Nucleoside Diphosphate Kinases (Ndpks) in Animal Development
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

METACYC ID Description A0AR23 GO:0004842 (Ubiquitin-Protein Ligase
Electronic Supplementary Material (ESI) for Integrative Biology This journal is © The Royal Society of Chemistry 2012 Heat Stress Responsive Zostera marina Genes, Southern Population (α=0. -

Phosphatidylinositol-3-Kinase in Tomato (Solanum Lycopersicum. L) Fruit and Its Role in Ethylene Signal Transduction and Senescence
Phosphatidylinositol-3-Kinase in Tomato (Solanum lycopersicum. L) Fruit and Its Role in Ethylene Signal Transduction and Senescence by Mohd Sabri Pak Dek A Thesis presented to The University of Guelph In partial fulfilment of requirements for the degree of Doctor of Philosophy in Plant Agriculture Guelph, Ontario, Canada © Mohd Sabri Pak Dek,June, 2015 ABSTRACT PHOSPHATIDYLINOSITOL-3-KINASE IN TOMATO (SOLANUM LYCOPERSICUM. L) FRUIT AND ITS ROLE IN ETHYLENE SIGNAL TRANSDUCTION AND SENESCENCE Mohd Sabri Pak Dek Co-Advisors: University of Guelph, 2015 Professor G. Paliyath Professor J. Subramanian The ripening process is initiated by ethylene through a signal transduction cascade leads to the expression of ripening-related genes and catabolism of membrane, cell wall, and storage components. One of the minor components in membrane phospholipids is phosphatidylinositol (PI). Phosphatidylinositol-3-kinase (PIK) is an enzyme that phosphorylates PI at the 3-OH position of inositol head group to produce phosphatidylinositol 3-phosphate (PI3P). Phosphorylation of PI may be an early event in the ethylene signal transduction pathway that generates negatively charged domains on the plasma membrane. PI3P domains may potentially serve as a docking site for phospholipase D (PLD) after ethylene stimulation. It is hypothesized that ethylene stimulation may activate PI3K resulting in enhanced level of phosphorylated phosphatidylinositol. However, the properties and function of PI3K is not well understood in plants. In the present study, the effect of PI3K inhibition during tomato fruit ripening was evaluated. This study demonstrated that PI3K activity is required for normal ripening process of the fruit. Inhibition of PI3K activity using wortmannin significantly reduced tomato ripening process. -

Structure-Function Analysis of the Catalytic Domain of the Histidine Kinase Chea
Loyola University Chicago Loyola eCommons Dissertations Theses and Dissertations 1997 Structure-Function Analysis of the Catalytic Domain of the Histidine Kinase Chea Dolph David Ellefson Loyola University Chicago Follow this and additional works at: https://ecommons.luc.edu/luc_diss Part of the Microbiology Commons Recommended Citation Ellefson, Dolph David, "Structure-Function Analysis of the Catalytic Domain of the Histidine Kinase Chea" (1997). Dissertations. 3425. https://ecommons.luc.edu/luc_diss/3425 This Dissertation is brought to you for free and open access by the Theses and Dissertations at Loyola eCommons. It has been accepted for inclusion in Dissertations by an authorized administrator of Loyola eCommons. For more information, please contact [email protected]. This work is licensed under a Creative Commons Attribution-Noncommercial-No Derivative Works 3.0 License. Copyright © 1997 Dolph David Ellefson LOYOLA UNIVERSITY MEDICAL CENTER LIBRARY LOYOLA UNIVERSITY OF CHICAGO STRUCTURE-FUNCTION ANALYSIS OF THE CATALYTIC DOMAIN OF THE HISTIDINE KINASE CHEA A DISSERTATION SUBMITTED TO THE FACULTY OF THE GRADUATE SCHOOL IN CANDIDACY FOR THE DEGREE OF DOCTOR OF PHILOSOPHY DEPARTMENT OF MICROBIOLOGY AND IMMUNOLOGY BY DOLPH DAVID ELLEFSON CHICAGO, ILLINOIS MAY, 1997 Copyright by Dolph David Ellefson, 1997 All Rights Reserved ii ACKNOWLEDGEMENTS I would like to thank my director, Dr. Alan J. Wolfe, for his support, advice, and encouragment during the many years in his laboratory. In his laboratory, I was given a rare opportunity to explore a new arena of science and interact with a field of gifted researchers who I would not known otherwise. I would also like to thank the members of my committee, Ors. -

A Thesis Entitled Phor, Phop and Mshc
A Thesis entitled PhoR, PhoP and MshC: Three essential proteins of Mycobacterium tuberculosis by Erica Loney Submitted to the Graduate Faculty as partial fulfillment of the requirements for the Master of Science Degree in Chemistry __________________________________ Dr. Donald R. Ronning, Committee Chair __________________________________ Dr. John J. Bellizzi, Committee Member __________________________________ Dr. Ronald Viola, Committee Member __________________________________ Dr. Patricia R. Komuniecki, Dean College of Graduate Studies The University of Toledo May 2014 Copyright 2014, Erica Loney This document is copyrighted material. Under copyright law, no parts of this document may be produced without the expressed permission of the author. An Abstract of PhoR, PhoP and MshC: Three essential proteins of Mycobacterium tuberculosis by Erica Loney Submitted to the Graduate Faculty as partial fulfillment of the requirements for the Master of Science Degree in Chemistry The University of Toledo May 2014 The tuberculosis (TB) pandemic is responsible for 1.6 million deaths annually, most of which occur in developing nations. TB is treatable, though patient non- compliance, co-infection with HIV, and the long, 6-9 month treatment regimen have resulted in the emergence of drug-resistant TB. For these reasons, the development of novel anti-tuberculin drugs is essential. Three proteins – PhoR, PhoP, and MshC – of Mycobacterium tuberculosis (M.tb), the causative agent of TB, are the focus of this thesis. The PhoPR two-component system is a phosphorelay system responsible for the virulence of M.tb. The histidine kinase PhoR responds to a yet-unknown environmental stimulus and autophosphorylates a conserved histidine. The phosphate is transferred to an aspartate of the response regulator PhoP, which then forms a head-to-head homodimer and initiates the transcription of 114 virulence genes. -

Protein Identities in Evs Isolated from U87-MG GBM Cells As Determined by NG LC-MS/MS
Protein identities in EVs isolated from U87-MG GBM cells as determined by NG LC-MS/MS. No. Accession Description Σ Coverage Σ# Proteins Σ# Unique Peptides Σ# Peptides Σ# PSMs # AAs MW [kDa] calc. pI 1 A8MS94 Putative golgin subfamily A member 2-like protein 5 OS=Homo sapiens PE=5 SV=2 - [GG2L5_HUMAN] 100 1 1 7 88 110 12,03704523 5,681152344 2 P60660 Myosin light polypeptide 6 OS=Homo sapiens GN=MYL6 PE=1 SV=2 - [MYL6_HUMAN] 100 3 5 17 173 151 16,91913397 4,652832031 3 Q6ZYL4 General transcription factor IIH subunit 5 OS=Homo sapiens GN=GTF2H5 PE=1 SV=1 - [TF2H5_HUMAN] 98,59 1 1 4 13 71 8,048185945 4,652832031 4 P60709 Actin, cytoplasmic 1 OS=Homo sapiens GN=ACTB PE=1 SV=1 - [ACTB_HUMAN] 97,6 5 5 35 917 375 41,70973209 5,478027344 5 P13489 Ribonuclease inhibitor OS=Homo sapiens GN=RNH1 PE=1 SV=2 - [RINI_HUMAN] 96,75 1 12 37 173 461 49,94108966 4,817871094 6 P09382 Galectin-1 OS=Homo sapiens GN=LGALS1 PE=1 SV=2 - [LEG1_HUMAN] 96,3 1 7 14 283 135 14,70620005 5,503417969 7 P60174 Triosephosphate isomerase OS=Homo sapiens GN=TPI1 PE=1 SV=3 - [TPIS_HUMAN] 95,1 3 16 25 375 286 30,77169764 5,922363281 8 P04406 Glyceraldehyde-3-phosphate dehydrogenase OS=Homo sapiens GN=GAPDH PE=1 SV=3 - [G3P_HUMAN] 94,63 2 13 31 509 335 36,03039959 8,455566406 9 Q15185 Prostaglandin E synthase 3 OS=Homo sapiens GN=PTGES3 PE=1 SV=1 - [TEBP_HUMAN] 93,13 1 5 12 74 160 18,68541938 4,538574219 10 P09417 Dihydropteridine reductase OS=Homo sapiens GN=QDPR PE=1 SV=2 - [DHPR_HUMAN] 93,03 1 1 17 69 244 25,77302971 7,371582031 11 P01911 HLA class II histocompatibility antigen, -

Expression, Purification and Characterisation of Full-Length
B doi:10.1016/S0022-2836(02)00424-2 available online at http://www.idealibrary.com on w J. Mol. Biol. (2002) 320, 201–213 Expression, Purification and Characterisation of Full-length Histidine Protein Kinase RegB from Rhodobacter sphaeroides Christopher A. Potter1, Alison Ward1, Cedric Laguri2 Michael P. Williamson2, Peter J.F. Henderson1 and Mary K. Phillips-Jones1* 1Division of Microbiology The global redox switch between aerobic and anaerobic growth in School of Biochemistry and Rhodobacter sphaeroides is controlled by the RegA/RegB two-component Molecular Biology, University system, in which RegB is the integral membrane histidine protein kinase, of Leeds, Leeds LS2 9JT, UK and RegA is the cytosolic response regulator. Despite the global regulatory importance of this system and its many homologues, there have been no 2Department of Molecular reported examples to date of heterologous expression of full-length RegB Biology and Biotechnology or any histidine protein kinases. Here, we report the amplified expression University of Sheffield of full-length functional His-tagged RegB in Escherichia coli, its purifi- Sheffield S10 2TN, UK cation, and characterisation of its properties. Both the membrane-bound and purified solubilised RegB protein demonstrate autophosphorylation activity, and the purified protein autophosphorylates at the same rate under both aerobic and anaerobic conditions confirming that an additional regulator is required to control/inhibit autophosphorylation. The intact protein has similar activity to previously characterised soluble forms, but is dephosphorylated more rapidly than the soluble form (half- life ca 30 minutes) demonstrating that the transmembrane segment present in the full-length RegB may be an important regulator of RegB activity. -

The Function of NM23-H1/NME1 and Its Homologs in Major Processes Linked to Metastasis
University of Dundee The Function of NM23-H1/NME1 and Its Homologs in Major Processes Linked to Metastasis Mátyási, Barbara; Farkas, Zsolt; Kopper, László; Sebestyén, Anna; Boissan, Mathieu; Mehta, Anil Published in: Pathology and Oncology Research DOI: 10.1007/s12253-020-00797-0 Publication date: 2020 Licence: CC BY Document Version Publisher's PDF, also known as Version of record Link to publication in Discovery Research Portal Citation for published version (APA): Mátyási, B., Farkas, Z., Kopper, L., Sebestyén, A., Boissan, M., Mehta, A., & Takács-Vellai, K. (2020). The Function of NM23-H1/NME1 and Its Homologs in Major Processes Linked to Metastasis. Pathology and Oncology Research, 26(1), 49-61. https://doi.org/10.1007/s12253-020-00797-0 General rights Copyright and moral rights for the publications made accessible in Discovery Research Portal are retained by the authors and/or other copyright owners and it is a condition of accessing publications that users recognise and abide by the legal requirements associated with these rights. • Users may download and print one copy of any publication from Discovery Research Portal for the purpose of private study or research. • You may not further distribute the material or use it for any profit-making activity or commercial gain. • You may freely distribute the URL identifying the publication in the public portal. Take down policy If you believe that this document breaches copyright please contact us providing details, and we will remove access to the work immediately and investigate your -

Development and Validation of a Protein-Based Risk Score for Cardiovascular Outcomes Among Patients with Stable Coronary Heart Disease
Supplementary Online Content Ganz P, Heidecker B, Hveem K, et al. Development and validation of a protein-based risk score for cardiovascular outcomes among patients with stable coronary heart disease. JAMA. doi: 10.1001/jama.2016.5951 eTable 1. List of 1130 Proteins Measured by Somalogic’s Modified Aptamer-Based Proteomic Assay eTable 2. Coefficients for Weibull Recalibration Model Applied to 9-Protein Model eFigure 1. Median Protein Levels in Derivation and Validation Cohort eTable 3. Coefficients for the Recalibration Model Applied to Refit Framingham eFigure 2. Calibration Plots for the Refit Framingham Model eTable 4. List of 200 Proteins Associated With the Risk of MI, Stroke, Heart Failure, and Death eFigure 3. Hazard Ratios of Lasso Selected Proteins for Primary End Point of MI, Stroke, Heart Failure, and Death eFigure 4. 9-Protein Prognostic Model Hazard Ratios Adjusted for Framingham Variables eFigure 5. 9-Protein Risk Scores by Event Type This supplementary material has been provided by the authors to give readers additional information about their work. Downloaded From: https://jamanetwork.com/ on 10/02/2021 Supplemental Material Table of Contents 1 Study Design and Data Processing ......................................................................................................... 3 2 Table of 1130 Proteins Measured .......................................................................................................... 4 3 Variable Selection and Statistical Modeling ........................................................................................ -

Assessing the Human Canonical Protein Count[Version 1; Peer Review
F1000Research 2017, 6:448 Last updated: 15 JUL 2020 REVIEW Last rolls of the yoyo: Assessing the human canonical protein count [version 1; peer review: 1 approved, 2 approved with reservations] Christopher Southan IUPHAR/BPS Guide to Pharmacology, Centre for Integrative Physiology, University of Edinburgh, Edinburgh, EH8 9XD, UK First published: 07 Apr 2017, 6:448 Open Peer Review v1 https://doi.org/10.12688/f1000research.11119.1 Latest published: 07 Apr 2017, 6:448 https://doi.org/10.12688/f1000research.11119.1 Reviewer Status Abstract Invited Reviewers In 2004, when the protein estimate from the finished human genome was 1 2 3 only 24,000, the surprise was compounded as reviewed estimates fell to 19,000 by 2014. However, variability in the total canonical protein counts version 1 (i.e. excluding alternative splice forms) of open reading frames (ORFs) in 07 Apr 2017 report report report different annotation portals persists. This work assesses these differences and possible causes. A 16-year analysis of Ensembl and UniProtKB/Swiss-Prot shows convergence to a protein number of ~20,000. The former had shown some yo-yoing, but both have now plateaued. Nine 1 Michael Tress, Spanish National Cancer major annotation portals, reviewed at the beginning of 2017, gave a spread Research Centre (CNIO), Madrid, Spain of counts from 21,819 down to 18,891. The 4-way cross-reference concordance (within UniProt) between Ensembl, Swiss-Prot, Entrez Gene 2 Elspeth A. Bruford , European Molecular and the Human Gene Nomenclature Committee (HGNC) drops to 18,690, Biology Laboratory, Hinxton, UK indicating methodological differences in protein definitions and experimental existence support between sources. -

Detailed Characterization of Human Induced Pluripotent Stem Cells Manufactured for Therapeutic Applications
Stem Cell Rev and Rep DOI 10.1007/s12015-016-9662-8 Detailed Characterization of Human Induced Pluripotent Stem Cells Manufactured for Therapeutic Applications Behnam Ahmadian Baghbaderani 1 & Adhikarla Syama2 & Renuka Sivapatham3 & Ying Pei4 & Odity Mukherjee2 & Thomas Fellner1 & Xianmin Zeng3,4 & Mahendra S. Rao5,6 # The Author(s) 2016. This article is published with open access at Springerlink.com Abstract We have recently described manufacturing of hu- help determine which set of tests will be most useful in mon- man induced pluripotent stem cells (iPSC) master cell banks itoring the cells and establishing criteria for discarding a line. (MCB) generated by a clinically compliant process using cord blood as a starting material (Baghbaderani et al. in Stem Cell Keywords Induced pluripotent stem cells . Embryonic stem Reports, 5(4), 647–659, 2015). In this manuscript, we de- cells . Manufacturing . cGMP . Consent . Markers scribe the detailed characterization of the two iPSC clones generated using this process, including whole genome se- quencing (WGS), microarray, and comparative genomic hy- Introduction bridization (aCGH) single nucleotide polymorphism (SNP) analysis. We compare their profiles with a proposed calibra- Induced pluripotent stem cells (iPSCs) are akin to embryonic tion material and with a reporter subclone and lines made by a stem cells (ESC) [2] in their developmental potential, but dif- similar process from different donors. We believe that iPSCs fer from ESC in the starting cell used and the requirement of a are likely to be used to make multiple clinical products. We set of proteins to induce pluripotency [3]. Although function- further believe that the lines used as input material will be used ally identical, iPSCs may differ from ESC in subtle ways, at different sites and, given their immortal status, will be used including in their epigenetic profile, exposure to the environ- for many years or even decades. -

Supplementary Information
Supplementary information (a) (b) Figure S1. Resistant (a) and sensitive (b) gene scores plotted against subsystems involved in cell regulation. The small circles represent the individual hits and the large circles represent the mean of each subsystem. Each individual score signifies the mean of 12 trials – three biological and four technical. The p-value was calculated as a two-tailed t-test and significance was determined using the Benjamini-Hochberg procedure; false discovery rate was selected to be 0.1. Plots constructed using Pathway Tools, Omics Dashboard. Figure S2. Connectivity map displaying the predicted functional associations between the silver-resistant gene hits; disconnected gene hits not shown. The thicknesses of the lines indicate the degree of confidence prediction for the given interaction, based on fusion, co-occurrence, experimental and co-expression data. Figure produced using STRING (version 10.5) and a medium confidence score (approximate probability) of 0.4. Figure S3. Connectivity map displaying the predicted functional associations between the silver-sensitive gene hits; disconnected gene hits not shown. The thicknesses of the lines indicate the degree of confidence prediction for the given interaction, based on fusion, co-occurrence, experimental and co-expression data. Figure produced using STRING (version 10.5) and a medium confidence score (approximate probability) of 0.4. Figure S4. Metabolic overview of the pathways in Escherichia coli. The pathways involved in silver-resistance are coloured according to respective normalized score. Each individual score represents the mean of 12 trials – three biological and four technical. Amino acid – upward pointing triangle, carbohydrate – square, proteins – diamond, purines – vertical ellipse, cofactor – downward pointing triangle, tRNA – tee, and other – circle. -
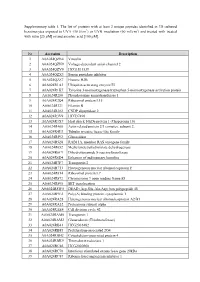
Supplementary Table 1. the List of Proteins with at Least 2 Unique
Supplementary table 1. The list of proteins with at least 2 unique peptides identified in 3D cultured keratinocytes exposed to UVA (30 J/cm2) or UVB irradiation (60 mJ/cm2) and treated with treated with rutin [25 µM] or/and ascorbic acid [100 µM]. Nr Accession Description 1 A0A024QZN4 Vinculin 2 A0A024QZN9 Voltage-dependent anion channel 2 3 A0A024QZV0 HCG1811539 4 A0A024QZX3 Serpin peptidase inhibitor 5 A0A024QZZ7 Histone H2B 6 A0A024R1A3 Ubiquitin-activating enzyme E1 7 A0A024R1K7 Tyrosine 3-monooxygenase/tryptophan 5-monooxygenase activation protein 8 A0A024R280 Phosphoserine aminotransferase 1 9 A0A024R2Q4 Ribosomal protein L15 10 A0A024R321 Filamin B 11 A0A024R382 CNDP dipeptidase 2 12 A0A024R3V9 HCG37498 13 A0A024R3X7 Heat shock 10kDa protein 1 (Chaperonin 10) 14 A0A024R408 Actin related protein 2/3 complex, subunit 2, 15 A0A024R4U3 Tubulin tyrosine ligase-like family 16 A0A024R592 Glucosidase 17 A0A024R5Z8 RAB11A, member RAS oncogene family 18 A0A024R652 Methylenetetrahydrofolate dehydrogenase 19 A0A024R6C9 Dihydrolipoamide S-succinyltransferase 20 A0A024R6D4 Enhancer of rudimentary homolog 21 A0A024R7F7 Transportin 2 22 A0A024R7T3 Heterogeneous nuclear ribonucleoprotein F 23 A0A024R814 Ribosomal protein L7 24 A0A024R872 Chromosome 9 open reading frame 88 25 A0A024R895 SET translocation 26 A0A024R8W0 DEAD (Asp-Glu-Ala-Asp) box polypeptide 48 27 A0A024R9E2 Poly(A) binding protein, cytoplasmic 1 28 A0A024RA28 Heterogeneous nuclear ribonucleoprotein A2/B1 29 A0A024RA52 Proteasome subunit alpha 30 A0A024RAE4 Cell division cycle 42 31