Megagametophyte Development of Crysanthemum Leuoanthemum L. Var. Pinnatifidum Lecoq. and Lamotte
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Heterospory: the Most Iterative Key Innovation in the Evolutionary History of the Plant Kingdom
Biol. Rej\ (1994). 69, l>p. 345-417 345 Printeii in GrenI Britain HETEROSPORY: THE MOST ITERATIVE KEY INNOVATION IN THE EVOLUTIONARY HISTORY OF THE PLANT KINGDOM BY RICHARD M. BATEMAN' AND WILLIAM A. DiMlCHELE' ' Departments of Earth and Plant Sciences, Oxford University, Parks Road, Oxford OXi 3P/?, U.K. {Present addresses: Royal Botanic Garden Edinburiih, Inverleith Rojv, Edinburgh, EIIT, SLR ; Department of Geology, Royal Museum of Scotland, Chambers Street, Edinburgh EHi ijfF) '" Department of Paleohiology, National Museum of Natural History, Smithsonian Institution, Washington, DC^zo^bo, U.S.A. CONTENTS I. Introduction: the nature of hf^terospon' ......... 345 U. Generalized life history of a homosporous polysporangiophyle: the basis for evolutionary excursions into hetcrospory ............ 348 III, Detection of hcterospory in fossils. .......... 352 (1) The need to extrapolate from sporophyte to gametophyte ..... 352 (2) Spatial criteria and the physiological control of heterospory ..... 351; IV. Iterative evolution of heterospory ........... ^dj V. Inter-cladc comparison of levels of heterospory 374 (1) Zosterophyllopsida 374 (2) Lycopsida 374 (3) Sphenopsida . 377 (4) PtiTopsida 378 (5) f^rogymnospermopsida ............ 380 (6) Gymnospermopsida (including Angiospermales) . 384 (7) Summary: patterns of character acquisition ....... 386 VI. Physiological control of hetcrosporic phenomena ........ 390 VII. How the sporophyte progressively gained control over the gametophyte: a 'just-so' story 391 (1) Introduction: evolutionary antagonism between sporophyte and gametophyte 391 (2) Homosporous systems ............ 394 (3) Heterosporous systems ............ 39(1 (4) Total sporophytic control: seed habit 401 VIII. Summary .... ... 404 IX. .•Acknowledgements 407 X. References 407 I. I.NIRODUCTION: THE NATURE OF HETEROSPORY 'Heterospory' sensu lato has long been one of the most popular re\ie\v topics in organismal botany. -

Structure and Function of Spores in the Aquatic Heterosporous Fern Family Marsileaceae
Int. J. Plant Sci. 163(4):485–505. 2002. ᭧ 2002 by The University of Chicago. All rights reserved. 1058-5893/2002/16304-0001$15.00 STRUCTURE AND FUNCTION OF SPORES IN THE AQUATIC HETEROSPOROUS FERN FAMILY MARSILEACEAE Harald Schneider1 and Kathleen M. Pryer2 Department of Botany, Field Museum of Natural History, 1400 South Lake Shore Drive, Chicago, Illinois 60605-2496, U.S.A. Spores of the aquatic heterosporous fern family Marsileaceae differ markedly from spores of Salviniaceae, the only other family of heterosporous ferns and sister group to Marsileaceae, and from spores of all ho- mosporous ferns. The marsileaceous outer spore wall (perine) is modified above the aperture into a structure, the acrolamella, and the perine and acrolamella are further modified into a remarkable gelatinous layer that envelops the spore. Observations with light and scanning electron microscopy indicate that the three living marsileaceous fern genera (Marsilea, Pilularia, and Regnellidium) each have distinctive spores, particularly with regard to the perine and acrolamella. Several spore characters support a division of Marsilea into two groups. Spore character evolution is discussed in the context of developmental and possible functional aspects. The gelatinous perine layer acts as a flexible, floating organ that envelops the spores only for a short time and appears to be an adaptation of marsileaceous ferns to amphibious habitats. The gelatinous nature of the perine layer is likely the result of acidic polysaccharide components in the spore wall that have hydrogel (swelling and shrinking) properties. Megaspores floating at the water/air interface form a concave meniscus, at the center of which is the gelatinous acrolamella that encloses a “sperm lake.” This meniscus creates a vortex-like effect that serves as a trap for free-swimming sperm cells, propelling them into the sperm lake. -
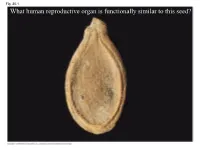
What Human Reproductive Organ Is Functionally Similar to This Seed?
Fig. 30-1 What human reproductive organ is functionally similar to this seed? Overview: Transforming the World • Seeds changed the course of plant evolution, enabling their bearers to become the dominant producers in most terrestrial ecosystems • A seed consists of an embryo and nutrients surrounded by a protective coat Copyright © 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings Fig. 30-1 What human reproductive organ is functionally similar to this seed? Seeds and pollen grains are key adaptations for life on land • In addition to seeds, the following are common to all seed plants – Reduced gametophytes – Heterospory – Ovules – Pollen Copyright © 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings Advantages of Reduced Gametophytes • The gametophytes of seed plants develop within the walls of spores that are retained within tissues of the parent sporophyte • Protect female gametophyte from UV and desiccation (good for the land life!) Copyright © 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings Fig. 30-2 PLANT GROUP Mosses and other Ferns and other seedless Seed plants (gymnosperms and angiosperms) nonvascular plants vascular plants Reduced, independent Reduced (usually microscopic), dependent on surrounding (photosynthetic and Gametophyte Dominant sporophyte tissue for nutrition free-living) Sporophyte Reduced, dependent on Dominant Dominant gametophyte for nutrition Gymnosperm Angiosperm Sporophyte Microscopic female (2n) gametophytes (n) inside ovulate cone Microscopic Sporophyte -

Stratigraphic Distribution of Species of the Megaspore Genus Minerisporites in North America
Stratigraphic Distribution of Species of the Megaspore Genus Minerisporites in North America GEOLOGICAL SURVEY PROFESSIONAL PAPER 743-E Stratigraphic Distribution of Species of the Megaspore Genus Minerisporites in North America By ROBERT H. TSCHUDY CONTRIBUTIONS TO PALEONTOLOGY GEOLOGICAL SURVEY PROFESSIONAL PAPER 743-E Taxonomy, stratigraphic ranges, and facies significance of Minerisporites megaspores UNITED STATES GOVERNMENT PRINTING OFFICE, WASHINGTON: 1976 UNITED STATES DEPARTMENT OF THE INTERIOR THOMAS S. KLEPPE, Secretary GEOLOGICAL SURVEY V. E. McKelvey, Director Tschudy, Robert H. Stratigraphic distribution of species of the megaspore genus Minerisporites in North America. (Contributions to Paleontology) (Geological Survey Professional Paper 743-E) Bibliography: p. Includes index. Supt. of Docs. no.: I 19.16:743-E 1. Minerisporites. 2. Paleobotany-Cretaceous. 3. Paleobotany-Paleocene. 4. Paleobotany-North America. I. Title. II. Series. III. Series: United States Geological Survey Professional Paper 743-E. QE996.T7 560' .13 76-608080 For sale by the Superintendent of Documents, U.S. Government Printing Office Washington, D.C. 20402 Stock Number 024-001-02892-1 CONTENTS Page Page Abstract ............................................................................................... EI Systematics-Continued New species of Minerisporites............................................................ I Genus Minerisporites-Continued Stratigraphic distribution................................................................... I Minerisporites -

Ovule Ontogenesis and Megasporogenesis in Adesmia Latifolia (Spreng.) Vog
Revista Brasil. Bot., V.26, n.4, p.495-502, out.-dez. 2003 Ovule ontogenesis and megasporogenesis in Adesmia latifolia (Spreng.) Vog. (Leguminosae-Papilionoideae)1 MARIA C.C. MOÇO2,3 and JORGE E.A. MARIATH2 (received: January 29, 2003; accepted: September 11, 2003) ABSTRACT – (Ovule ontogenesis and megasporogenesis in Adesmia latifolia (Spreng.) Vog. (Leguminosae-Papilionoideae)). The ovule ontogenesis and the megasporogenesis events were studied under bright field, fluorescence and scanning electron microscopy. The primordium is 3-zonate and gives rise to a hemianatropous, bitegmic and crassinucellate ovule. The archesporium may consist of one or more archesporial cells, but only one undergoes meiosis, forming a linear tetrad. Normally, only a single megaspore is functional in the chalazal position, but occasionally two functional chalazal megaspores arise. The present study provides additional information on embryological characters in the Adesmieae tribe and discusses their taxonomic significance to the Leguminosae family. Key words - Adesmia, embryology, Leguminosae, megaspore, ovule RESUMO – (Ontogênese do óvulo e megasporogênese em Adesmia latifolia (Spreng.) Vog. (Leguminosae - Papilionoideae)). A ontogênese do óvulo e os eventos da megasporogênese foram estudados sob microscopia de campo claro, fluorescência e eletrônica de varredura. O primórdio é trizonado e origina um óvulo hemianátropo, bitegumentado e crassinucelado. O arquespório pode ser composto de mais de uma célula arquesporial, mas apenas uma sofre meiose formando uma tétrade linear. Normalmente, apenas um megásporo é funcional na posição calazal, mas foram registrados alguns casos em que os dois megásporos calazais eram, simultâneamente, funcionais. O presente estudo acrescenta informações sobre caracteres embriológicos na tribo Adesmieae e discute sua importância taxonômica na família Leguminosae. -

Gametophyte Development in Near the Interior Base
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Magazine R718 Primer tissues. These form multicellular The first gametophytic division bud-like structures, each of which is asymmetric, producing one develops into a leafy shoot. The large and one small cell. In C. mature gametophytes produce richardii, the large cell divides Gametophyte male and female sexual organs, again asymmetrically to produce development the antheridia and archegonia, another small cell, which later respectively. The gametophyte is develops into the rhizoid, a root- often sexually distinct, and plants like structure. The small cell from Wuxing Li and Hong Ma are either male or female. the first division becomes the Each antheridium has an outer protonemal initial, which divides Unlike animals, which produce layer that encloses and protects further to form a linear three- single-celled gametes directly thousands of motile sperm, which celled protonema. The middle cell from meiotic products, plants swim through available external then undergoes a transition in have generations which alternate water layer to the egg. Fertilization divisional plane, forming a two- between the diploid sporophyte at the base of the cylindrical dimensional structure. During and haploid gametophyte (Figure archegonium produces a diploid hermaphrodite development, a 1A). The diploid sporophytic zygote which develops into an meristem is formed in the two- generation develops from the unbranched sporophyte. The dimensional plane and gives rise zygote, the fusion product of sporophyte consists of a thin stalk to the male antheridia and the haploid gametes. Sporophytic attached to the gametophyte, and female archegonia. -

Lecture 7 Integrative Biology 168: Spring 2009
Key concepts -- Lecture 7 Integrative Biology 168: Spring 2009 INTRODUCTION TO SEED PLANTS Any understanding of the origin and morphology of the seed requires us to revisit the concept of heterospory (also see p. 103 in Simpson's Plant Systematics for diagram). Heterospory = production of two different types of spores (in two different types of sporangia): (1) megaspores (developing into female gametophytes = megagametophytes) and (2) microspores (developing into male gametophytes = microgametophytes). Sperm and egg are produced by different types of gametophytes in heterosporous plants. Each of the two principal free-sporing lineages of vascular plants (lycophytes and ferns, that is) have both homosporous and heterosporous taxa, with heterospory being independently derived from homospory in both groups. In heterosporous lycophytes and heterosporous ferns (as in homosporous lycophytes and ferns), the spore is the unit of dispersal and is released from the sporangium of the parent sporophyte prior to spore germination. From now on, we will be studying heterosporous plants that produce only one functional megaspore and retain it within the megasporangium -- the seed plants. All of the seed plants are heterosporous; the transition to heterospory from homospory happened before all characteristics of the seed evolved. [See p. 102, 104, and 105 of Simpson's Plant Systematics for diagrams that illustrate the following points] General points about evolution of seed: 1) Maternal protection and care by the sporophyte for the megagametophyte was taken to a new level in the seed plants; not only is the only functional megaspore retained in the megasporangium (of the sporophyte) but also the megagametophyte that germinates from that megaspore develops inside the megasporangium. -

Club Mosses, Ferns & Horsetails: the Seed-Free Vascular Plants
Club Mosses, Ferns & Horsetails: the Seed-free Vascular Plants Vascular Plants - a quick review Two unrelated groups within “cryptogams” – seed free vascular plants – are recognized as phyla: 1. Lycopodiophyta : lycopods 2. Polypodiophyta: ferns, horsetails, and whisk ferns Vascular Plants - a quick review Why were the seed-free plants “grouped” together? They produce free spores, the principal dispersal units, via meiosis. Spore: a reproductive cell, capable of developing into an adult without fusion with another cell. spores Vascular Plants - a quick review Why were the seed-free plants “grouped” together? Spores develop within a sporangium (pl. sporangia) sporangium spores Vascular Plants - a quick review Why were the seed-free plants “grouped” together? Spores germinate and develop into gametophytes that exist independently of the spore-producing plants. The gametophytes (haploid, n) tend to be inconspicuous and short-lived. sporangium gametophyte spores Vascular Plants - a quick review Why were the seed-free plants “grouped” together? Like all plants, seed-free plants produce two kinds of gametes in their gametophytes: sperm and egg that unite to form a zygote (2n or diploid) via fertilization! sporangium zygote gametophyte spores Vascular Plants - a quick review Why were the seed-free plants “grouped” together? The sporophyte (2n) develops from the zygote and is more conspicuous, usually perennial and lives for an indefinite period! sporangium sporophyte zygote gametophyte spores Wisconsin Seed-free Plants The best website to identify -

Seed Plant Synapomorphies Introduction to Non-Flowering Seed Plants (Gymnosperms) a Seed Is a Highly Modified Megasporangium, So Seed Plants Are Heterosporous
LAB 06: Seed Plant Synapomorphies Introduction to non-flowering seed plants (Gymnosperms) A seed is a highly modified megasporangium, so seed plants are heterosporous. We will review important differences between heterosporous non-seed plants and seed plants. There are five lineages of extant seed plants, four of which are gymnosperms (seeds not enclosed in a fruit) and one lineage of angiosperms (seeds in a fruit). We will also review some potentially confusing differences in what the terms dioecious and monoecious refer to when applied to homosporous versus heterosporous plants. MICROSPORANGIA, MICROSPORES, MALE GAMETOPHYTES Microsporangia. There is no fundamental difference in the function of microsporangia in heterosporous non-seed and seed plants. The structure of the endosporic male gametophytes and the way they function, however, is drastically different in the two groups of plants. Microspores and Male Gametophytes: In heterosporous, non-seed plants an entire antheridium develops within the microspore wall (review the structure of the Selaginella male gametophyte). It has jacket cells surrounding a substantial number of spermatocytes. The microspore wall eventually cracks open and many sperm are released and swim to the female gametophyte (in dew, rainwater, pond water). Because seed plants are heterosporous, the gametophytes are endosporic just as they are in heterosporous non-seed plants. But the male gametophyte has undergone substantial reduction so that there is no longer any trace of an antheridium. Male gametophytes of seed plants consist of about 2-6 cells. The microspore, with its tiny internal gametophyte, is carried by wind or animals to somatic tissues in the vicinity of the female gametophyte. -

The Life Cycle of a Heterosporous Pteridophyte
Jan., 1905.] Heterosporous Pteridophyte. 255 THE LIFE CYCLE OF A HETEROSPOROUS PTERIDOPHYTE. JOHN H SCHAFFNER. The Heterosporous Pteridophytes represent the highest stage of development in the second or intermediate series of plants. The term heterosporous is applied to plants in which there are two kinds of nonsexual spores, large and small, called respectively megaspores and microspores. This peculiar spore condition is also present in the seed plants. The megaspore always gives rise to a female individual and the microspore to a male. In the lower Pteridophytes there is only one kind of nonsexual spores and they are, therefore, called homosporous. The Homosporous Pteridophytes should be kept distinct from the Heterosporous, since the development of heterospory represents one of the advancing waves of evolution which brought about profound changes in the character of the vegetation of the earth. The elimination of chance environment in the development of uni- sexual individuals by predetermining the sex in the spore and the reduction in the size of the gametophytes appear to have been necessary conditions in the evolution of the seed plants. The living species of Heterosporous Pteridophytes fall into six genera, the remnants of former extensive and dominant groups. The total number of species is about 635, somewhat more than the living Gymnosperms which constitute the next higher sub- kingdom. Because of their position as the lowest of heterospor- ous plants a thorough knowledge of their morphology and life history is necessary to a proper understanding of the structures and processes found in the Gymnosperms and Angiosperms. Nevertheless, much of their life history is still obscure and vari- ous statements rest on conjecture or imperfectly established facts. -

Effect of Low Temperature on Changes in AGP Distribution During Development of Bellis Perennis Ovules and Anthers
cells Article Effect of Low Temperature on Changes in AGP Distribution during Development of Bellis perennis Ovules and Anthers Agata Leszczuk 1,* , Ewa Szczuka 2, Kinga Lewtak 2, Barbara Chudzik 3 and Artur Zdunek 1 1 Institute of Agrophysics, Polish Academy of Sciences, 20-290 Lublin, Poland; [email protected] 2 Department of Cell Biology, Institute of Biological Sciences, Maria Curie-Skłodowska University, 20-033 Lublin, Poland; [email protected] (E.S.); [email protected] (K.L.) 3 Department of Biological and Environmental Education with Zoological Museum, Institute of Biological Sciences, Maria Curie-Skłodowska University, 20-033 Lublin, Poland; [email protected] * Correspondence: [email protected] Abstract: Arabinogalactan proteins (AGPs) are a class of heavily glycosylated proteins occurring as a structural element of the cell wall-plasma membrane continuum. The features of AGPs described earlier suggest that the proteins may be implicated in plant adaptation to stress conditions in important developmental phases during the plant reproduction process. In this paper, the microscopic and immunocytochemical studies conducted using specific antibodies (JIM13, JIM15, MAC207) recognizing the carbohydrate chains of AGPs showed significant changes in the AGP distribution in female and male reproductive structures during the first stages of Bellis perennis development. In typical conditions, AGPs are characterized by a specific persistent spatio-temporal pattern of distribution. AGP epitopes are visible in the cell walls of somatic cells and in the megasporocyte walls, Citation: Leszczuk, A.; Szczuka, E.; megaspores, and embryo sac at every stage of formation. During development in stress conditions, Lewtak, K.; Chudzik, B.; Zdunek, A. -

Pteridophyta – Vascular Cryptogams
Pteridophyta – Vascular cryptogams Dr. Pallavi J.N.L College Khagaul Division of Pteridophyta Pteridophytes includes four classes. • Psilopsida: Psilotum • Lycopsida: Lycopodium,Selaginella • Sphenopsida: Equisetum (Horse tail) • Pteropsida or Filicopsida: Pteridium, Adiantum GENERAL CHARACTERISTICS • Pteridophytes (pteron= feather, phyton= plant, i.e. plants with feather like leaves). • These are also known as vascular cryptogams. • They are represented by about 400 living and fossils genera and some 10,500 species. • These plants originated about 380 million years ago in the Silurian period of the Paleozoic era and formed dominant vegetation on the earth during the Devonian period. HABITAT • Most of the living pteridophytes are terrestrial, growing in moist and shady places. • Some members are aquatic (Azolla, Marsilea, Salvinia) usually occurring in permanent ponds. • A few forms are xerophytic like Equisetum. • the greatest diversity of pteridophytes is found in the tropics, with only about six hundred species adapted for life in temperate climates. Species living today are relics of ancient lineages that once dominated • Ferns reproduce by releasing spores rather than seeds. • There is a rich fossil record showing that pteridophytes have ancestors dating back nearly four hundred million years. Before there were seed plants, there were pteridophytes such as large, treelike (up to 36.5 meters [120 feet] tall) Lepidodendron, an ancestor of modern club mosses (which are no more than .30 meter [1 foot] tall), and shrubby Sphenophyllum PLANT BODY • They show much variation in their form, size and habit. They ranges from small annual (e.g. Azolla) to large tree-like perennials. • The main plant body is sporophytic, differentiated into root, stem and leaves.