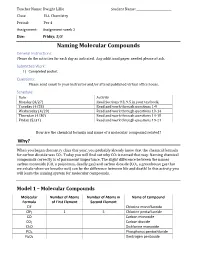
Naming Molecular Compounds General Instructions: Please Do the Activities for Each Day As Indicated
Teacher Name: Dwight Lillie Student Name: ________________________ Class: ELL Chemistry Period: Per 4 Assignment: Assignment week 2 Due: Friday, 5/8 Naming Molecular Compounds General Instructions: Please do the activities for each day as indicated. Any additional paper needed please attach. Submitted Work: 1) Completed packet. Questions: Please send email to your instructor and/or attend published virtual office hours. Schedule: Date Activity Monday (4/27) Read Sections 9.3, 9.5 in your textbook. Tuesday (4/28) Read and work through questions 1-9 Wednesday (4/29) Read and work through questions 10-14 Thursday (4/30) Read and work through questions 14-18 Friday (5/31) Read and work through questions 19-21 How are the chemical formula and name of a molecular compound related? Why? When you began chemistry class this year, you probably already knew that the chemical formula for carbon dioxide was CO2. Today you will find out why CO2 is named that way. Naming chemical compounds correctly is of paramount importance. The slight difference between the names carbon monoxide (CO, a poisonous, deadly gas) and carbon dioxide (CO2, a greenhouse gas that we exhale when we breathe out) can be the difference between life and death! In this activity you will learn the naming system for molecular compounds. Model 1 – Molecular Compounds Molecular Number of Atoms Number of Atoms in Name of Compound Formula of First Element Second Element ClF Chlorine monofluoride ClF5 1 5 Chlorine pentafluoride CO Carbon monoxide CO2 Carbon dioxide Cl2O Dichlorine monoxide PCl5 Phosphorus pentachloride N2O5 Dinitrogen pentoxide 1. Fill in the table to indicate the number of atoms of each type in the molecular formula. -

Pp-03-25-New Dots.Qxd 10/23/02 2:16 PM Page 136
pp-03-25-new dots.qxd 10/23/02 2:16 PM Page 136 136 BROMIC ACID / BROMINE BROMIC ACID [7789–31–3] Formula: HBrO3; MW 128.91 Uses Bromic acid is used as an oxidizing agent; and also as intermediate in the preparation of dyes and pharmaceuticals . Physical Properties Unstable compound; stable only in dilute aqueous solutions; solution turns yellow on standing; decomposes when heated to 100°C. Preparation Bromic acid is prepared by adding sulfuric acid to barium bromate. Ba(BrO3)2 + H2SO4 → 2HBrO3 + BaSO4 The product is distilled and absorbed in water. A 50% solution may be obtained by slow evaporation of the dilute solution in vacuum at –12°C. Toxicity Contact with skin and eyes can cause severe irritation. BROMINE [7726–95–6] Symbol Br; atomic number 35; atomic weight 79.904; a halogen group ele- ment; electron affinity 3.36359 eV; electronegativity 2.8; electron configura- tion [Ar] 3d104s24p5; most stable valence states –1 and +5, less stable valence states +1 and +3; a diatomic molecule (Br2) in liquid and vapor states over a wide range of temperature; two stable isotopes, Br–79 (50.57%) and Br–81 (49.43%). Occurrence and Uses Bromine occurs in nature as bromide in many natural brine wells and salt deposits. It also is found in seawater at a concentration of 85 mg/L. The ele- ment was discovered by A. J. Balard and C. Lowig, independently in 1826. Bromine is used in bleaching fibers and as a disinfectant for water purifica- tion. Other applications are in organic synthesis as an oxidizing or brominat- ing agent; in the manufacture of ethylene dibromide, methyl bromide and other bromo compounds for dyes and pharmaceutical uses; as a fire retardant for plastics; and in chemical analysis. -

Iodine Monobromide Original Commentary
IODINE MONOBROMIDE 1 Iodine Monobromide O OH t 1. IBr, PhMe O -BuO O –80 to –85 °C (2) BrI 2. K2CO3, MeOH 71% OBn 13.9:1 [7789-33-5] Brl (MW 206.80) OBn InChI = 1S/BrI/c1-2 InChIKey = CBEQRNSPHCCXSH-UHFFFAOYAO (diastereoselective cyclizations of homoallylic carbonates; elec- trophilic addition to alkenes; -bromination of steroidal aldehy- Table 1 Regiochemistry of IBr addition to alkenes des and ketones;, cleavage of carbon–metal bonds) Alkene % Markovnikov % anti-Markovnikov Physical Data: mp 40 ◦ C; bp 116 ◦C (dec); d 4.416 g cm−3. Solubility: sol alcohol, ether, CS2, acetic acid, acetone, CH2Cl2, MeCH=CH2 65 35 MeCN, DME, toluene (but with limited solubility below –80 ErCH=CH2 45 55 ◦ C); soluble in H2O, but hydrolyzes to HBr and IOH. i-PrCH=CH2 15 85 Form Supplied in: brownish-black crystals or very hard, black t-BuCH=CH2 0 100 solid; widely available (usually 97–98% purity). PhCH=CH2 100 0 Handling, Storage, and Precautions: corrosive solid and vapor; readily absorbed through skin and mucous membranes; use in a fume hood and wear gloves and appropriate protective clothing; air-, moisture-, and light-sensitive; should be stored refrigerated (under N ) in a tightly sealed amber bottle. Forms explosive 2 mixtures with potassium and sodium, and reacts extremely -Bromination of Steroidal Aldehydes and Ketones. Io- exothermically with phosphorus and tin. dine monobromide is an effective monobrominating reagent for steroidal aldehydes (eq 3). The ratio of diastereomeric products is in part a function of the reaction site’s proximity to an asymmet- Original Commentary ric center, with -orientation favored for the entering bromine. -

Chemical Names and CAS Numbers Final
Chemical Abstract Chemical Formula Chemical Name Service (CAS) Number C3H8O 1‐propanol C4H7BrO2 2‐bromobutyric acid 80‐58‐0 GeH3COOH 2‐germaacetic acid C4H10 2‐methylpropane 75‐28‐5 C3H8O 2‐propanol 67‐63‐0 C6H10O3 4‐acetylbutyric acid 448671 C4H7BrO2 4‐bromobutyric acid 2623‐87‐2 CH3CHO acetaldehyde CH3CONH2 acetamide C8H9NO2 acetaminophen 103‐90‐2 − C2H3O2 acetate ion − CH3COO acetate ion C2H4O2 acetic acid 64‐19‐7 CH3COOH acetic acid (CH3)2CO acetone CH3COCl acetyl chloride C2H2 acetylene 74‐86‐2 HCCH acetylene C9H8O4 acetylsalicylic acid 50‐78‐2 H2C(CH)CN acrylonitrile C3H7NO2 Ala C3H7NO2 alanine 56‐41‐7 NaAlSi3O3 albite AlSb aluminium antimonide 25152‐52‐7 AlAs aluminium arsenide 22831‐42‐1 AlBO2 aluminium borate 61279‐70‐7 AlBO aluminium boron oxide 12041‐48‐4 AlBr3 aluminium bromide 7727‐15‐3 AlBr3•6H2O aluminium bromide hexahydrate 2149397 AlCl4Cs aluminium caesium tetrachloride 17992‐03‐9 AlCl3 aluminium chloride (anhydrous) 7446‐70‐0 AlCl3•6H2O aluminium chloride hexahydrate 7784‐13‐6 AlClO aluminium chloride oxide 13596‐11‐7 AlB2 aluminium diboride 12041‐50‐8 AlF2 aluminium difluoride 13569‐23‐8 AlF2O aluminium difluoride oxide 38344‐66‐0 AlB12 aluminium dodecaboride 12041‐54‐2 Al2F6 aluminium fluoride 17949‐86‐9 AlF3 aluminium fluoride 7784‐18‐1 Al(CHO2)3 aluminium formate 7360‐53‐4 1 of 75 Chemical Abstract Chemical Formula Chemical Name Service (CAS) Number Al(OH)3 aluminium hydroxide 21645‐51‐2 Al2I6 aluminium iodide 18898‐35‐6 AlI3 aluminium iodide 7784‐23‐8 AlBr aluminium monobromide 22359‐97‐3 AlCl aluminium monochloride -

Hazardous Substances (Chemicals) Transfer Notice 2006
16551655 OF THURSDAY, 22 JUNE 2006 WELLINGTON: WEDNESDAY, 28 JUNE 2006 — ISSUE NO. 72 ENVIRONMENTAL RISK MANAGEMENT AUTHORITY HAZARDOUS SUBSTANCES (CHEMICALS) TRANSFER NOTICE 2006 PURSUANT TO THE HAZARDOUS SUBSTANCES AND NEW ORGANISMS ACT 1996 1656 NEW ZEALAND GAZETTE, No. 72 28 JUNE 2006 Hazardous Substances and New Organisms Act 1996 Hazardous Substances (Chemicals) Transfer Notice 2006 Pursuant to section 160A of the Hazardous Substances and New Organisms Act 1996 (in this notice referred to as the Act), the Environmental Risk Management Authority gives the following notice. Contents 1 Title 2 Commencement 3 Interpretation 4 Deemed assessment and approval 5 Deemed hazard classification 6 Application of controls and changes to controls 7 Other obligations and restrictions 8 Exposure limits Schedule 1 List of substances to be transferred Schedule 2 Changes to controls Schedule 3 New controls Schedule 4 Transitional controls ______________________________ 1 Title This notice is the Hazardous Substances (Chemicals) Transfer Notice 2006. 2 Commencement This notice comes into force on 1 July 2006. 3 Interpretation In this notice, unless the context otherwise requires,— (a) words and phrases have the meanings given to them in the Act and in regulations made under the Act; and (b) the following words and phrases have the following meanings: 28 JUNE 2006 NEW ZEALAND GAZETTE, No. 72 1657 manufacture has the meaning given to it in the Act, and for the avoidance of doubt includes formulation of other hazardous substances pesticide includes but -

Determination of Iodine Values Using 1,3-Dibromo-5,5-Dimethylhydantoin
ORIGINAL ARTICLES Institut fu¨r Pharmazeutische Chemie der Philipps-Universita¨t, Marburg, Germany Determination of iodine values using 1,3-dibromo-5,5-dimethylhydantoin (DBH) and ethyl acetate as solvent Analytical methods with DBH in respect to environmental and economical concern, part 18* M. Hilp Received November 03, 2003, accepted November 13, 2003 Dr. Manfred Hilp, private address: Haselhecke 42, D-35041 Marburg, Germany [email protected] Pharmazie 59: 612–614 (2004) Iodine values (iodine numbers) of several fixed oils and lard can be determined in ethyl acetate, an easily biodegredable solvent, instead of chloroform according to PH. EUR. 2002. Iodine monobromide has been replaced by 1,3-dibromo-5,5-dimethylhydantoin (DBH) and potassium iodide (KI) and the reaction time was reduced to 5 min only. However, cod-liver oil and linseed oil require a reaction time of 30 min and a smaller weight of sample. Longer reaction times are also necessary for soya oil and wheat germ oil. Iodine values of linseed oil determined according to method A of PH. EUR. 2002, are dependent on the amount of sample, even in the range prescribed by the pharmacopoeia. 1. Introduction oils and fats using only ethyl acetate instead of chloro- form and without the employment of nonionogenic emul- The iodine value (iodine number) is characteristic for the sifiers. content of unsaturated fatty acids in fats, fixed oils, emul- sifiers and solubilizers (Hartke et al. 1999). Halogen (Hilp 2002a; Imming and Germershaus 2002) is added to the 2. Investigations, results, and discussion double bonds. After the addition of potassium iodide the As shown in the Table chloroform can be replaced by excess of the halogenating reagent reacts to iodine, which ethyl acetate analysing fixed oils, and lard. -

A Critical Study of the Precision and Value of Halogen Addition Reactions Applied to Dairy Research Delbert F
Iowa State University Capstones, Theses and Retrospective Theses and Dissertations Dissertations 1938 A critical study of the precision and value of halogen addition reactions applied to dairy research Delbert F. Breazeale Iowa State College Follow this and additional works at: https://lib.dr.iastate.edu/rtd Part of the Biochemistry Commons, and the Chemistry Commons Recommended Citation Breazeale, Delbert F., "A critical study of the precision and value of halogen addition reactions applied to dairy research " (1938). Retrospective Theses and Dissertations. 13448. https://lib.dr.iastate.edu/rtd/13448 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Retrospective Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. INFORMATION TO USERS This manuscript has been reproduced from the microfilm master. UMI films the text directly from the original or copy submitted. Thus, some thesis and dissertation copies are in typewriter face, while others may be from any type of computer printer. The quality of this reproduction is dependent upon the quality of the copy submitted. Broken or indistinct print, colored or poor quality illustrations and photographs, print bleedthrough, substandard margins, and improper alignment can adversely affect reproduction. In the unlikely event that the author did not send UMI a complete manuscript and there are missing pages, these will be noted. Also, if unauthorized copyright material had to be removed, a note will indicate the deletion. -

Safety Data Sheet
SAFETY DATA SHEET Creation Date 08-April-2009 Revision Date 14-February-2020 Revision Number 2 1. Identification Product Name Iodine monobromide Cat No. : A16158 CAS-No 7789-33-5 Synonyms Iodine Bromide. Recommended Use Laboratory chemicals. Uses advised against Food, drug, pesticide or biocidal product use. Details of the supplier of the safety data sheet Company Importer/Distributor Manufacturer Fisher Scientific Alfa Aesar 112 Colonnade Road, Thermo Fisher Scientific Chemicals, Inc. Ottawa, ON K2E 7L6, 30 Bond Street, Ward Hill, MA 01835-8099 Canada Tel: 800-343-0660 Fax: 800-322-4757 Tel: 1-800-234-7437 Email: [email protected] www.alfa.com Emergency Telephone Number During normal business hours (Monday-Friday, 8am-7pm EST), call (800) 343-0660. After normal business hours, call Carechem 24 at (800) 579-7421. 2. Hazard(s) identification Classification WHMIS 2015 Classification Classified as hazardous under the Hazardous Products Regulations (SOR/2015-17) Skin Corrosion/Irritation Category 1 B Serious Eye Damage/Eye Irritation Category 1 Specific target organ toxicity (single exposure) Category 3 Target Organs - Respiratory system. Label Elements Signal Word Danger Hazard Statements Causes severe skin burns and eye damage May cause respiratory irritation ______________________________________________________________________________________________ Page 1 / 7 Iodine monobromide Revision Date 14-February-2020 ______________________________________________________________________________________________ Precautionary Statements Manufacturer Alfa Aesar Thermo Fisher Scientific Chemicals, Inc. 30 Bond Street, Ward Hill, MA 01835-8099 Tel: 800-343-0660 Fax: 800-322-4757 Email: [email protected] www.alfa.com Prevention Do not breathe dust/fumes/gas/mist/vapours/spray Wash face, hands and any exposed skin thoroughly after handling Use only outdoors or in a well-ventilated area Wear protective gloves/protective clothing/eye protection/face protection Response IF SWALLOWED: Rinse mouth. -
The Complexes of Benzene with Halogens and Interhalogens in Carbon Tetrachloride* **
Daehan Hwahak Hwoejee Volume 9, Number 4, 1965 Printed in Republic of Korea. The Complexes of Benzene with Halogens and Interhalogens in Carbon Tetrachloride* ** by Sang Up Choi, Suck Joo Chang and Shoon Ja Kwon Department of Chemistry, Hanyang University (Received September 17, 1965) 벤젠과 할로겐 또는 할로겐間化合物 사이의 錯物에 관한 연구 漢陽大學校 化學科 崔相案 -張石柱 -權純子 (1965. 9. 17 受理 ) 要 糸勺 벤젠과 요오드 , 브롬 , 염소 또는 일브롬화요오드 와의 각 系를 사염화탄소 용액에서 分光光度法 에 의하 여 연구한 결과 C6H6.X2 또는 C&H6-IX형의 錯物이 형성됨을 알았다 (X 는 할로겐 原子를 표시 함 ) . 이들 錯物의 생성에 대한 25。에서의 평형상수 는 다음과 같다 . C6H6.I2 0.173 I mole-i, C6HS-Br2 0.137 I mole-\ C6H6-CI2 0.0643 I mo!e-\ C6H6-IBr 0. 341 I mole-L 이 결과와 다른 사람들의 결과를 종합 하면 벤젠과의 錯物의 상대적 안정도가 다음 순서로 감소함을 알 수 있다 . lCl>IBr>I2>Br2>Cl2 이 순서는 그들의 상대적 산도를 의미하며 할로겐 분자의 편극율 및 할로겐 원자의 전기음성도의 차이 에 의하여 설명할 수 있다 . Abstract Ultraviolet spectrophotometric investigations have been carried out on the systems of benzene with iodine, bromine, chlorine and iodine monobromide in carbon tetrachloride. The results reveal the formation of one to one molecular complexes of the type, CgHe* X2 or CgHg, IX (X denotes halogen atoms). The equilibrium constants obtained at 25° for the complex formation are 0.173, 0.137, 0. 0643 and 0- 341 / mole-1 for *Br2CeH6 t CgHg-C^ and CeHg'IBr, respectively. These results combined with those obtained by other workers indicate that the relative stabilities of the benzene complexes decrease in the order, lCl>IBr>I2>Br2>Cl2. -

Wo 2007/044666 A2
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (43) International Publication Date PCT (10) International Publication Number 19 April 2007 (19.04.2007) WO 2007/044666 A2 (51) International Patent Classification: Not classified (81) Designated States (unless otherwise indicated, for every kind of national protection available): AE, AG, AL, AM, (21) International Application Number: AT,AU, AZ, BA, BB, BG, BR, BW, BY, BZ, CA, CH, CN, PCT/US2006/039394 CO, CR, CU, CZ, DE, DK, DM, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, HN, HR, HU, ID, IL, IN, IS, JP, (22) International Filing Date: 6 October 2006 (06.10.2006) KE, KG, KM, KN, KP, KR, KZ, LA, LC, LK, LR, LS, LT, LU, LV,LY,MA, MD, MG, MK, MN, MW, MX, MY, MZ, (25) Filing Language: English NA, NG, NI, NO, NZ, OM, PG, PH, PL, PT, RO, RS, RU, SC, SD, SE, SG, SK, SL, SM, SV, SY, TJ, TM, TN, TR, (26) Publication Language: English TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW (30) Priority Data: (84) Designated States (unless otherwise indicated, for every 60/724,129 6 October 2005 (06.10.2005) US kind of regional protection available): ARIPO (BW, GH, GM, KE, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, ZM, (71) Applicant (for all designated States except US): E. I. DU ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, TM), PONT DE NEMOURS AND COMPANY [US/US] ; 1007 European (AT,BE, BG, CH, CY, CZ, DE, DK, EE, ES, FI, Market Street, Wilmington, DE 19898 (US). -

Interhalogen.Pdf
Interhalogen 1 Interhalogen The halogens react with each other to form interhalogen compounds. The general formula of most interhalogen compounds is XY , where n = 1, 3, 5 or 7, and X is the less n electronegative of the two halogens. They are all prone to hydrolysis, and ionise to give rise to polyatomic ions. Diatomic interhalogens The interhalogens of form XY have physical properties intermediate between those of the two parent halogens. The covalent bond between the two atoms has some ionic character, the less electronegative element, X, being oxidised and having a partial positive charge. Most combinations of F, Cl, Br and I are known, but not all are stable. Chlorine monofluoride (ClF) The lightest interhalogen compound, ClF is a colorless gas with a normal boiling point of Ä100ÄÅC. Bromine monofluoride (BrF) BrF has not been obtained pure Å it dissociates into the trifluoride and free bromine. Iodine monofluoride (IF) IF is unstable and decomposes at 0 C, disproportionating into elemental iodine and iodine pentafluoride. Bromine monochloride (BrCl) A red-brown gas with a boiling point of 5 ÅC. Iodine monochloride (ICl) Red transparent crystals which melt at 27.2Å C to form a choking brownish liquid (similar in appearance and weight to bromine). It reacts with HCl to form the strong acid HICl . The crystal structure of iodine monochloride consists 2 of puckered zig-zag chains, with strong interactions between the chains. Iodine monobromide (IBr) Made by direct combination of the elements to form a dark red crystalline solid. It melts at 42Å C and boils at 116Å C to form a partially dissociated vapour.