United States Patent Office 2,220,845 Aromatic
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Phosphorus Oxychloride CAS No
Product Safety Summary Phosphorus Oxychloride CAS No. 10025-87-3 This Product Safety Summary is intended to provide a general overview of the chemical substance. The information in the summary is basic information and is not intended to provide emergency response information, medical information or treatment information. The summary should not be used to provide in-depth safety and health information. In-depth safety and health information can be found in the Safety Data Sheet (SDS) for the chemical substance. Names Phosphorus oxychloride Phosphorus trichloride oxide Phosphoric trichloride POCl 3 Product Overview Solvay Novecare does not sell phosphorus oxychloride directly to consumers. Phosphorus oxychloride (POCl3) is used as a chemical intermediate to produce a variety of products which are used in several applications including manufacture of triarylphosphate esters which are used as flame retardants as well as an intermediate in the production of pharmaceutical, textile and agricultural chemicals. Phosphorus oxychloride is used in industrial applications and other processes where workplace exposures can occur. Consumer exposure does not occur as phosphorus oxychloride is not used in any commercially available product. Phosphorus oxychloride is dangerous to human health. Phosphorus oxychloride may be fatal if inhaled, highly toxic if swallowed, harmful if absorbed through skin and can cause severe burns which may result in scarring. Phosphorus oxychloride is consumed in manufacturing processes. POCl3 can make its way into the environment through unintentional releases (spills). POCl3 will not bioaccumulate but is not biodegradable. POCl3 in higher concentrations can be harmful to aquatic life due to formation of acids from the hydrolysis of POCl3. When released into the atmosphere, phosphorus oxychloride exists as vapor. -

5 Phosphorus Oxychloride1 Acute Exposure Guideline Levels
Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 10 Committee on Acute Exposure Guideline Levels Committee on Toxicology Board on Environmental Studies and Toxicology Division on Earth and Life Studies Copyright © National Academy of Sciences. All rights reserved. Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 10 THE NATIONAL ACADEMIES PRESS 500 FIFTH STREET, NW WASHINGTON, DC 20001 NOTICE: The project that is the subject of this report was approved by the Governing Board of the National Research Council, whose members are drawn from the councils of the National Academy of Sciences, the National Academy of Engineering, and the Insti- tute of Medicine. The members of the committee responsible for the report were chosen for their special competences and with regard for appropriate balance. This project was supported by Contract No. W81K04-06-D-0023 and EP-W-09-007 be- tween the National Academy of Sciences and the U.S. Department of Defense and the U.S. Environmental Protection Agency. Any opinions, findings, conclusions, or recom- mendations expressed in this publication are those of the author(s) and do not necessarily reflect the view of the organizations or agencies that provided support for this project. International Standard Book Number-13: 978-0-309-21987-7 International Standard Book Number-10: 0-309-21987-6 Additional copies of this report are available from The National Academies Press 500 Fifth Street, NW Box 285 Washington, DC 20055 800-624-6242 202-334-3313 (in the Washington metropolitan area) http://www.nap.edu Copyright 2011 by the National Academy of Sciences. -

NPR72: the Pavlodar Chemical Weapons Plant in Kazakhstan
GULBARSHYN BOZHEYEVA Report The Pavlodar Chemical Weapons Plant in Kazakhstan: History and Legacy GULBARSHYN BOZHEYEVA Gulbarshyn Bozheyeva holds a Ph.D. degree in Chemical Physics from the Kazakh State University. From 1996-97, she was a Visiting Scholar and Research Associate at the Center for Nonproliferation Studies at the Monterey Institute of International Studies. She is now completing a master’s degree in International Development Policy at Duke University. he former Soviet Union’s chemical weapons theless, manufacturing lines and equipment for primary (CW) program consisted of many production and intermediate CW precursors and buildings for fill- plants that created the world’s largest stockpile ing CW munitions were constructed at Pavlodar. The T 1 of chemical weapons. Most of the CW production and plant also acquired personnel with expertise in CW pro- storage facilities were located in Russia, but a few fa- duction.5 cilities existed in other Soviet republics. In recent years, This report is devoted to the role of the Pavlodar Western countries have provided significant financial Chemical Plant in the former Soviet CW program and assistance for dismantling former CW facilities in Rus- its current status. The first part of the report describes sia and converting former CW production facilities for the history of the Pavlodar plant and its military and 2 commercial use. Although a fair amount has been writ- civilian infrastructures. The second part deals with the ten about Russian CW facilities, little is known about CW capability of the plant and the nature of the chemi- the CW programs in other former Soviet republics. -

United States Patent Office Patented Oct
3,346,562 United States Patent Office Patented Oct. 10, 1967 2 3,346,562 cg METHOD FOR THE PRODUCTION OF PO-CE Base RBONUCLEOSDE-5'-PHOSPHATE / O Mikio Honjo, Takatsuki, and Ryuji Maremoto, Minoo, Cl EO Japan, assignors to Takeda Chemical industries, Ltd., Osaka, Japan No Drawing. Filed May 31, 1966, Ser. No. 553,718 Claims priority, application Japan, May 29, 1965, R. X R. 40/31,814 9 Claims. (Cl. 260-21.5) HO. O. BIO O 10 N1 N1 This invention is concerned with a method for the pro Po-H, Base Po-H, Base duction of ribonucleoside-5'-phosphate. EIO k" wE+ EO k". Ribonucleoside-5'-phosphate is very useful as condi H HO - ment for food and also in the pharmaceutical industry, O O OH OH and has been chemically produced by at first protecting 15 X the hydroxyl groups at the 2'- and 3'-positions of its ribose R1 R2 moiety with acyl or isopropylidene groups and then phos phorylating the 5'-hydroxyl group of the thus-protected RN compound with pentavalent phosphorus compound such C=O: aliphatic ketone or aromatic aldehyde as phosphorus pentachloride, phosphorus oxychloride, 20 R?2 etc., followed by removing the protecting groups. As "ribonucleoside' in the present method there are However, this hitherto-known method requires a long used those containing purine base such as adenosine, time (about 7 to about 30 hours) for completing the pro inosine, etc. or those containing pyrimidine base such as tection and phosphorylation, and therefore is not desirable uridine, cytidine, etc. As the aliphatic ketone having 3 from an industrial viewpoint. -
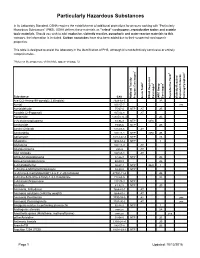
Particularly Hazardous Substances
Particularly Hazardous Substances In its Laboratory Standard, OSHA requires the establishment of additional protections for persons working with "Particularly Hazardous Substances" (PHS). OSHA defines these materials as "select" carcinogens, reproductive toxins and acutely toxic materials. Should you wish to add: explosive, violently reactive, pyrophoric and water-reactve materials to this category, the information is included. Carbon nanotubes have also been added due to their suspected carcinogenic properties. This table is designed to assist the laboratory in the identification of PHS, although it is not definitively conclusive or entirely comprehensive. *Notes on the proper use of this table appear on page 12. 1 6 5 2 3 4 Substance CAS National Toxicity National Program Carcinogen Toxin Acute Regulated OSHA Carcinogen Group IARC Carcinogen Toxin Reproductive Violently Reactive/ Explosive/Peroxide Forming/Pyrophoric A-a-C(2-Amino-9H-pyrido[2,3,b]indole) 2648-68-5 2B Acetal 105-57-7 yes Acetaldehyde 75-07-0 NTP AT 2B Acrolein (2-Propenal) 107-02-8 AT Acetamide 126850-14-4 2B 2-Acetylaminofluorene 53-96-3 NTP ORC Acrylamide 79-06-6 NTP 2B Acrylyl Chloride 814-68-6 AT Acrylonitrile 107-13-1 NTP ORC 2B Adriamycin 23214-92-8 NTP 2A Aflatoxins 1402-68-2 NTP 1 Allylamine 107-11-9 AT Alkylaluminums varies AT Allyl Chloride 107-05-1 AT ortho-Aminoazotoluene 97-56-3 NTP 2B para-aminoazobenzene 60-09-3 2B 4-Aminobiphenyl 92-67-1 NTP ORC 1 1-Amino-2-Methylanthraquinone 82-28-0 NTP (2-Amino-6-methyldipyrido[1,2-a:3’,2’-d]imidazole) 67730-11-4 2B -

Product Safety Summary
Product Safety Summary Phosphorus Trichloride CAS No. 7719-12-2 This Product Safety Summary is intended to provide a general overview of the chemical substance. The information in the summary is basic information and is not intended to provide emergency response information, medical information or treatment information. The summary should not be used to provide in-depth safety and health information. In-depth safety and health information can be found in the Safety Data Sheet (SDS) for the chemical substance. Names Phosphorus trichloride Phosphorus (III) chloride Phosphorous chloride PCl 3 Product Overview Solvay Novecare does not sell phosphorus trichloride directly to consumers and phosphorus trichloride has no known uses except as an intermediate to produce other chemicals. Phosphorus trichloride (PCl3) is used as a chemical intermediate to produce a variety of products which are used in several applications including agricultural products, surfactants and metal extractants, flame retardants, additives for lubricants and stabilizers for plastics. Phosphorus trichloride is used in industrial applications and other processes where workplace exposures can occur. Consumer exposure does not occur as phosphorus trichloride is not used in any commercially available product. Phosphorus trichloride is dangerous to human health. Phosphorus trichloride may be fatal if inhaled, highly toxic if swallowed, harmful if absorbed through skin and can cause severe burns which may result in scarring. Phosphorus trichloride is consumed in manufacturing processes. PCl3 can make its way into the environment through unintentional releases (spills). PCl3 will not bioaccumulate but is not biodegradable. Based on ecotoxicological testing performed on fish and fresh-water invertebrates, PCl3 in higher concentrations can be harmful to aquatic life due to formation of acids from the hydrolysis of PCl3. -

UNITED STATES PATENT OFFICE 2,683,168 PREPARATHON of ORGANO PHOSPHONY, CHELORADES Warren Jensen, Ponca City, Okla., and James O
Patented July 6, 1954 2,683,168 UNITED STATES PATENT OFFICE 2,683,168 PREPARATHON OF ORGANO PHOSPHONY, CHELORADES Warren Jensen, Ponca City, Okla., and James O. Claytoia, Berkeley, Calif., assigno's to California, Research Corporation, San Francisco, Calif., a corporation of Delaware No Drawing. Application December 22, 1950, Serial No. 202,396 8 Claims. (CI. 260-543) 1. 2 This invention relates to a method of prepar vide a means of preparing phosphonyl compounds ing phosphonyl chlorides and the like by the wherein the phosphorus atom is directly con reaction of an organic compound with phosphorus nected to an aliphatic carbon aton. trichloride in the presence of oxygen. it is another object of this invention to provide This application is a continuation-in-part of 5 a means of converting an aliphatic carbon-to Our Copending application, Serial No. 86,856 hydrogen bond to an aliphatic carbon-to-phoS (filed April 11, 1949 which has been abandoned, phorus bond. and was a continuation-in-part of application It is a further object of this invention to pro Serial No. 716,182 (filed December 12, 1946), which wide a more economical means of obtaining or has also been abandoned). O gano phosphorus compounds having a carbon-to Phosphonyl chloride and their derivatives are phosphorus linkage. useful in various arts. For example, certain It is a still further object of this invention to phosphonyl chloride derivatives (e.g., phosphonic provide a means of obtaining organo phosphorus acids and Salts and esters thereof) are useful compounds having -

Bureau of Industry and Security, Commerce Pt. 742, Supp. 1
Bureau of Industry and Security, Commerce Pt. 742, Supp. 1 that an export or reexport could make N,N-diethylethanolamine is December 12, a significant contribution to the mili- 1989. tary potential of North Korea, includ- (6) The contract sanctity date for exports ing its military logistics capability, or to all destinations (except Iran or Syria) of could enhance North Korea’s ability to phosphorus trichloride, trimethyl phosphite, support acts of international ter- and thionyl chloride is December 12, 1989. rorism, the Secretaries of State and For exports to Iran or Syria, paragraph (2) of Commerce will notify the Congress 30 this supplement applies. days prior to issuance of a license. (7) The contract sanctity date for exports to all destinations (except Iran or Syria) of [65 FR 38151, June 19, 2000, as amended at 66 2-chloroethanol and triethanolamine is Jan- FR 36682, July 12, 2001; 68 FR 16212, Apr. 3, uary 15, 1991. For exports of 2-chloroethanol 2003; 70 FR 54628, Sept. 16, 2005; 71 FR 20885, to Iran or Syria, paragraph (1) of this Sup- Apr. 24, 2006; 72 FR 3725, Jan. 26, 2007; 72 FR plement applies. For exports of triethanol- 62532, Nov. 5, 2007; 75 FR 14340, Mar. 25, 2010] amine to Iran or Syria, paragraph (5) of this Supplement applies. SUPPLEMENT NO. 1 TO PART 742—NON- (8) The contract sanctity date for exports PROLIFERATION OF CHEMICAL AND BI- to all destinations (except Iran or Syria) of OLOGICAL WEAPONS chemicals controlled by ECCN 1C350 is March 7, 1991, except for applications to export the NOTE: Exports and reexports of items in performance of contracts entered into before following chemicals: 2-chloroethanol, di- the applicable contract sanctity date(s) will methyl methylphosphonate, dimethyl be eligible for review on a case-by-case basis phosphite (dimethyl hydrogen phosphite), or other applicable licensing policies that phosphorus oxychloride, phosphorous tri- were in effect prior to the contract sanctity chloride, thiodiglycol, thionyl chloride tri- date. -

United States Patent Office Patented Aug
3,463,840 United States Patent Office Patented Aug. 26, 1969 1. 2 phonates in small yields. When at least three moles of mer 3,463,840 captan are employed, the alkylthioalkyldithiophospho PROCESS FOR PRODUCING ALKYLTHOALKYL nates are obtained in good yields. PHOSPHONODTHOC ESTERS Alkylthioalkyldithiophosphonates are known com Marion F. Botts, Independence, Mo., and Erik K. Regel, Mission, Kans, assignors to Chemagro Corporation, pounds and are useful as defoliants, Regal patent 3,193,- New York, N.Y., a corporation of New York 372. The procedure for preparing such compounds in the No Drawing. Original application June 24, 1966, Ser. No. Regal patent, namely reacting PCls with a hydrocarbon 560,097. Divided and this application Jan. 3, 1968, Ser. and a disulfide in the presence of a Friedel-Crafts catalyst, No. 708,745 however, is relatively expensive considering the overall Int, Cl, C07f 9/40, A01n 9/36 O yields and reaction conditions employed. U.S. C. 260-972 8 Claims Accordingly, it is an object of the present invention to develop a novel method for making alkylthioalkyldithio phosphonates. ABSTRACT OF THE DISCLOSURE Another object is to develop a novel method for Compounds are prepared having the formula 5 making analogues of alkylthioalkyldithiophosphonates wherein the alkyl thio group is replaced by haloalkylthio. O SR3 A further object is to develop a method of preparing RSCIP N dithiophosphonates which cannot be synthesized by other R SR4 routes. where R, Ra, and R4 are alkyl or monohaloalkyl and R2 20 An additional object is to develop a novel method for is hydrogen, alkyl, monohaloalkyl, aryl, haloaryl or al killing nematodes. -

The Reaction of Diphenyl Cadium with Phosphorus Trichloride
Union College Union | Digital Works Honors Theses Student Work 6-1940 The er action of diphenyl cadium with phosphorus trichloride Alfred Henry Case Union College - Schenectady, NY Follow this and additional works at: https://digitalworks.union.edu/theses Part of the Chemistry Commons Recommended Citation Case, Alfred Henry, "The er action of diphenyl cadium with phosphorus trichloride" (1940). Honors Theses. 1745. https://digitalworks.union.edu/theses/1745 This Open Access is brought to you for free and open access by the Student Work at Union | Digital Works. It has been accepted for inclusion in Honors Theses by an authorized administrator of Union | Digital Works. For more information, please contact [email protected]. The Reaction of Diphenyl Cadmium wj_th Phosphorus Trichloride J A thesis presented to the Department of Chem• istry of Union College, in partial fulfillment of the requirements for the Degree of Bachelor of Science in Chemistry, by ;,-. J ..,' ,., .... -. ' - " .. ' .. -..,, .. June, 1940 '"' ' - ' -1- Historical Any study of the stereochernistry of tricovalent atoms would necessarily involve members of the third and fifth periodic groups. Tertiary phosphines with heavy grol .pa would be most valuable for this work since phosphorus seems to be better suited to ezhi- bit asymmetry than any other element 01' the fifth group.<24) It is believed that the heavy groups would slow down the oscillations of the phosphorus atom and make possible the resolution of the optical isomers .. However, this work has been retarded by the lack of a convenient means of preparing asymmetrical sub- stituted triaryl phosphines. The usual method of preparing triaryl phosphines is by the reaction of the appropriate phosphenyl chloride with a Grignard reagent. -

List of Reactive Chemicals
LIST OF REACTIVE CHEMICALS Chemical Prefix Chemical Name Reactive Reactive Reactive CAS# Chemical Chemical Chemical Stimulus 1 Stimulus 2 Stimulus 3 111-90-0 "CARBITOL" SOLVENT D 111-15-9 "CELLOSOLVE" ACETATE D 110-80-5 "CELLOSOLVE" SOLVENT D 2- (2,4,6-TRINITROPHENYL)ETHYL ACETATE (1% IN ACETONE & BENZENE S 12427-38-2 AAMANGAN W 88-85-7 AATOX S 40487-42-1 AC 92553 S 105-57-7 ACETAL D 75-07-0 ACETALDEHYDE D 105-57-7 ACETALDEHYDE, DIETHYL ACETAL D 108-05-4 ACETIC ACID ETHENYL ESTER D 108-05-4 ACETIC ACID VINYL ESTER D 75-07-0 ACETIC ALDEHYDE D 101-25-7 ACETO DNPT T 126-84-1 ACETONE DIETHYL ACETAL D 108-05-4 ACETOXYETHYLENE D 108-05-4 1- ACETOXYETHYLENE D 37187-22-7 ACETYL ACETONE PEROXIDE, <=32% AS A PASTE T 37187-22-7 ACETYL ACETONE PEROXIDE, <=42% T 37187-22-7 ACETYL ACETONE PEROXIDE, >42% T S 644-31-5 ACETYL BENZOYL PEROXIDE (SOLID OR MORE THAN 45% IN SOLUTION) T S 644-31-5 ACETYL BENZOYL PEROXIDE, <=45% T 506-96-7 ACETYL BROMIDE W 75-36-5 ACETYL CHLORIDE W ACETYL CYCLOHEXANE SULFONYL PEROXIDE (>82% WITH <12% WATER) T S 3179-56-4 ACETYL CYCLOHEXANE SULFONYL PEROXIDE, <=32% T 3179-56-4 ACETYL CYCLOHEXANE SULFONYL PEROXIDE, <=82% T 674-82-8 ACETYL KETENE (POISON INHALATION HAZARD) D 110-22-5 ACETYL PEROXIDE, <=27% T 110-22-5 ACETYL PEROXIDE, SOLID, OR MORE THAN 27% IN SOLUTION T S 927-86-6 ACETYLCHOLINE PERCHLORATE O S 74-86-2 ACETYLENE D 74-86-2 ACETYLENE (LIQUID) D ACETYLENE SILVER NITRATE D 107-02-08 ACRALDEHYDE (POISON INHALATION HAZARD) D 79-10-7 ACROLEIC ACID D 107-02-08 ACROLEIN, INHIBITED (POISON INHALATION HAZARD) D 107-02-08 ACRYLALDEHYDE (POISON INHALATION HAZARD) D 79-10-7 ACRYLIC ACID D 141-32-2 ACRYLIC ACID BUTYL ESTER D 140-88-5 ACRYLIC ACID ETHYL ESTER D 96-33-3 ACRYLIC ACID METHYL ESTER D Stimulus - Stimuli is the thermal, physical or chemical input needed to induce a hazardous reaction. -

Acetylcholinesterase: the “Hub” for Neurodegenerative Diseases And
Review biomolecules Acetylcholinesterase: The “Hub” for NeurodegenerativeReview Diseases and Chemical Weapons Acetylcholinesterase: The “Hub” for Convention Neurodegenerative Diseases and Chemical WeaponsSamir F. de A. Cavalcante Convention 1,2,3,*, Alessandro B. C. Simas 2,*, Marcos C. Barcellos 1, Victor G. M. de Oliveira 1, Roberto B. Sousa 1, Paulo A. de M. Cabral 1 and Kamil Kuča 3,*and Tanos C. C. França 3,4,* Samir F. de A. Cavalcante 1,2,3,* , Alessandro B. C. Simas 2,*, Marcos C. Barcellos 1, Victor1 Institute G. M. ofde Chemical, Oliveira Biological,1, Roberto Radiological B. Sousa and1, Paulo Nuclear A. Defense de M. Cabral (IDQBRN),1, Kamil Brazilian Kuˇca Army3,* and TanosTechnological C. C. França Center3,4,* (CTEx), Avenida das Américas 28705, Rio de Janeiro 23020-470, Brazil; [email protected] (M.C.B.); [email protected] (V.G.M.d.O.); [email protected] 1 Institute of Chemical, Biological, Radiological and Nuclear Defense (IDQBRN), Brazilian Army (R.B.S.); [email protected] (P.A.d.M.C.) Technological Center (CTEx), Avenida das Américas 28705, Rio de Janeiro 23020-470, Brazil; 2 [email protected] Mors Institute of Research (M.C.B.); on Natural [email protected] Products (IPPN), Federal (V.G.M.d.O.); University of Rio de Janeiro (UFRJ), CCS,[email protected] Bloco H, Rio de Janeiro (R.B.S.); 21941-902, [email protected] Brazil (P.A.d.M.C.) 32 DepartmentWalter Mors of Institute Chemistry, of Research Faculty of on Science, Natural Un Productsiversity (IPPN),