Representation of Molecules
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Catalytic Pyrolysis of Plastic Wastes for the Production of Liquid Fuels for Engines
Electronic Supplementary Material (ESI) for RSC Advances. This journal is © The Royal Society of Chemistry 2019 Supporting information for: Catalytic pyrolysis of plastic wastes for the production of liquid fuels for engines Supattra Budsaereechaia, Andrew J. Huntb and Yuvarat Ngernyen*a aDepartment of Chemical Engineering, Faculty of Engineering, Khon Kaen University, Khon Kaen, 40002, Thailand. E-mail:[email protected] bMaterials Chemistry Research Center, Department of Chemistry and Center of Excellence for Innovation in Chemistry, Faculty of Science, Khon Kaen University, Khon Kaen, 40002, Thailand Fig. S1 The process for pelletization of catalyst PS PS+bentonite PP ) t e PP+bentonite s f f o % ( LDPE e c n a t t LDPE+bentonite s i m s n HDPE a r T HDPE+bentonite Gasohol 91 Diesel 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-1) Fig. S2 FTIR spectra of oil from pyrolysis of plastic waste type. Table S1 Compounds in oils (%Area) from the pyrolysis of plastic wastes as detected by GCMS analysis PS PP LDPE HDPE Gasohol 91 Diesel Compound NC C Compound NC C Compound NC C Compound NC C 1- 0 0.15 Pentane 1.13 1.29 n-Hexane 0.71 0.73 n-Hexane 0.65 0.64 Butane, 2- Octane : 0.32 Tetradecene methyl- : 2.60 Toluene 7.93 7.56 Cyclohexane 2.28 2.51 1-Hexene 1.05 1.10 1-Hexene 1.15 1.16 Pentane : 1.95 Nonane : 0.83 Ethylbenzen 15.07 11.29 Heptane, 4- 1.81 1.68 Heptane 1.26 1.35 Heptane 1.22 1.23 Butane, 2,2- Decane : 1.34 e methyl- dimethyl- : 0.47 1-Tridecene 0 0.14 2,2-Dimethyl- 0.63 0 1-Heptene 1.37 1.46 1-Heptene 1.32 1.35 Pentane, -

Synthetic Turf Scientific Advisory Panel Meeting Materials
California Environmental Protection Agency Office of Environmental Health Hazard Assessment Synthetic Turf Study Synthetic Turf Scientific Advisory Panel Meeting May 31, 2019 MEETING MATERIALS THIS PAGE LEFT BLANK INTENTIONALLY Office of Environmental Health Hazard Assessment California Environmental Protection Agency Agenda Synthetic Turf Scientific Advisory Panel Meeting May 31, 2019, 9:30 a.m. – 4:00 p.m. 1001 I Street, CalEPA Headquarters Building, Sacramento Byron Sher Auditorium The agenda for this meeting is given below. The order of items on the agenda is provided for general reference only. The order in which items are taken up by the Panel is subject to change. 1. Welcome and Opening Remarks 2. Synthetic Turf and Playground Studies Overview 4. Synthetic Turf Field Exposure Model Exposure Equations Exposure Parameters 3. Non-Targeted Chemical Analysis Volatile Organics on Synthetic Turf Fields Non-Polar Organics Constituents in Crumb Rubber Polar Organic Constituents in Crumb Rubber 5. Public Comments: For members of the public attending in-person: Comments will be limited to three minutes per commenter. For members of the public attending via the internet: Comments may be sent via email to [email protected]. Email comments will be read aloud, up to three minutes each, by staff of OEHHA during the public comment period, as time allows. 6. Further Panel Discussion and Closing Remarks 7. Wrap Up and Adjournment Agenda Synthetic Turf Advisory Panel Meeting May 31, 2019 THIS PAGE LEFT BLANK INTENTIONALLY Office of Environmental Health Hazard Assessment California Environmental Protection Agency DRAFT for Discussion at May 2019 SAP Meeting. Table of Contents Synthetic Turf and Playground Studies Overview May 2019 Update ..... -

Sampling Dynamics for Volatile Organic Compounds Using Headspace Solid-Phase Microextraction Arrow for Microbiological Samples
separations Communication Sampling Dynamics for Volatile Organic Compounds Using Headspace Solid-Phase Microextraction Arrow for Microbiological Samples Kevin E. Eckert 1, David O. Carter 2 and Katelynn A. Perrault 1,* 1 Laboratory of Forensic and Bioanalytical Chemistry, Forensic Sciences Unit, Division of Natural Sciences and Mathematics, Chaminade University of Honolulu, 3140 Waialae Ave., Honolulu, HI 96816, USA; [email protected] 2 Laboratory of Forensic Taphonomy, Forensic Sciences Unit, Division of Natural Sciences and Mathematics, Chaminade University of Honolulu, 3140 Waialae Avenue, Honolulu, HI 96816, USA; [email protected] * Correspondence: [email protected] Received: 10 August 2018; Accepted: 27 August 2018; Published: 10 September 2018 Abstract: Volatile organic compounds (VOCs) are monitored in numerous fields using several commercially-available sampling options. Sorbent-based sampling techniques, such as solid-phase microextraction (SPME), provide pre-concentration and focusing of VOCs prior to gas chromatography–mass spectrometry (GC–MS) analysis. This study investigated the dynamics of SPME Arrow, which exhibits an increased sorbent phase volume and improved durability compared to traditional SPME fibers. A volatile reference mixture (VRM) and saturated alkanes mix (SAM) were used to investigate optimal parameters for microbiological VOC profiling in combination with GC–MS analysis. Fiber type, extraction time, desorption time, carryover, and reproducibility were characterized, in addition to a comparison with traditional SPME fibers. The developed method was then applied to longitudinal monitoring of Bacillus subtilis cultures, which represents a ubiquitous microbe in medical, forensic, and agricultural applications. The carbon wide range/polydimethylsiloxane (CWR/PDMS) fiber was found to be optimal for the range of expected VOCs in microbiological profiling, and a statistically significant increase in the majority of VOCs monitored was observed. -

Vapor Pressures and Vaporization Enthalpies of the N-Alkanes from 2 C21 to C30 at T ) 298.15 K by Correlation Gas Chromatography
BATCH: je1a04 USER: jeh69 DIV: @xyv04/data1/CLS_pj/GRP_je/JOB_i01/DIV_je0301747 DATE: October 17, 2003 1 Vapor Pressures and Vaporization Enthalpies of the n-Alkanes from 2 C21 to C30 at T ) 298.15 K by Correlation Gas Chromatography 3 James S. Chickos* and William Hanshaw 4 Department of Chemistry and Biochemistry, University of MissourisSt. Louis, St. Louis, Missouri 63121 5 6 The temperature dependence of gas chromatographic retention times for n-heptadecane to n-triacontane 7 is reported. These data are used to evaluate the vaporization enthalpies of these compounds at T ) 298.15 8 K, and a protocol is described that provides vapor pressures of these n-alkanes from T ) 298.15 to 575 9 K. The vapor pressure and vaporization enthalpy results obtained are compared with existing literature 10 data where possible and found to be internally consistent. Sublimation enthalpies for n-C17 to n-C30 are 11 calculated by combining vaporization enthalpies with fusion enthalpies and are compared when possible 12 to direct measurements. 13 14 Introduction 15 The n-alkanes serve as excellent standards for the 16 measurement of vaporization enthalpies of hydrocarbons.1,2 17 Recently, the vaporization enthalpies of the n-alkanes 18 reported in the literature were examined and experimental 19 values were selected on the basis of how well their 20 vaporization enthalpies correlated with their enthalpies of 21 transfer from solution to the gas phase as measured by gas 22 chromatography.3 A plot of the vaporization enthalpies of 23 the n-alkanes as a function of the number of carbon atoms 24 is given in Figure 1. -
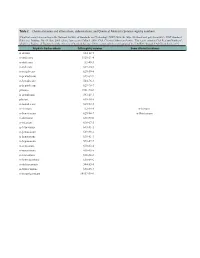
Table 2. Chemical Names and Alternatives, Abbreviations, and Chemical Abstracts Service Registry Numbers
Table 2. Chemical names and alternatives, abbreviations, and Chemical Abstracts Service registry numbers. [Final list compiled according to the National Institute of Standards and Technology (NIST) Web site (http://webbook.nist.gov/chemistry/); NIST Standard Reference Database No. 69, June 2005 release, last accessed May 9, 2008. CAS, Chemical Abstracts Service. This report contains CAS Registry Numbers®, which is a Registered Trademark of the American Chemical Society. CAS recommends the verification of the CASRNs through CAS Client ServicesSM] Aliphatic hydrocarbons CAS registry number Some alternative names n-decane 124-18-5 n-undecane 1120-21-4 n-dodecane 112-40-3 n-tridecane 629-50-5 n-tetradecane 629-59-4 n-pentadecane 629-62-9 n-hexadecane 544-76-3 n-heptadecane 629-78-7 pristane 1921-70-6 n-octadecane 593-45-3 phytane 638-36-8 n-nonadecane 629-92-5 n-eicosane 112-95-8 n-Icosane n-heneicosane 629-94-7 n-Henicosane n-docosane 629-97-0 n-tricosane 638-67-5 n-tetracosane 643-31-1 n-pentacosane 629-99-2 n-hexacosane 630-01-3 n-heptacosane 593-49-7 n-octacosane 630-02-4 n-nonacosane 630-03-5 n-triacontane 638-68-6 n-hentriacontane 630-04-6 n-dotriacontane 544-85-4 n-tritriacontane 630-05-7 n-tetratriacontane 14167-59-0 Table 2. Chemical names and alternatives, abbreviations, and Chemical Abstracts Service registry numbers.—Continued [Final list compiled according to the National Institute of Standards and Technology (NIST) Web site (http://webbook.nist.gov/chemistry/); NIST Standard Reference Database No. -

Comparative Analysis of Volatile Compounds of Gamma-Irradiated Mutants of Rose (Rosa Hybrida)
plants Article Comparative Analysis of Volatile Compounds of Gamma-Irradiated Mutants of Rose (Rosa hybrida) Jaihyunk Ryu 1 , Jae Il Lyu 1 , Dong-Gun Kim 1, Jung-Min Kim 1, Yeong Deuk Jo 1 , Si-Yong Kang 2, Jin-Baek Kim 1 , Joon-Woo Ahn 1 and Sang Hoon Kim 1,* 1 Advanced Radiation Technology Institute, Korea Atomic Energy Research Institute, Jeollabuk-do 56212, Korea; [email protected] (J.R.); [email protected] (J.I.L.); [email protected] (D.-G.K.); [email protected] (J.-M.K.); [email protected] (Y.D.J.); [email protected] (J.-B.K.); [email protected] (J.-W.A.) 2 Department of Horticulture, College of Industrial Sciences, Kongju National University, Yesan 32439, Korea; [email protected] * Correspondence: [email protected]; Tel.: +82-63-570-3318 Received: 22 July 2020; Accepted: 14 September 2020; Published: 17 September 2020 Abstract: Roses are one of the most important floricultural crops, and their essential oils have long been used for cosmetics and aromatherapy. We investigated the volatile compound compositions of 12 flower-color mutant variants and their original cultivars. Twelve rose mutant genotypes were developed by treatment with 70 Gy of 60Co gamma irradiation of six commercial rose cultivars. Essential oils from the flowers of the 18 genotypes were analyzed by gas chromatography–mass spectrometry. Seventy-seven volatile compounds were detected, which were categorized into six classes: Aliphatic hydrocarbons, aliphatic alcohols, aliphatic ester, aromatic compounds, terpene alcohols, and others. Aliphatic (hydrocarbons, alcohols, and esters) compounds were abundant categories in all rose flowers. -

Exhaust Emission Profiles for EPA SPECIATE Database
Exhaust Emission Profiles for EPA SPECIATE Database: Energy Policy Act (EPAct) Low-Level Ethanol Fuel Blends and Tier 2 Light- Duty Vehicles Exhaust Emission Profiles for EPA SPECIATE Database: Energy Policy Act (EPAct) Low-Level Ethanol Fuel Blends and Tier 2 Light- Duty Vehicles Assessment and Standards Division Office of ransportationT and Air Quality U.S. Environmental Protection Agency EPA-420-R-09-002 June 2009 Table of Contents 1.0 Introduction....................................................................................................................... 1 1.1 Background..................................................................................................................... 1 1.2 Purpose of the Project ..................................................................................................... 1 2.0 Methods.............................................................................................................................. 2 2.1 Exhaust Emissions Data.................................................................................................. 2 2.2 Calculation of Composite Profiles.................................................................................. 3 2.3 Adjustment of E0 and E10 profiles with an In-Use Gasoline......................................... 3 2.4 Assignment of SPECIATE Identification Numbers ....................................................... 4 3.0 Results ............................................................................................................................... -

ASTM D2887 Boiling Range Distribution of Petroleum Fractions by GC
Visit us at accustandard.com ASTM D2887 Boiling Range Distribution of Petroleum Fractions by GC Total Petroleum Hydrocarbons (C8 - C20) Total Petroleum Hydrocarbons (C8 - C30) TPH-LOW-01 $ 50 / 1 x 1 mL TPH-MID-01 $ 65 / 1 x 1 mL TPH-LOW-01-PAK SAVE 20% $ 200 / 5 x 1 mL TPH-MID-01-PAK SAVE 20% $ 260 / 5 x 1 mL 500 µg/mL each in Chloroform 13 comps. 500 µg/mL each in Chloroform 25 comps. n-Octane n-Pentadecane UPDATED n-Octane n-Heptadecane n-Tricosane UPDATED n-Nonane n-Hexadecane n-Nonane n-Octadecane n-Tetracosane n-Decane n-Heptadecane n-Decane Pristane n-Pentacosane n-Undecane n-Octadecane n-Undecane n-Nonadecane n-Hexacosane n-Dodecane n-Nonadecane n-Dodecane Phytane n-Heptacosane n-Tridecane n-Eicosane n-Tridecane n-Eicosane n-Octacosane n-Tetradecane n-Tetradecane n-Heneicosane n-Nonacosane n-Pentadecane n-Docosane n-Triacontane n-Hexadecane Calibration Mixture (C6 - C44) Hydrocarbon Window Defining Standard (C8 - C40) DRH-002N $ 30 / 100 mg DRH-008S-R2 $ 65 / 1 x 1 mL DRH-002N-10X $ 40 / 1 gm DRH-008S-R2-PAK SAVE 20% $ 260 / 5 x 1 mL At stated Wt.% 17 comps. 500 µg/mL each in Chloroform 35 comps. n-Hexane 6 n-Octadecane 5 n-Octane n-Nonadecane n-Triacontane n-Heptane 6 n-Eicosane 2 n-Nonane Phytane n-Hentriacontane n-Octane 8 n-Tetracosane 2 n-Decane n-Eicosane n-Dotriacontane n-Nonane 8 n-Octacosane 1 n-Undecane n-Heneicosane n-Tritriacontane n-Decane 12 n-Dotriacontane 1 n-Dodecane n-Docosane n-Tetratriacontane n-Undecane 12 n-Hexatriacontane 1 n-Tridecane n-Tricosane n-Pentatriacontane n-Dodecane 12 n-Tetracontane 1 n-Tetradecane n-Tetracosane n-Hexatriacontane n-Tetradecane 12 n-Tetratetracontane 1 n-Pentadecane n-Pentacosane n-Heptatriacontane n-Hexadecane 10 n-Hexadecane n-Hexacosane n-Octatriacontane n-Heptadecane n-Heptacosane n-Nonatriacontane n-Octadecane n-Octacosane n-Tetracontane Calibration Solution (C6 - C44) DRH-002S-R1 $ 50 / 1 x 1 mL Pristane n-Nonacosane DRH-002S-R1-PAK SAVE 20% $ 200 / 5 x 1 mL At stated conc. -

Control Technology Center EPA-600 12-91-061 November 1991
United States Envimnmentai Protection Agency Control Technology Center EPA-600 12-91-061 November 1991 EVALUATION OF VOC EMISSIONS FROM HEATED ROOFING ASPHALT RESEARCH REPORTING SERIES Research reports of the Office of Research and Development. U.S. Environmental Protection Agency, have been grouped into nine series. These nine broad cate- gories were established to facilitate further development and application of en- vironmental technology. Elimination of traditional grouping was consciously planned to foster technology transfer and a maximum interface in related fields. The nine series are: 1. Environmental Health Effects Research 2. Environmental Protection Technology 3. Ecological Research 4. Environmental Monitoring 5. Socioeconomic Environmental Studies 6. Scientific and Technical Assessment Reports (STAR) 7. Interagency Energy-Environment Research and Development 8. "Special" Reports 9. Miscellaneous Reports This report has been assigned to the ENVIRONMENTAL PROTECT1ON TECH- NOLOGY series. This series describes research performed to develop and dem- onstrate instrumentation, equipment, and methodology to repair or prevent en- vironmental degradation from point and non-point sources of pollution. This work provides the new or improved technology required for the control and treatment of pollution sources to meet environmental quality standards. EPA REVIEW NOTICE This report has been reviewed by the U.S. Environmental Protection Agency, and approved for publication. Approval does not signify that the contents necessarily reflect the views and policy of the Agency, nor does mention of trade names or 'commercial products constitute endorsement or recommendation for use. This document is available to the public through the National Technical Informa- tion Service. Springfield. Virginia 22161. EPA-600/2-91-061 November 1991 EVALUATION OF VOC EMISSIONS FROM HEATED ROOFING ASPHALT Prepared by: Peter Kariher, Michael Tufts, and Larry Hamel Acurex Corporation Environmental Systems Division 49 15 Prospectus Drive P.O. -

Analysis of N-Alkanes and N-Alkenes in Chukchi Sea Sediments By
Analysis of n-Alkanes and n-Alkenes in Chukchi Sea Sediments By Salomey Asantewaa Sasu Bachelor of Science, University of Cape Coast Master of Science, Stephen F. Austin State University A dissertation submitted to the Department of Chemistry and Florida Institute of Technology In partial fulfillment of the requirements for the degree of Doctor of Philosophy In Chemistry Melbourne, Florida July, 2019 Analysis of n-Alkanes and n-Alkenes in Chukchi Sea Sediments A dissertation by Salomey Asantewaa Sasu Approved as to style and content _______________________________________________________________ Kurt Winkelmann, Ph.D., Committee Chairperson Associate Professor, Chemistry _______________________________________________________________ Mary L. Sohn, Ph.D. Professor Emerita, Chemistry ________________________________________________________________ Alan B. Brown, Ph.D. Associate Professor, Chemistry ________________________________________________________________ Robert Weaver, Ph.D. Associate Professor, Ocean Engineering ________________________________________________________________ Ted Conway, Ph.D. Professor and Head, Biomedical and Chemical Engineering and Sciences Abstract Analysis of n-Alkanes and n-Alkenes in Chukchi Sea Sediments By Salomey Asantewaa Sasu Major Advisor: Kurt Winkelmann, Ph.D. This study determined the concentrations, distribution and sources of n-alkanes in core sediments from four locations: Stations H24, H30, H32 and BarC5 in the Chukchi Sea, which is in the Arctic Ocean. n-Alkanes are valuable tracers of aquatic, terrigenous and petroleum inputs and may give evidence to the extent of climate change. The study of climate change in the Arctic is important since the Arctic is warming faster than any other part of Earth. The increase in temperature is causing enhanced evaporation leading to a higher precipitation rate and hence enhanced erosion. The n-alkanes C12-C35 were analysed and characterized using GC-MS in all four core sediments. -

Retention Index Mixture for GC (R8769)
RETENTION INDEX STANDARD For Gas Chromatography SIGMA TECHNICAL BULLETIN #R8769 Product No. R 8769 Description: Sigma Retention Index Standard consists of a mixture of aliphatic hydrocarbons ranging from C8 through C32, dissolved in hexane. It is designed to be used to obtain Kovats-type gas chromatographic retention indices, which are useful for preliminary identification of unknowns and as an aid in GC method development. Components with carbon numbers that are a multiple of five are at 2X concentration to allow easy determination of carbon numbers for peaks of interest. Composition: All components used are 98+% pure and are dissolved in GC grade Hexane at the nominal concentrations listed below: Component µg/mL Component µg/mL Component µg/mL n-Octane (C8) 1000 n-Hexadecane (C16) 1000 n-Tetracosane (C24) 1000 n-Nonane (C9) 1000 n-Heptadecane (C17) 1000 n-Pentacosane (C25) 2000 n-Decane (C10) 2000 n-Octadecane (C18) 1000 n-Hexacosane (C26) 1000 n-Undecane (C11) 1000 n-Nonadecane (C19) 1000 n-Heptacosane (C27) 1000 n-Dodecane (C12) 1000 n-Eicosane (C20) 2000 n-Octacosane (C28) 1000 n-Tridecane (C13) 1000 n-Heneicosane (C21) 1000 n-Nonacosane (C29) 1000 n-Tetradecane (C14) 1000 n-Docosane (C22) 1000 n-Triacontane (C30) 2000 n-Pentadecane (C15) 2000 n-Tricosane (C23) 1000 n-Dotriacontane (C32) 1000 C10 C15 C20 C25 C30 5.00 10.00 15.00 20.00 25.00 30.00 35.00 40.00 45.00 50.00 Time (min.) Typical Temperature Programmed Chromatogram Column 15m x 0.20mm x 0.2 µm Supelco SPB-1 (Cat. No. 2-4162) Oven Temperature 30°C (0 min.), then 5°/min. -

Fuel & Hydrocarbon Standards
Techlab - Matériaux de référence certifiés et Equipements de laboratoire - www.techlab.fr Fuel & Hydrocarbon Standards Fuels & Hydrocarbon Standards (in 1 mL of solvent, unless otherwise noted) Individual Fuel & Hydrocarbons FUEL & HYDROCARBON STANDARDS CAS NO. QTY./CONC. MATRIX CAT. NO. UNIT PRICE 5-alpha Androstane 438-22-2 1 mg/mL CH2Cl2 GRH-IS 1 mL 15 10 mg/mL CH2Cl2 GRH-IS-10X 1 mL 30 Arctic Diesel Fuel 68334-30-5-AR 0.5 mg/mL MeOH FU-023 1 mL 10 20 mg/mL MeOH FU-023-40X 1 mL 20 20 mg/mL CH2Cl2 FU-023-D-40X 1 mL 20 Aviation (gas) grade 100-LL 0.5 mg/mL MeOH GA-004 1 mL 10 20 mg/mL MeOH GA-004-40X 1 mL 20 20 mg/mL CH2Cl2 GA-004-D-40X 1 mL 20 p-Bromofluorobenzene 460-00-4 2.5 mg/mL Acetone GARH-SS 1 mL 20 1-Chloro-4-fluorobenzene 352-33-0 2 mg/mL MeOH AK-101.0-IS-10X 1 mL 20 1-Chlorooctadecane 3386-33-2 1 mg/mL Hexane DRH-007-SS 1 mL 15 1-Chloro-4-fluorobenzene 352-33-0 1 mg/mL CH2Cl2 GARH-IS 1 mL 15 2,5-Dibromotoluene 615-59-8 50 µg/mL MeOH GRH-004-SS 1 mL 10 500 µg/mL MeOH GRH-004-SS-10X 1 mL 20 5 mg/mL MeOH GRH-004-SS-100X 1 mL 20 Diesel Fuel 0.5 mg/mL MeOH FU-009 1 mL 10 5 mg/mL CH2Cl2 FU-009-D-10X 1 mL 20 20 mg/mL MeOH FU-009-40X 1 mL 20 20 mg/mL CH2Cl2 FU-009-D-40X 1 mL 20 #1 Diesel - Low Sulfur 0.5 mg/mL MeOH FU-013 1 mL 10 20 mg/mL MeOH FU-013-40X 1 mL 20 20 mg/mL CH2Cl2 FU-013-D-40X 1 mL 20 #2 Diesel Fuel 68334-30-5 50 mg/mL Acetone AK-102-LCS-R1-10X 1 mL 20 #2 Diesel Fuel (Extra Low Sulfur) 68476-43-6 0.5 mg/mL MeOH FU-017 1 mL 10 5 mg/mL CH2Cl2 FU-017-D-10X 1 mL 20 5 mg/mL Acetone AK-102.0-LCS 1 mL 10 50 mg/mL Acetone AK-102.0-LCS-10X