Orange Book Cumulative Supplement 07 July 2019
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
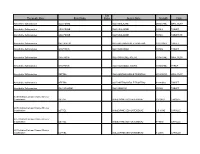
Therapeutic Class Brand Name P a Status Generic
P A Therapeutic Class Brand Name Status Generic Name Strength Form Absorbable Sulfonamides AZULFIDINE SULFASALAZINE 250MG/5ML ORAL SUSP Absorbable Sulfonamides AZULFIDINE SULFASALAZINE 500MG TABLET Absorbable Sulfonamides AZULFIDINE SULFASALAZINE 500MG TABLET DR Absorbable Sulfonamides BACTRIM DS SULFAMETHOXAZOLE/TRIMETHO 800-160MG TABLET Absorbable Sulfonamides GANTRISIN SULFISOXAZOLE 500MG TABLET Absorbable Sulfonamides GANTRISIN SULFISOXAZOLE ACETYL 500MG/5ML ORAL SUSP Absorbable Sulfonamides GANTRISIN SULFISOXAZOLE ACETYL 500MG/5ML SYRUP Absorbable Sulfonamides SEPTRA SULFAMETHOXAZOLE/TRIMETHO 200-40MG/5 ORAL SUSP Absorbable Sulfonamides SEPTRA SULFAMETHOXAZOLE/TRIMETHO 400-80MG TABLET Absorbable Sulfonamides SULFADIAZINE SULFADIAZINE 500MG TABLET ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 10-20MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 2.5-10MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 5-10MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 5-20MG CAPSULE P A Therapeutic Class Brand Name Status Generic Name Strength Form ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 5-40MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 10-40MG CAPSULE Acne Agents, Systemic ACCUTANE ISOTRETINOIN 10MG CAPSULE Acne Agents, Systemic ACCUTANE ISOTRETINOIN 20MG CAPSULE Acne Agents, Systemic ACCUTANE -

Design, Synthesis, and Evaluation of Antiepileptic Compounds Based on Β-Alanine and Isatin
Design, Synthesis, and Evaluation of Antiepileptic Compounds Based on β-Alanine and Isatin by Robert Philip Colaguori A thesis submitted in conformity with the requirements for the degree of Master of Science Department of Pharmaceutical Sciences University of Toronto © Copyright by Robert Philip Colaguori, 2016 ii Design, Synthesis, and Evaluation of Antiepileptic Compounds Based on β-Alanine and Isatin Robert Philip Colaguori Master of Science Department of Pharmaceutical Sciences University of Toronto 2016 Abstract Epilepsy is the fourth-most common neurological disorder in the world. Approximately 70% of cases can be controlled with therapeutics, however 30% remain pharmacoresistant. There is no cure for the disorder, and patients affected are subsequently medicated for life. Thus, there is a need to develop compounds that can treat not only the symptoms, but also delay/prevent progression. Previous work resulted in the discovery of NC-2505, a substituted β-alanine with activity against chemically induced seizures. Several N- and α-substituted derivatives of this compound were synthesized and evaluated in the kindling model and 4-AP model of epilepsy. In the kindling model, RC1-080 and RC1-102 were able to decrease the mean seizure score from 5 to 3 in aged mice. RC1-085 decreased the interevent interval by a factor of 2 in the 4-AP model. Future studies are focused on the synthesis of further compounds to gain insight on structure necessary for activity. iii Acknowledgments First and foremost, I would like to thank my supervisor Dr. Donald Weaver for allowing me to join the lab as a graduate student and perform the work ultimately resulting in this thesis. -

High-Throughput Brain Activity Mapping and Machine Learning As a Foundation for Systems Neuropharmacology
High-throughput brain activity mapping and machine learning as a foundation for systems neuropharmacology Lin, Xudong; Duan, Xin; Jacobs, Claire; Ullmann, Jeremy; Chan, Chung-Yuen; Chen, Siya; Cheng, Shuk-Han; Zhao, Wen-Ning; Poduri, Annapurna; Wang, Xin; Haggarty, Stephen J.; Shi, Peng Published in: Nature Communications Published: 03/12/2018 Document Version: Final Published version, also known as Publisher’s PDF, Publisher’s Final version or Version of Record License: CC BY Publication record in CityU Scholars: Go to record Published version (DOI): 10.1038/s41467-018-07289-5 Publication details: Lin, X., Duan, X., Jacobs, C., Ullmann, J., Chan, C-Y., Chen, S., Cheng, S-H., Zhao, W-N., Poduri, A., Wang, X., Haggarty, S. J., & Shi, P. (2018). High-throughput brain activity mapping and machine learning as a foundation for systems neuropharmacology. Nature Communications, 9, [5142]. https://doi.org/10.1038/s41467-018-07289- 5 Citing this paper Please note that where the full-text provided on CityU Scholars is the Post-print version (also known as Accepted Author Manuscript, Peer-reviewed or Author Final version), it may differ from the Final Published version. When citing, ensure that you check and use the publisher's definitive version for pagination and other details. General rights Copyright for the publications made accessible via the CityU Scholars portal is retained by the author(s) and/or other copyright owners and it is a condition of accessing these publications that users recognise and abide by the legal requirements associated with these rights. Users may not further distribute the material or use it for any profit-making activity or commercial gain. -

"2-Oxo-1-Pyrrolidine Derivative and Its Pharmaceutical Uses"
(19) & (11) EP 1 452 524 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: C07D 207/27 (2006.01) A61K 31/4015 (2006.01) 14.10.2009 Bulletin 2009/42 A61P 25/08 (2006.01) (21) Application number: 04007878.4 (22) Date of filing: 21.02.2001 (54) "2-oxo-1-pyrrolidine derivative and its pharmaceutical uses" "2-Oxo-1-Pyrrolidinderivate und ihre pharmazeutische Verwendung" "Dérivé de 2-oxo-1-pyrrolidine et ses applications pharmaceutique" (84) Designated Contracting States: • Talaga, Patrice AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU 1170 Watermael-Boitsfort (BE) MC NL PT SE TR (74) Representative: UCB (30) Priority: 23.02.2000 GB 0004297 Intellectual Property c/o UCB S.A. (43) Date of publication of application: Intellectual Property Department 01.09.2004 Bulletin 2004/36 Allée de la Recherche 60 1070 Brussels (BE) (62) Document number(s) of the earlier application(s) in accordance with Art. 76 EPC: (56) References cited: 01925354.1 / 1 265 862 WO-A-99/13911 GB-A- 1 309 692 (73) Proprietor: UCB Pharma, S.A. • PROUS: "LEVETIRACETAM" DRUGS OF THE 1170 Brussels (BE) FUTURE, BARCELONA, ES, vol. 19, no. 2, 1994, pages 111-113, XP000908882 ISSN: 0377-8282 (72) Inventors: • FISCHER W ET AL: "Die Wirksamkeit von • Differding, Edmond Piracetam, Meclofenoxat und Vinpocetin in 1348 Ottignies-Louvain-La-Neuve (BE) verschiedenen Krampfmodellen bei der Maus" • Kenda, Benoît PHARMAZIE, VEB VERLAG VOLK UND 5080 Emines (BE) GESUNDHEIT. BERLIN, DD, vol. 46, no. -

DILANTIN Oral Suspension Page 1 of 23 LPDPYC102019 Pfleet Number: 2019-0051655, 2019-0053053 4.2 Posology and Method of Administration
For the use only of a Registered Medical Practitioners or a Hospital or a Laboratory Phenytoin Oral Suspension USP DILANTIN Suspension 1. NAME OF MEDICINAL PRODUCT DILANTIN SUSPENSION 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Phenytoin is an anticonvulsant drug, related to the barbiturates in chemical structure, but has a five-membered ring. The chemical name is 5,5-diphenyl-2,4-imidazolidinedione. Each 5 mL of oral suspension contains 125 mg of phenytoin I.P. All strengths mentioned in this document might not be available in the market. For full list of excipients please see section 6.1. 3. PHARMACEUTICAL FORM Oral suspension 4. CLINICAL PARTICULARS 4.1 Therapeutic Indications Phenytoin is indicated for the control of generalized tonic-clonic (grand mal) and complex partial (psychomotor, temporal lobe) seizures and prevention and treatment of seizures occurring during or following neurosurgery. Trademark of Parke, Davis & Company, LLC, USA. Licensed user: Pfizer Limited, India DILANTIN Oral Suspension Page 1 of 23 LPDPYC102019 PfLEET Number: 2019-0051655, 2019-0053053 4.2 Posology and Method of Administration General NOT FOR PARENTERAL USE Phenytoin capsules and solution for injection are formulated with the sodium salt of phenytoin. The free acid form of phenytoin is used in the phenytoin suspensions (30 mg/5 mL (pediatric) and 125 mg/5 mL). Because there is approximately an 8% increase in drug content with the free acid form over that of the sodium salt, dosage adjustments and serum level monitoring may be necessary when switching from a product formulated with the free acid to a product formulated with the sodium salt and vice versa. -

Reproductive Performance and Embriotoxicity of Rats Exposed to Carbamazepine
Revista Brasileira de Ciências Farmacêuticas Brazilian Journal of Pharmaceutical Sciences vol. 44, n. 3, jul./set., 2008 Reproductive performance and embriotoxicity of rats exposed to carbamazepine Marli Gerenutti*, Carolina Clavijos de Oliveira, Aliana Conceição Ribeiro de Miranda, Raquel Mendonça Rosa, Fernando de Sá Del Fiol Curso de Farmácia, Universidade de Sorocaba Uniterms To study the possible effects of carbamazepine in rats during • Carbamazepine pregnancy and fetuses´ physical development, carbamazepine • Teratology solubilized in propilenglycol was orally administered (20 and • Embriotoxicity 40 mg/kg) to the female rats from the 2nd to the 19th days of • Pregnancy pregnancy. Propilenglycol was administered to the control group. • Reproduction The animals were sacrificed on the 20th day, when 50% of the offspring were fixed in Bouin´s solution and the remaining 50% were submitted to diaphanization. The carbamazepine administration caused reduction on weight gain of pregnant rats and did not damage the females´ reproductive performance. In the fetuses´ physical development, it was observed a flattening on the skull soft tissues and bones; delay in the skull bone development; cartilage calcification increase between hip and *Correspondência: femur and reduction in the number of the sternum ossifications. M.Gerenutti, Universidade de Sorocaba Although carbamazepine has not caused general changes over Av. Dr. Eugênio Salerno, 140 female rats´ reproductive performance, it produced significant 18035-430 - Sorocaba - SP, Brasil E-mail [email protected]. alterations in the development of the fetuses´ skeletal parameters. INTRODUCTION to anatomical and/or biochemical abnormalities specially when specifying a group of epileptic pregnant females Drugs usage during pregnancy and breast feeding (Kaneko et al., 2001; Samren et al., 2003). -

Antiepileptic Drug Treatment in 1959
Antiepileptic Drug Treatment in 1959 The two decades between 1938 and 1958 were notable for a large number of new medicinal com- pounds.Phenytoin was of course the most important, but other drugs, some of which are still prescribed, were introduced at this time. Not only drugs – for the ketogenic diet was also popu- larised in this period. Lennox in his book in 1960 gave a long treatise on drug therapy. Head- ing his list were the bromides, which he found rather ineffective, but barbiturates were greeted enthusiastically. Lennox mentioned that 2,500 compounds had been synthesised, and of these 50 compounds were marketed of which phenobarbital was the most frequently used for epilepsy. He was also enthusiastic about phenytoin, especially for patients ‘with long–standing convul- sions previously unrelieved by phenobarbital’ although Mesantoin outranked it on several points, and indeed he favoured their combined use: ‘Mesantoin and Dilantin are Damon and Pythias in respect to their suitability for joint action. Similarity of action gives a doubled therapeutic effect; the dissimilarity of their side reactions keeps these within bounds.’ Phenacemide was ‘what in athletics might be called a triple threat because, more than any other drug, it acts against each of the three main types of seizures, and especially against the most feared psychomotor seizures. However, it is also a triple threat to the patient himself because of possible effect on the marrow, the liver or the psyche.’ Given one chance in 250 of not surviving this treatment, he Drugs introduced into clinical asked, ‘Is the risk too great?’ Trimethadione practice between 1938 and 1958 (Tridione) ‘heads the list of drugs that are 1938 Dilantin Phenytoin peculiarly beneficial to persons subject to 1941 Diamox Acetazolamide 1946 Tridione Trimethadione petits’. -

Use of Prescribed Psychotropics During Pregnancy: a Systematic Review of Pregnancy, Neonatal, and Childhood Outcomes
brain sciences Review Use of Prescribed Psychotropics during Pregnancy: A Systematic Review of Pregnancy, Neonatal, and Childhood Outcomes Catherine E. Creeley * and Lisa K. Denton Department of Psychology, State University of New York at Fredonia, Fredonia, NY 14063 USA; [email protected] * Correspondence: [email protected]; Tel.: +1-716-673-3890 Received: 19 July 2019; Accepted: 9 September 2019; Published: 14 September 2019 Abstract: This paper reviews the findings from preclinical animal and human clinical research investigating maternal/fetal, neonatal, and child neurodevelopmental outcomes following prenatal exposure to psychotropic drugs. Evidence for the risks associated with prenatal exposure was examined, including teratogenicity, neurodevelopmental effects, neonatal toxicity, and long-term neurobehavioral consequences (i.e., behavioral teratogenicity). We conducted a comprehensive review of the recent results and conclusions of original research and reviews, respectively, which have investigated the short- and long-term impact of drugs commonly prescribed to pregnant women for psychological disorders, including mood, anxiety, and sleep disorders. Because mental illness in the mother is not a benign event, and may itself pose significant risks to both mother and child, simply discontinuing or avoiding medication use during pregnancy may not be possible. Therefore, prenatal exposure to psychotropic drugs is a major public health concern. Decisions regarding drug choice, dose, and duration should be made carefully, by balancing severity, chronicity, and co-morbidity of the mental illness, disorder, or condition against the potential risk for adverse outcomes due to drug exposure. Globally, maternal mental health problems are considered as a major public health challenge, which requires a stronger focus on mental health services that will benefit both mother and child. -

Drugs Which Can Affect Near Vision: a Useful List
Drugs Which Can Affect Near Vision: A Useful List Joanne L. Smith B.Sc., Ph.Phm.* J. Raymond Buncic, M.D., F.R.C.S.(C)t ABSTRACT This paper documents a list of drugs that cause problems with near vision, by virtue of effects on accommodation, occasionally refractive error and diplopia. It is meant as a reference aid to the clinician when confronted with problems of focusing on near objects or print. There are many drugs that have been reported to interfere with near or reading vision, producing blurring, decreased accommodation and diplopia. This paper lists the drugs that have been reported in the literature to produce symptoms which interfere with near vision. Case reports for the listed drugs vary greatly from many to few. The drugs have been divided into the following categories: those causing (A) blurring at near, (B) diplopia and (C) induced myopia. Those drugs which only rarely cause these symptoms have been omitted. From the Departments of Pharmacy* and Ophthalmologyt, The Hospital For Sick Children, Toronto, Ontario, Canada Requests for reprints should be addressed to: Dr. J. Raymond Buncic, Department of Ophthalmology, The Hospital For Sick Children, 555 University Ave., Toronto, Ontario, Canada M5G lX8 TABLE 1 DRUGS COMMONLY CAUSING DIFFICULTY WITH FOCUSING AT NEAR OR BLURRED VISION. DRUG INCIDENCE REFERENCE Antipsychotics Chlorpromazine 14-23 8 Clozapine 5 8,14 Fluphenazine 1.2-4.3 8 Haloperidol 6.8-16 8 Loxapine 12,14 Perphenazine 7.4-17.8 8 Pimozide 20 8 Risperidone 1-2%, >/= 10% 11 Thioridazine 0.6-18 8 Thiothixene 20 8 -

Bypassing the Blood–Brain Barrier: Direct Intracranial Drug Delivery in Epilepsies
pharmaceutics Review Bypassing the Blood–Brain Barrier: Direct Intracranial Drug Delivery in Epilepsies Manuela Gernert 1,2,* and Malte Feja 1,2 1 Department of Pharmacology, Toxicology, and Pharmacy, University of Veterinary Medicine Hannover, Bünteweg 17, D-30559 Hannover, Germany; [email protected] 2 Center for Systems Neuroscience, D-30559 Hannover, Germany * Correspondence: [email protected]; Tel.: +49-(0)511-953-8527 Received: 30 October 2020; Accepted: 21 November 2020; Published: 24 November 2020 Abstract: Epilepsies are common chronic neurological diseases characterized by recurrent unprovoked seizures of central origin. The mainstay of treatment involves symptomatic suppression of seizures with systemically applied antiseizure drugs (ASDs). Systemic pharmacotherapies for epilepsies are facing two main challenges. First, adverse effects from (often life-long) systemic drug treatment are common, and second, about one-third of patients with epilepsy have seizures refractory to systemic pharmacotherapy. Especially the drug resistance in epilepsies remains an unmet clinical need despite the recent introduction of new ASDs. Apart from other hypotheses, epilepsy-induced alterations of the blood–brain barrier (BBB) are thought to prevent ASDs from entering the brain parenchyma in necessary amounts, thereby being involved in causing drug-resistant epilepsy. Although an invasive procedure, bypassing the BBB by targeted intracranial drug delivery is an attractive approach to circumvent BBB-associated drug resistance mechanisms and to lower the risk of systemic and neurologic adverse effects. Additionally, it offers the possibility of reaching higher local drug concentrations in appropriate target regions while minimizing them in other brain or peripheral areas, as well as using otherwise toxic drugs not suitable for systemic administration. -

Fetal Drug Syndromes NEIL GORDON M.D., F.R.C.P
Postgrad Med J: first published as 10.1136/pgmj.54.638.796 on 1 December 1978. Downloaded from Postgraduate Medical Journal (December 1978) 54, 796-798. Fetal drug syndromes NEIL GORDON M.D., F.R.C.P. Booth Hall Children's Hospital, Manchester Summary been previously reported as an example of the Certain drugs which affect fetal development are Coffin-Siris syndrome (Weisswasser et al., 1973). considered. Phenytoin taken during pregnancy may This syndrome consists of absence of the nails of result in infants who remain mentally and physically fingers and toes, growth deficiency, mental retard- retarded, with abnormal craniofacial appearance and ation with coarse features, sparse scalp hair and limb defects. A boy with such a syndrome is reported. hypertrichosis. The craniofacial features include Trimethadione is another drug which has been said to heavy eyebrows, bulbous anteverted nose, small be teratogenic, and to cause a typical phenotype teeth, full lips and a broad mouth (Coffin and Siris, which is described. The fetal alcohol syndrome is 1970). Children described by Senior (1971) with short now well recognized and has similar features to those stature, small toe nails, shortened fifth fingers and related to anti-epileptic drugs taken during pregnancy. broadened nose and mouth have been considered aProtected by copyright. A high level of suspicion of possible associations possible variant of the Coffin-Siris syndrome. between congenital defects and drugs must be main- A number of findings among the children born to tained. mothers taking phenytoin during pregnancy have been reported. There is defective growth and mental Introduction retardation, and limb defects including hypoplasia Among children who are mentally handicapped of phalanges and nails, finger-like thumbs and there are a number whose appearance is strikingly variations in palmar creases and dermatoglyphics. -

FDA Listing of Established Pharmacologic Class Text Phrases January 2021
FDA Listing of Established Pharmacologic Class Text Phrases January 2021 FDA EPC Text Phrase PLR regulations require that the following statement is included in the Highlights Indications and Usage heading if a drug is a member of an EPC [see 21 CFR 201.57(a)(6)]: “(Drug) is a (FDA EPC Text Phrase) indicated for Active Moiety Name [indication(s)].” For each listed active moiety, the associated FDA EPC text phrase is included in this document. For more information about how FDA determines the EPC Text Phrase, see the 2009 "Determining EPC for Use in the Highlights" guidance and 2013 "Determining EPC for Use in the Highlights" MAPP 7400.13.