Do Tip-Up Mounds Create Unique Microsites That Enhance Biodiversity?
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Plant Coefficient Listing.Xlsx
Wisconsin Native Plants Coefficient of Conservativsm Table August 2013 Coefficient of Latin Name Common Name Family Conservatism Acalypha rhomboidea Rhombic Cooper Leaf Euphorbiaceae 0 Achillea millefolium Yarrow Asteraceae 1 Aconitum columbianum Columbia Monk's Hood Ranunculaceae 10 Aconitum noveboracense Northern Blue Monkshood Ranunculaceae 10 Acorus americanus Sweet flag Acoraceae 7 Actaea pachypoda White baneberry Ranunculaceae 6 Actaea rubra Red baneberry Ranunculaceae 7 Adiantum pendatum Maidenhair Fern Pteridaceae 7 Adlumia fungosa Allegheny Vine Fumariaceae 7 Adoxa moschatellina Moschatel Adoxaceae 10 Agalinis aspera Rough false foxglove Scrophulariaceae 7 Agalinis gattingeri Round-stem False Foxglove Scrophulariaceae 8 Agalinis paupercula Small-flowered False Foxglove Scrophulariaceae 7 Agalinis purpurea Purple False Foxglove Scrophulariaceae 7 Agalinis skinneriana Pale False Foxglove Scrophulariaceae 9 Agalinis tenuifolia Common Foxglove Scrophulariaceae 7 Agastache foeniculum Blue Giant Hyssop Lamiaceae 5 Agastache nepetoides Yellow giant hyssop Lamiaceae 5 Agastache scrophulariaefolia Purple giant hyssop Lamiaceae 4 Agrimonia gryposepala Tall agrimony Rosaceae 2 Agrimonia parviflora Southern Agrimony Rosaceae 4 Agrimonia pubescens Downy Agrimony Rosaceae 5 Agrimonia striata Grooved Agrimony Rosaceae 3 Agrostis hyemalis Ticklegrass Poaceae 4 Aletris farinosa Colic root Liliaceae 9 Alisma gramineum Grass-leaved Water Plantain Alismataceae 5 Alisma subcordatum Common Water Plantain Alismataceae 3 Alisma triviale Northern Water -

Hardwood Swamps of Vermont: Distribution, Ecology, Classification, and Some Sites of Ecological Significance
Hardwood Swamps of Vermont: Distribution, Ecology, Classification, and Some Sites of Ecological Significance Red Maple-Black Ash Seepage Swamp, illustration by Libby Davidson Eric Sorenson, Robert Popp, Michael Lew-Smith, Brett Engstrom, Marc Lapin, and Mark Ferguson for Nongame and Natural Heritage Program Vermont Fish and Wildlife Department Agency of Natural Resources Waterbury, Vermont March, 2004 Acknowledgements This study of Vermont's hardwood swamps was funded by the United States Environmental Protection Agency through the Performance Partnership with Vermont Department of Environmental Conservation. Julia Watson identified and contacted the numerous landowners with hardwood swamps on their properties. Ian Worley piloted the plane for aerial reconnaissance, as well as sharing his expertise on Vermont's landscape ecology during the flights. Everett Marshall and Jodi Shippee helped with many aspects of the project, including botanical and ecological issues, data management, and GIS mapping. Everett Marshall, Alan Quackenbush, Ted Allen, and Doug Blodgett all assisted in conducting breeding bird surveys. Kristen Rose conducted the majority of the field work for amphibian surveys. Leif Richardson provided information on hardwood swamps recently identified on state lands. Mark Rahill carefully identified many mosses and liverworts collected during field work. District Wildlife Biologists Kim Royar, Doug Blodgett, and John Gobeille shared their expertise during site visits or on wildlife use of hardwood swamps. Libby Davidson created the pen and ink illustrations of hardwood swamps. Laura Cadmus and Eric Hillmuth both provided assistance with GIS. Jenny Ramstetter and her Marlboro College ecology class helped sample a few southern Vermont swamps. Anne Turner entered most of the plot data. -

The Delaware Wetland Plant Field Guide
Compiled by DNREC’s Wetland Monitoring & Assessment Program 1 This Field Guide was prepared by the Delaware Department of Natural Resources and Environmental Control's (DNREC) Wetland Monitoring & Assessment Program (WMAP). WMAP provides state leadership to conserve wetlands for their water quality, wildlife habitat, and flood control benefits. This project has been funded wholly or in part by the United States Environmental Protection Agency under assistance agreement CD-96347201 CFDA 66.461 to Delaware Department of Natural Resources and Environmental Control. The contents of this document do not necessarily reflect the views and policies of the Environmental Protection Agency, nor does the EPA endorse trade names or recommend the use of commercial products mentioned in this document. Acknowledgements: Special thanks to Bill McAvoy, LeeAnn Haaf, Kari St. Laurent, Susan Guiteras, and Andy Howard for reviewing the guide and providing helpful feedback. Photo credits are listed below pictures. All photos that do not have credits listed were taken or drawn by WMAP. Cover illustrations courtesy of the University of Wisconsin Extension and the Wisconsin Department of Natural Resources. Recommended Citation: Delaware Department of Natural Resources and Environmental Control. 2018. The Delaware Wetland Plant Field Guide. Dover, Delaware, USA. 146pp. 2 to this illustrated guide of the most common wetland plants found in Delaware. All wetlands have 3 characteristics: 1. Water at or near the surface for some part of the year 2. Hydrophytic plants, which are specially adapted to living in wet conditions 3. Hydric soils, which are soils that are permanently or seasonally soaked in water, resulting in oxygen deprivation If you have water on the area of interest for at least some part of the year, the next step in determining if you’re in a wetland is to take a look at the plants. -

A Mi'kmaq Traditional and Ecological Knowledge Review of Three Wind
A Mi’kmaq Traditional and Ecological Knowledge Review of three Wind Project Development Properties Historical and Field Survey Information Submitted to: Affinity Renewables Inc 1383 Mt Thom Rd Saltspring, NS. Canada B0K 1P0 Submitted by: AMEC Environment & Infrastructure a division of AMEC Americas Ltd. Dartmouth, Nova Scotia December 2013 TV134003 MEKS DRAFT Affinity Renewables Inc. December 2013 TABLE OF CONTENTS 1.0 INTRODUCTION .................................................................................................. 3 1.1 INDIGENOUS KNOWLEDGE AND KNOWLEDGE SYSTEMS ........................................................................... 3 2.0 DATA ACQUISITION ........................................................................................... 7 2.1 GATHERING OF LOCAL KNOWLEDGE OF PROJECT SITE ............................................................................. 7 2.1.1 Data Collection and Verification‐ WORKSHOP ..................................................................... 7 2.2 FIELD SURVEY FOR PLANT SPECIES OF MI’KMAQ CULTURAL SIGNIFICANCE .................................................. 7 2.2.1 Review of Available Data ...................................................................................................... 7 2.2.2 Field Survey ........................................................................................................................... 7 2.3 WILDLIFE SURVEY AND HABITAT MODELING EXERCISE ............................................................................ 8 3.0 -

Invasive Plant Species Management at Gwynedd Wildlife Preserve, Ambler, PA
University of Pennsylvania ScholarlyCommons Master of Environmental Studies Capstone Department of Earth and Environmental Projects Science 2014 Invasive Plant Species Management at Gwynedd Wildlife Preserve, Ambler, PA Nathan Hartshorne University of Pennsylvania Follow this and additional works at: https://repository.upenn.edu/mes_capstones Part of the Environmental Sciences Commons Hartshorne, Nathan, "Invasive Plant Species Management at Gwynedd Wildlife Preserve, Ambler, PA" (2014). Master of Environmental Studies Capstone Projects. 57. https://repository.upenn.edu/mes_capstones/57 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/mes_capstones/57 For more information, please contact [email protected]. Invasive Plant Species Management at Gwynedd Wildlife Preserve, Ambler, PA Disciplines Environmental Sciences | Physical Sciences and Mathematics This presentation is available at ScholarlyCommons: https://repository.upenn.edu/mes_capstones/57 INVASIVE PLANT SPECIES MANAGEMENT AT GWYNEDD WILDLIFE PRESERVE, AMBLER, PA Nathan Hartshorne Spring 2014 Daniel Barringer, Natural Lands Trust Sarah A. Willig, PhD, University of Pennsylvania Hartshorne 2 Hartshorne 3 Acknowledgements: When choosing a project for school, it is important to find a topic that forces you to really learn. If you are already something of an expert, you may create a better work, but you will get less out of it. There is a balance, however, in trying not to choose something too far over your own head. Trial by fire leads to many a burned person. Fortunately, to keep from getting overly scorched, there were several people without whom I never would have managed to complete this project. First and foremost is my academic advisor, Dr. Sarah Willig who met with me at Gwynedd Wildlife Preserve on multiple occasions and on campus at the University of Pennsylvania on several other occasions. -

Ecological Assessment and Rare Species Surveys of the Coleman Parcel, Barry State Game Area, Barry Co., Michigan
Ecological Assessment and Rare Species Surveys of the Coleman Parcel, Barry State Game Area, Barry Co., Michigan Prepared By: Bradford S. Slaughter and Yu Man Lee Michigan Natural Features Inventory Michigan State University Extension P.O. Box 30444 Lansing, MI 48909-7944 Prepared For: Michigan Department of Natural Resources Wildlife Division September 2, 2016 MNFI Report Number 2016–17 Michigan Natural Features Inventory Discover. Define. Deliver. Funding for this project was provided by the Michigan Department of Natural Resources, Wildlife Division. Suggested Citation: Slaughter, B.S., and Y.Lee. 2016. Ecological Assessment and Rare Species Surveys of the Coleman Parcel, Barry State Game Area, Barry Co., Michigan. Michigan Natural Features Inventory, Report No. 2016-17, Lansing, MI. 37 pp. Copyright 2016 Michigan State University Board of Trustees. Michigan State University Extension programs and materials are open to all without regard to race, color, national origin, gender, religion, age, disability, political beliefs, sexual orientations, marital status, or family status. Cover photograph: Seeps along Hill Creek, Barry State Game Area, Barry Co., Mich., July 27, 2016. Photograph by B.S. Slaughter. Executive Summary In 2015—2016, Michigan Natural Features Inventory (MNFI) contracted with Michigan Department of Natural Resources (MDNR), Wildlife Division (WLD), to conduct ecological assessments at two state game/wildlife areas, including a recently acquired 15 hectare (37 acre) parcel in Barry State Game Area (SGA) in Barry County, Michigan, previously owned by Dave Coleman. MNFI conducted surveys in June— August 2016. Surveys resulted in the extension of existing element occurrences for prairie fen and Myrica pensylvanica (bayberry) and the redetection of eastern massasauga (Sistrurus catenatus) at the adjacent Hill Creek fen. -
Vascular Plant Richness in an Electric Power Right-Of-Way Wetland at Greenwoods Conservancy, Summer 1999
329 Vascular plant richness in an electric power right-of-way wetland at Greenwoods Conservancy, summer 1999 Sarah Groff INTRODUCTION Greenwoods Conservancy is a thousand-acre nature preserve located in Burlington, New York. A conservation easement through the Otsego Land Trust, a non-profit corporation, protects Greenwoods from development in perpetuity (Taylor, 1994). In 1986, the New York State Power Authority built the Marcy-South power line through Greenwoods. In building the power line, a corridor, or Right-of-Way (ROW), was created. Since the creation of Marcy-South, the ROW has been maintained through selective cutting and chemical stump treatment. The objective of this study was to determine the effect of the ROW management on a wetland that runs through the ROW. A similar study was conducted in 1997 at Belford, Caledonia, Croghan, Eden, Empeyfield and Fulton by the Empire State Electric Energy Research Corporation (ESEERCO). This work concluded that "maintaining ROW within wetlands does not adversely affect one indicator of biodiversity, i.e., vascular species richness, nor the ability to provide habitat for plant species of state or regional concern" (Podniesinski et aI., 1997). This study followed the basic methodology of the ESEERCO's survey to determine whether the ESEERCO's conclusion is applicable to the Greenwoods ROW wetland. METHODS Two 20 x 50 meter plots were laid out, one within the ROWand the other adjacent to the ROW plot (Figure 1). The deciduous wood surrounding the off-ROW plot limited the size ofthe plot. To minimize the influence of edge effects, the first 10 meters on either side of the forest edge were excluded from the study (MacLellan and Stewart, 1986). -
DEWBERRIES and BRAMBLES Rubus Spp
DEWBERRIES AND BRAMBLES Rubus spp. University of Massachusetts, Cranberry Experiment Station Hilary A. Sandler Two different species of Rubus commonly occur on Taxonomy commercial cranberry bogs: bristly dewberry (Rubus The taxonomy of Rubus is difficult to sort out as many hispidus) and prickly dewberry (Rubus flagellaris). Upright species hybridize and exhibit polyploidy (more than twice bramble (Rubus allegheniensis) was more prevalent in the the usual number of chromosomes). The genus is in the 1970’s-1980’s, but is typically controlled in the field by a family, Rosaceae (the rose family), and 25 species are rust pathogen (see photo next page). Currently, it is rarely described in an authoritative flora (Gleason and Cronquist, seen on bogs in Massachusetts. Thus, further mention of 1991). The genus as a whole is known by the common dewberries or brambles in this fact sheet refers only to R. name, brambles, but the species that occur in MA are hispidus and R. flagellaris. The range of these weed pests identified by their common names, swamp dewberry (R. extends through a wide range of habitats, from Quebec hispidus) and Northern dewberry (R. flagellaris). In and Nova Scotia west to Wisconsin and Minnesota and Massachusetts, growers sometimes interchange the group south to North Carolina and Georgia. Rubus species are names, brambles and briers, but these are two distinct characterized by growth in disturbed habitats, often genera of plants. In fact, they fall into two separate major representing an early stage of plant succession (in non- categories of plant types. Briers are monocotyledons (one agricultural settings). On cranberry plantings, infestations true leaf emerges from the seed) classified to the genus, tend to occur in small to large patches on the high edges. -

A Checklist of New York State Plants
THt U: '"'■I Digitized by the Internet Archive in 2017 with funding from IMLS LG-70-15-0138-15 https://archive.org/details/checklistofnewyo4581 mite j.' fORK 0QTAR1CAL GARDEN A Checklist of New York State Plants Richard S. Mitchell State Botanist New York State Museum Contributions to a Flora of New York State Checklist III Richard S. Mitchell, Editor 1986 Bulletin No. 458 New York State Museum The State Education Department does not discriminate on the basis of age, color, reli¬ gion, creed, disability, marital status, veteran status, national origin, race, or sex in the educational programs and activities which it operates. Inquiries concerning this policy of equal opportunity and affirmative action should be referred to the Department’s Affirma¬ tive Action Officer, Education Building, Albany, NY 12234. Phone (518) 473-1265. The University of the State of New York THE STATE EDUCATION DEPARTMENT Albany, New York 12230 THE UNIVERSITY OF THE STATE OF NEW YORK Regents of The University (with years when terms expire) 1987 MARTIN C. Barell, Chancellor, B.A., I.A., LL.B., LL.D., Muttontown 1987 R. Carlos Carballada, Vice Chancellor, B.S., L.H.D., D.Se. Rochester 1988 Willard A. Genrich, LL.B., L.H.D., LL.D., Litt.D., D.C.S., D.C.L., Sc.I). Buffalo 1989 Emlyn I. Griffith, A.B„ J.D., L.H.D., Sc.D.Rome 1991 Jorge L. Batista, B.A., J.D., LL.D. Bronx 1993 Laura Bradley Chodos, B.A., M.A.. Viseher Ferry 1991 Louise P. Matteoni, B.A., M.A., Ph.D. -

Fragaria Virginiana - Wild Strawberry
Diversity of Wisconsin Rosids . roses, currants, raspberries . we will be seeing, in the next few lectures, many of the woody plants (trees/shrubs) present at your sites Trees & Shrubs ID Helps Trees & Shrubs ID Helps John Kress – Smithsonian Institution Trees & Shrubs ID Helps Trees & Shrubs ID Helps Gary Fewless – UW Green Bay Herbarium Rosids Rosids are one of the two large groups of dicots; the other group are the Asterids Rosids: Separate petals Asterids: Fused petals Hamamelidaceae - witch hazel family Trees and shrubs in subtropical and temperate areas but only 1 species in Wisconsin - witch hazel found in rich deciduous woods. A very distinctive shrub with asymmetrical crenate leaves and late-blooming flowers. Hamamelis virginiana Witch hazel Hamamelidaceae - witch hazel family Simple, alternate leaves on flattened branches; these leaves usually with palmate venation or at least pronounced basal veins Hamamelis virginiana Witch hazel Hamamelidaceae - witch hazel family Witch hazel yields an astringent and soothing lotion for cuts and bruises, hemorrhoids 18th century 20th century Water diviners favor witch hazel for their dowsers (the divining rod to find water underground) Hamamelidaceae - witch hazel family __ The flowers 4 merous (the family can CA 4-5 CO 4-5 A 4-5 G (2) be five merous), and insect pollinated in the fall (images from Sept) Petals are ribbon-like 4 stamens opposite the sepals, but 4 small staminodia are opposite the petals Hamamelis virginiana Witch hazel Hamamelidaceae - witch hazel family __ CA 4-5 CO 4-5 -

Diversity and Evolution of Rosids
Diversity and Evolution of Rosids . roses, currants, peonies . Eudicots • continue survey through the eudicots or tricolpates • vast majority of eudicots are Rosids (polypetalous) and Asterids core (sympetalous) eudicots rosid asterid Eudicots • unlike Asterids, Rosids (in orange) now represent a diverse set of families *Saxifragales • before examining the large Rosid group, look at a small but important order of flowering plants - Saxifragales Paeonia Sedum *Saxifragales • small group of 16 families and about 2500 species sister to Rosids • ancient lineage from 120 mya and underwent rapid radiation Paeonia Sedum *Saxifragales • part of this ancient radiation may involve this small family of holo-parasites - Cynomoriaceae *Saxifragales • they generally can be identified by their two or more separate or semi-fused carpels, but otherwise quite variable Paeonia Sedum Paeoniaceae 1 genus / 33 species • like many of these families, Paeonia exhibits an Arcto-Tertiary distribution Paeoniaceae 1 genus / 33 species • small shrubs with primitive features of perianth and stamens • hypogynous with 5-8 separate carpels developing into follicles Cercidiphyllaceae 1 genus / 2 species • small trees (kadsura-tree) restricted to eastern China and Japan . • . but fossils in North America and Europe from Tertiary Cercidiphyllaceae 1 genus / 2 species • unisexual, wind-pollinated but do produce follicles Hamamelidaceae 27 genera and 80 species - witch hazels • family of trees and shrubs in subtropical and temperate areas but only 1 species in Wisconsin - witch -
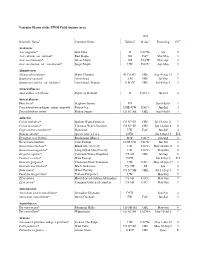
Cedarburg Bog Plants List
Vascular Plants of the UWM Field Station Area Wet Scientific Name1 Common Name Habitat2 Status3 Flowering CC4 Aceraceae Acer negundo* Box Elder D FACW- Apr 0 Acer rubrum var. rubrum* Red Maple SH FAC Mar-May 3 Acer saccharinum* Silver Maple SH FACW Mar-Apr 2 Acer saccharum var. saccharum* Sugar Maple UW FACU Apr-May 5 Alismataceae Alisma subcordatum Water-Plantain M CS SH OBL Aug 4-Aug 21 3 Sagittaria cuneata* Arrowhead A M OBL Jul-Sep 7 Sagittaria latifolia var. latifolia* Arrowhead, Wapato A M SC OBL Jul 4-Sep 5 3 Amaranthaceae Amaranthus retroflexus Pigweed, Redroot D FACU+ Jul-Oct 0 Anacardiaceae Rhus hirta* Staghorn Sumac FD Jun 8-Jul 8 2 Toxicodendron radicans subsp. negundo Poison-Ivy D FD UW FAC+ Jun-Jul 4 Toxicodendron vernix Poison Sumac CS SC SH OBL Jun 8-Jul 8 7 Apiaceae Cicuta bulbifera* Bulblet Water-Hemlock CS SC SH OBL Jul 19-Sep 11 7 Cicuta maculata* Common Water-Hemlock CS SC SH OBL Jun 16-Sep 8 6 Cryptotaenia canadensis* Honewort UW FAC Jun-Jul 4 Daucus carota* Queen Anne's-Lace D FD Jul 4-Sep 24 E:I Eryngium yuccifolium Rattlesnake-Master FD P FAC+ Jul-Aug 8 Heracleum lanatum Cow-Parsnip D SH UW FACW Jun-Jul 3 Osmorhiza claytonii* Bland Sweet Cicely UW FACU- May 24-Jun 11 5 Osmorhiza longistylis* Long-Styled Sweet Cicely UW FACU- May-Jun 4 Oxypolis rigidior* Common Water-Dropwort CS SH OBL Jul-Sep 6 Pastinaca sativa* Wild Parsnip D FD Jun 8-Sep 2 E:I Sanicula gregaria* Clustered Black Snakeroot UW FAC+ May 26-Jun 29 3 Sanicula marilandica* Black Snakeroot CS UW NI Jun 5 Sium suave* Water-Parsnip CS SC SH OBL Jul 14-Sep 5 5 Taenidia integerrima* Yellow-Pimpernel UW May-Jun 7 Zizia aptera Heart-Leaved Golden Alexanders CS SH FACU May-Jun 9 Zizia aurea* Common Golden Alexanders CS SH FAC+ May-Jun 7 Apocynaceae Apocynum androsaemifolium* Spreading Dogbane D FD Jun-Sep 2 Apocynum cannabinum* Hemp-Dogbane FW FAC Jun-Jul 3 Aquifoliaceae Ilex mucronata* Mountain Holly CS OB SH OBL May-Jun 8 Ilex verticillata* Winterberry CS SC SH FACW+ May 10-Jul 5 7 Araceae Arisaema triphyllum subsp.