Efficient Generation of Human Natural Killer Cell Lines by Viral Transformation
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Costimulation of T-Cell Activation and Virus Production by B7 Antigen on Activated CD4+ T Cells from Human Immunodeficiency Virus Type 1-Infected Donors OMAR K
Proc. Natl. Acad. Sci. USA Vol. 90, pp. 11094-11098, December 1993 Immunology Costimulation of T-cell activation and virus production by B7 antigen on activated CD4+ T cells from human immunodeficiency virus type 1-infected donors OMAR K. HAFFAR, MOLLY D. SMITHGALL, JEFFREY BRADSHAW, BILL BRADY, NITIN K. DAMLE*, AND PETER S. LINSLEY Bristol-Myers Squibb Pharmaceutical Research Institute, Seattle, WA 98121 Communicated by Leon E. Rosenberg, August 3, 1993 (receivedfor review April 29, 1993) ABSTRACT Infection with the human immunodeficiency sequence (CTLA-4) (34), a protein structurally related to virus type 1 (HIV-1) requires T-cefl activation. Recent studies CD28 but only expressed on T cells after activation (12). have shown that interactions of the T-lymphocyte receptors CTLA-4 acts cooperatively with CD28 to bind B7 and deliver CD28 and CTLA-4 with their counter receptor, B7, on antigen- T-cell costimulatory signals (13). presenting cells are required for optimal T-cell activation. Here Because of the importance of the CD28/CTLA-4 and B7 we show that HIV-1 infection is associated with decreased interactions in immune responses, it is likely that these expression of CD28 and increased expression of B7 on CD4+ interactions are also important during HIV-1 infection. Stud- T-cell lines generated from seropositive donors by afloantigen ies with anti-CD28 monoclonal antibodies (mAbs) suggested stimulation. Loss of CD28 expression was not seen on CD4+ a role for CD28 in up-regulating HIV-1 long terminal repeat- T-ceU lines from seronegative donors, but up-regulation of B7 driven transcription of a reporter gene in leukemic cell lines expression was observed upon more prolonged culture. -

Human and Mouse CD Marker Handbook Human and Mouse CD Marker Key Markers - Human Key Markers - Mouse
Welcome to More Choice CD Marker Handbook For more information, please visit: Human bdbiosciences.com/eu/go/humancdmarkers Mouse bdbiosciences.com/eu/go/mousecdmarkers Human and Mouse CD Marker Handbook Human and Mouse CD Marker Key Markers - Human Key Markers - Mouse CD3 CD3 CD (cluster of differentiation) molecules are cell surface markers T Cell CD4 CD4 useful for the identification and characterization of leukocytes. The CD CD8 CD8 nomenclature was developed and is maintained through the HLDA (Human Leukocyte Differentiation Antigens) workshop started in 1982. CD45R/B220 CD19 CD19 The goal is to provide standardization of monoclonal antibodies to B Cell CD20 CD22 (B cell activation marker) human antigens across laboratories. To characterize or “workshop” the antibodies, multiple laboratories carry out blind analyses of antibodies. These results independently validate antibody specificity. CD11c CD11c Dendritic Cell CD123 CD123 While the CD nomenclature has been developed for use with human antigens, it is applied to corresponding mouse antigens as well as antigens from other species. However, the mouse and other species NK Cell CD56 CD335 (NKp46) antibodies are not tested by HLDA. Human CD markers were reviewed by the HLDA. New CD markers Stem Cell/ CD34 CD34 were established at the HLDA9 meeting held in Barcelona in 2010. For Precursor hematopoetic stem cell only hematopoetic stem cell only additional information and CD markers please visit www.hcdm.org. Macrophage/ CD14 CD11b/ Mac-1 Monocyte CD33 Ly-71 (F4/80) CD66b Granulocyte CD66b Gr-1/Ly6G Ly6C CD41 CD41 CD61 (Integrin b3) CD61 Platelet CD9 CD62 CD62P (activated platelets) CD235a CD235a Erythrocyte Ter-119 CD146 MECA-32 CD106 CD146 Endothelial Cell CD31 CD62E (activated endothelial cells) Epithelial Cell CD236 CD326 (EPCAM1) For Research Use Only. -
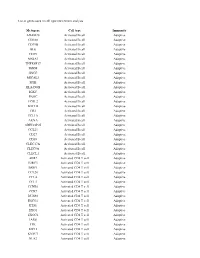
List of Genes Used in Cell Type Enrichment Analysis
List of genes used in cell type enrichment analysis Metagene Cell type Immunity ADAM28 Activated B cell Adaptive CD180 Activated B cell Adaptive CD79B Activated B cell Adaptive BLK Activated B cell Adaptive CD19 Activated B cell Adaptive MS4A1 Activated B cell Adaptive TNFRSF17 Activated B cell Adaptive IGHM Activated B cell Adaptive GNG7 Activated B cell Adaptive MICAL3 Activated B cell Adaptive SPIB Activated B cell Adaptive HLA-DOB Activated B cell Adaptive IGKC Activated B cell Adaptive PNOC Activated B cell Adaptive FCRL2 Activated B cell Adaptive BACH2 Activated B cell Adaptive CR2 Activated B cell Adaptive TCL1A Activated B cell Adaptive AKNA Activated B cell Adaptive ARHGAP25 Activated B cell Adaptive CCL21 Activated B cell Adaptive CD27 Activated B cell Adaptive CD38 Activated B cell Adaptive CLEC17A Activated B cell Adaptive CLEC9A Activated B cell Adaptive CLECL1 Activated B cell Adaptive AIM2 Activated CD4 T cell Adaptive BIRC3 Activated CD4 T cell Adaptive BRIP1 Activated CD4 T cell Adaptive CCL20 Activated CD4 T cell Adaptive CCL4 Activated CD4 T cell Adaptive CCL5 Activated CD4 T cell Adaptive CCNB1 Activated CD4 T cell Adaptive CCR7 Activated CD4 T cell Adaptive DUSP2 Activated CD4 T cell Adaptive ESCO2 Activated CD4 T cell Adaptive ETS1 Activated CD4 T cell Adaptive EXO1 Activated CD4 T cell Adaptive EXOC6 Activated CD4 T cell Adaptive IARS Activated CD4 T cell Adaptive ITK Activated CD4 T cell Adaptive KIF11 Activated CD4 T cell Adaptive KNTC1 Activated CD4 T cell Adaptive NUF2 Activated CD4 T cell Adaptive PRC1 Activated -

Markers of T Cell Senescence in Humans
International Journal of Molecular Sciences Review Markers of T Cell Senescence in Humans Weili Xu 1,2 and Anis Larbi 1,2,3,4,5,* 1 Biology of Aging Program and Immunomonitoring Platform, Singapore Immunology Network (SIgN), Agency for Science Technology and Research (A*STAR), Immunos Building, Biopolis, Singapore 138648, Singapore; [email protected] 2 School of Biological Sciences, Nanyang Technological University, Singapore 637551, Singapore 3 Department of Microbiology, National University of Singapore, Singapore 117597, Singapore 4 Department of Geriatrics, Faculty of Medicine, University of Sherbrooke, Sherbrooke, QC J1K 2R1, Canada 5 Faculty of Sciences, University ElManar, Tunis 1068, Tunisia * Correspondence: [email protected]; Tel.: +65-6407-0412 Received: 31 May 2017; Accepted: 26 July 2017; Published: 10 August 2017 Abstract: Many countries are facing the aging of their population, and many more will face a similar obstacle in the near future, which could be a burden to many healthcare systems. Increased susceptibility to infections, cardiovascular and neurodegenerative disease, cancer as well as reduced efficacy of vaccination are important matters for researchers in the field of aging. As older adults show higher prevalence for a variety of diseases, this also implies higher risk of complications, including nosocomial infections, slower recovery and sequels that may reduce the autonomy and overall quality of life of older adults. The age-related effects on the immune system termed as “immunosenescence” can be exemplified by the reported hypo-responsiveness to influenza vaccination of the elderly. T cells, which belong to the adaptive arm of the immune system, have been extensively studied and the knowledge gathered enables a better understanding of how the immune system may be affected after acute/chronic infections and how this matters in the long run. -

Human Β‑1,3‑Glucuronyltransferase 1/B3GAT1 Antibody Monoclonal Mouse Igg2a Clone # 1002707 Catalog Number: MAB8560
Human β‑1,3‑Glucuronyltransferase 1/B3GAT1 Antibody Monoclonal Mouse IgG2A Clone # 1002707 Catalog Number: MAB8560 DESCRIPTION Species Reactivity Human Specificity Detects human β-1,3-Glucuronyltransferase 1/B3GAT1 in direct ELISAs. Source Monoclonal Mouse IgG2A Clone # 1002707 Purification Protein A or G purified from hybridoma culture supernatant Immunogen Chinese Hamster Ovary cell line, CHO-derived human β‑1,3‑Glucuronyltransferase 1/B3GAT1 His25-Ile334 Accession # Q9P2W7 Formulation Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details. *Small pack size (-SP) is supplied either lyophilized or as a 0.2 μm filtered solution in PBS. APPLICATIONS Please Note: Optimal dilutions should be determined by each laboratory for each application. General Protocols are available in the Technical Information section on our website. Recommended Sample Concentration Immunohistochemistry 5-25 µg/mL See Below DATA Immunohistochemistry β-1,3-Glucuronyltransferase 1/B3GAT1 in Human Brain. β-1,3-Glucuronyltransferase 1/B3GAT1 was detected in immersion fixed paraffin-embedded sections of human brain (cortex) using Mouse Anti-Human β-1,3- Glucuronyltransferase 1/B3GAT1 Monoclonal Antibody (Catalog # MAB8560) at 5 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Mouse IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC001). Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm in neurons. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents. PREPARATION AND STORAGE Reconstitution Reconstitute at 0.5 mg/mL in sterile PBS. -

Papers J Clin Pathol: First Published As 10.1136/Jcp.49.7.539 on 1 July 1996
Clin Pathol 1996;49:539-544 539 Papers J Clin Pathol: first published as 10.1136/jcp.49.7.539 on 1 July 1996. Downloaded from Differential expression of T cell antigens in normal peripheral blood lymphocytes: a quantitative analysis by flow cytometry L Ginaldi, N Farahat, E Matutes, M De Martinis, R Morilla, D Catovsky Abstract diagnosis by comparison with findings in Aims-To obtain reference values of the normal counterparts. level of expression of T cell antigens on ( Clin Pathol 1996;49:539-544) normal lymphocyte subsets in order to disclose differences which could reflect Keywords: flow cytometry, T cell antigens, peripheral their function or maturation stages, or blood lymphocytes. both. Methods-Peripheral blood from 15 healthy donors was processed by flow The most common use of flow cytometry is to cytometry with triple colour analysis. For determine the percent of "positive" or "nega- each sample phycoerythrin (PE) conju- tive" cells for each antigen in different cell gated CD2, CD4, CD5, CD8, and CD56 populations. However, valuable information is monoclonal antibodies were combined lost when the relative intensities of the positive with Cy5-R-phycoerythrin (TC) conju- cells, reflecting antigenic densities, are not considered. A cell population with a well gated CD3 and fluorescein isothiocyanate http://jcp.bmj.com/ (FITC) conjugated CD7; CD2- and defined phenotype, positive for one antigen, CD7-PE were also combined with might be heterogeneous with regard to its level CD3-TC and CD4-FITC. Standard mi- of expression. This may reflect different crobeads with different capacities to bind functional or maturational states, or both, or mouse immunoglobulins were used to identify subpopulations on the basis of the dif- convert the mean fluorescence intensity ferent numbers of molecules of antigen per cell. -

CD18) in a Patient with Leukocyte Adhesion Molecule (Leu-CAM) Deficiency
Point mutations impairing cell surface expression of the common beta subunit (CD18) in a patient with leukocyte adhesion molecule (Leu-CAM) deficiency. M A Arnaout, … , D G Tenen, D M Fathallah J Clin Invest. 1990;85(3):977-981. https://doi.org/10.1172/JCI114529. Research Article The leukocyte adhesion molecules CD11a/CD18, CD11b/CD18, and CD11c/CD18 (Leu-CAM) are members of the integrin receptor family and mediate crucial adhesion-dependent functions in leukocytes. The molecular basis for their deficient cell surface expression was sought in a patient suffering from severe and recurrent bacterial infections. Previous studies revealed that impaired cell surface expression of Leu-CAM is secondary to heterogeneous structural defects in the common beta subunit (CD18). Cloning and sequencing of complementary DNA encoding for CD18 in this patient revealed two mutant alleles, each representing a point mutation in the coding region of CD18 and resulting in an amino acid substitution. Each mutant allele results in impaired CD18 expression on the cell surface membrane of transfected COS M6 cells. One substitution involves an arginine residue (Arg593----cysteine) that is conserved in the highly homologous fourth cysteine-rich repeats of other mammalian integrin subfamilies. The other substitution involves a lysine residue (Lys196----threonine) located within another highly conserved region in integrins. These data identify crucial residues and regions necessary for normal cell surface expression of CD18 and possibly other integrin beta subunits and define a molecular basis for impaired cell surface expression of CD18 in this patient. Find the latest version: https://jci.me/114529/pdf Rapid Publication Point Mutations Impairing Cell Surface Expression of the Common ,B Subunit (CD18) in a Patient with Leukocyte Adhesion Molecule (Leu-CAM) Deficiency M. -

Differential Integrin Adhesome Expression Defines Human Natural
bioRxiv preprint doi: https://doi.org/10.1101/2020.12.01.404806; this version posted December 1, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. Differential integrin adhesome expression defines human natural killer cell residency and developmental stage Everardo Hegewisch Solloa1, Seungmae Seo1, Bethany L. Mundy-Bosse2, Anjali Mishra3,a, Erik Waldman4,b, Sarah Maurrasse4,b, Eli Grunstein4, Thomas J. Connors5, Aharon G. Freud2,6, and Emily M. Mace1 1Department of Pediatrics, Columbia University College of Physicians and Surgeons, New York NY 10032 2Division of Hematology, Department of Internal Medicine, The Ohio State University, Columbus, OH 43210, USA; Comprehensive Cancer Center and The James Cancer Hospital and Solove Research Institute, The Ohio State University, Columbus, OH 43210 33Division of Dermatology, Department of Internal Medicine, The Ohio State University, Columbus, OH 43210, USA; Comprehensive Cancer Center and The James Cancer Hospital and Solove Research Institute, The Ohio State University, Columbus, OH 43210 4Department of Otolaryngology - Head and Neck Surgery, Columbia University Medical Center, New York, New York 10032 5Department of Pediatrics, Division of Pediatric Critical Care and Hospital Medicine, Columbia University Irving Medical Center, New York, NY 10024 6Department of Pathology, The Ohio State University, Columbus, -

The Immunological Synapse and CD28-CD80 Interactions Shannon K
© 2001 Nature Publishing Group http://immunol.nature.com ARTICLES The immunological synapse and CD28-CD80 interactions Shannon K. Bromley1,Andrea Iaboni2, Simon J. Davis2,Adrian Whitty3, Jonathan M. Green4, Andrey S. Shaw1,ArthurWeiss5 and Michael L. Dustin5,6 Published online: 19 November 2001, DOI: 10.1038/ni737 According to the two-signal model of T cell activation, costimulatory molecules augment T cell receptor (TCR) signaling, whereas adhesion molecules enhance TCR–MHC-peptide recognition.The structure and binding properties of CD28 imply that it may perform both functions, blurring the distinction between adhesion and costimulatory molecules. Our results show that CD28 on naïve T cells does not support adhesion and has little or no capacity for directly enhancing TCR–MHC- peptide interactions. Instead of being dependent on costimulatory signaling, we propose that a key function of the immunological synapse is to generate a cellular microenvironment that favors the interactions of potent secondary signaling molecules, such as CD28. The T cell receptor (TCR) interaction with complexes of peptide and as CD2 and CD48, which suggests that CD28 might have a dual role as major histocompatibility complex (pMHC) is central to the T cell an adhesion and a signaling molecule4. Coengagement of CD28 with response. However, efficient T cell activation also requires the partici- the TCR has a number of effects on T cell activation; these include pation of additional cell-surface receptors that engage nonpolymorphic increasing sensitivity to TCR stimulation and increasing the survival of ligands on antigen-presenting cells (APCs). Some of these molecules T cells after stimulation5. CD80-transfected APCs have been used to are involved in the “physical embrace” between T cells and APCs and assess the temporal relationship of TCR and CD28 signaling, as initiat- are characterized as adhesion molecules. -

(P -Value<0.05, Fold Change≥1.4), 4 Vs. 0 Gy Irradiation
Table S1: Significant differentially expressed genes (P -Value<0.05, Fold Change≥1.4), 4 vs. 0 Gy irradiation Genbank Fold Change P -Value Gene Symbol Description Accession Q9F8M7_CARHY (Q9F8M7) DTDP-glucose 4,6-dehydratase (Fragment), partial (9%) 6.70 0.017399678 THC2699065 [THC2719287] 5.53 0.003379195 BC013657 BC013657 Homo sapiens cDNA clone IMAGE:4152983, partial cds. [BC013657] 5.10 0.024641735 THC2750781 Ciliary dynein heavy chain 5 (Axonemal beta dynein heavy chain 5) (HL1). 4.07 0.04353262 DNAH5 [Source:Uniprot/SWISSPROT;Acc:Q8TE73] [ENST00000382416] 3.81 0.002855909 NM_145263 SPATA18 Homo sapiens spermatogenesis associated 18 homolog (rat) (SPATA18), mRNA [NM_145263] AA418814 zw01a02.s1 Soares_NhHMPu_S1 Homo sapiens cDNA clone IMAGE:767978 3', 3.69 0.03203913 AA418814 AA418814 mRNA sequence [AA418814] AL356953 leucine-rich repeat-containing G protein-coupled receptor 6 {Homo sapiens} (exp=0; 3.63 0.0277936 THC2705989 wgp=1; cg=0), partial (4%) [THC2752981] AA484677 ne64a07.s1 NCI_CGAP_Alv1 Homo sapiens cDNA clone IMAGE:909012, mRNA 3.63 0.027098073 AA484677 AA484677 sequence [AA484677] oe06h09.s1 NCI_CGAP_Ov2 Homo sapiens cDNA clone IMAGE:1385153, mRNA sequence 3.48 0.04468495 AA837799 AA837799 [AA837799] Homo sapiens hypothetical protein LOC340109, mRNA (cDNA clone IMAGE:5578073), partial 3.27 0.031178378 BC039509 LOC643401 cds. [BC039509] Homo sapiens Fas (TNF receptor superfamily, member 6) (FAS), transcript variant 1, mRNA 3.24 0.022156298 NM_000043 FAS [NM_000043] 3.20 0.021043295 A_32_P125056 BF803942 CM2-CI0135-021100-477-g08 CI0135 Homo sapiens cDNA, mRNA sequence 3.04 0.043389246 BF803942 BF803942 [BF803942] 3.03 0.002430239 NM_015920 RPS27L Homo sapiens ribosomal protein S27-like (RPS27L), mRNA [NM_015920] Homo sapiens tumor necrosis factor receptor superfamily, member 10c, decoy without an 2.98 0.021202829 NM_003841 TNFRSF10C intracellular domain (TNFRSF10C), mRNA [NM_003841] 2.97 0.03243901 AB002384 C6orf32 Homo sapiens mRNA for KIAA0386 gene, partial cds. -

(12) United States Patent (405
USOO6291239B1 (12) United States Patent (10) Patent No.: US 6,291,239 B1 Prodinger et al. (45) Date of Patent: Sep. 18, 2001 (54) MONOCLONAL ANTIBODY Recess Formed Between SCR1 and SCR 2 of Complement Receptor Type Two', Prodinger. (76) Inventors: Wolfgang Prodinger, Kirschentalgasse Immunopharmacology, vol. 38, No. 1-2, Dec., 1997, pp. 16, A-6020 Innsbruck; Michael 141-148, XP002089604, “Expression in Insect Cells of the Schwendinger, Semmelweisgasse 4, Functional Domain of CD21 (Complement Receptor Type A-7100 Neusiedl/See, both of (AT) Two) as a Truncated... ", Prodinger. Journal of Immunology, vol. 156, No. 7 Apr. 1, 1996, pp. (*) Notice: Subject to any disclaimer, the term of this 2580–2584, XP002089605, “Ligation of the Functional patent is extended or adjusted under 35 Domain of Complement Receptor Type 2 (CR2, CD21) is U.S.C. 154(b) by 0 days. Relevant for Complex . , Prodinger. Journal of Immunology, vol. 154, No. 10, May 15, 1995, pp. (21) Appl. No.: 09/276,296 5426–5435, XP002089606, “Characterization of a Comple (22) Filed: Mar. 25, 1999 ment Receptor 2 (CR2, CD21) Ligand Binding Site for C3”, H. Molina et al. (51) Int. Cl. ............................... C12N 5/06; C12O 1/70; Journal of Immunology, vol. 161, Nov. 1, 1998, pp. G01N 33/53; CO7K 16/00; A61K 39/395 4604–4610, XP002089607, “Characterization of C3dg Binding to a Recess Formed Between Short Consensus (52) U.S. Cl. ............................ 435/339.1; 435/5; 435/7.1; Repeats 1 and 2 ... ', Prodinger. 435/334; 530/388.1; 530/388.35; 424/141.1; 424/143.1; 424/159.1 Primary Examiner Hankyel T. -

Identification of Key Pathways and Genes in Dementia Via Integrated Bioinformatics Analysis
bioRxiv preprint doi: https://doi.org/10.1101/2021.04.18.440371; this version posted July 19, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Identification of Key Pathways and Genes in Dementia via Integrated Bioinformatics Analysis Basavaraj Vastrad1, Chanabasayya Vastrad*2 1. Department of Biochemistry, Basaveshwar College of Pharmacy, Gadag, Karnataka 582103, India. 2. Biostatistics and Bioinformatics, Chanabasava Nilaya, Bharthinagar, Dharwad 580001, Karnataka, India. * Chanabasayya Vastrad [email protected] Ph: +919480073398 Chanabasava Nilaya, Bharthinagar, Dharwad 580001 , Karanataka, India bioRxiv preprint doi: https://doi.org/10.1101/2021.04.18.440371; this version posted July 19, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Abstract To provide a better understanding of dementia at the molecular level, this study aimed to identify the genes and key pathways associated with dementia by using integrated bioinformatics analysis. Based on the expression profiling by high throughput sequencing dataset GSE153960 derived from the Gene Expression Omnibus (GEO), the differentially expressed genes (DEGs) between patients with dementia and healthy controls were identified. With DEGs, we performed a series of functional enrichment analyses. Then, a protein–protein interaction (PPI) network, modules, miRNA-hub gene regulatory network and TF-hub gene regulatory network was constructed, analyzed and visualized, with which the hub genes miRNAs and TFs nodes were screened out. Finally, validation of hub genes was performed by using receiver operating characteristic curve (ROC) analysis.