Aspen Plus7 10 STEADY STATE SIMULATION
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

1998 LIST of PUBLICATIONS
Instytut Niskich Temperatur i Badań Strukturalnych PAN Wrocław, 31 sierpnia 2007 LISTA PUBLIKACJI 1998 LIST of PUBLICATIONS KSIĄŻKI, MONOGRAFIE i ARTYKUŁY PRZEGLĄDOWE Books, Monographs & Reviews 1. D.Aoki, Y.Katayama, S.Nojiri, R.Settai, Y.Inada, K.Sugiyama, Y.Onuki,¯ H.Harima, Z. Kletowski, Antiferroquadrupolar Ordering and Fermi Surface Property in PrPb3 . In: Japanese Journal of Applied Physics, Series 11., ed. by *** (Tokyo: The Institute of Pure and Applied Physics, 1999) pp. 188–90. 2. E. Gałdecka, X-ray Diffraction Methods: Single Crystal. In: International Tables for Crystallography, Vol. C, 2nd ed., ed. by E. Prince & A.J.C. Wilson (Dordrecht: Kluwer Acad. 1998) Ch. 5.3, pp. 501–31. 3. M. Kazimierski (Editor) Proceedings of 11th Seminar on Phase Transitions & Critical Phenomena (Wrocław & Polanica Zdrój, Poland, May 1998) (Wrocław: Inst. of Low Temperature and Structure Research, 1998) 198pp. 4. K. Maruszewski, Spektroskopia aktywowanych zeolitów i materiałów zol–żelowych. [Spectroscopic Investigation of Activated Molecular Sieves and Sol–Gel Materials.] (Wrocław: Instytut Niskich Temperatur i Badań Strukturalnych., 1998) 126 pp [in Polish]. 5. W. Suski, T.Palewski, [1 Magnetic and Related Properties of Pnictides and Chalcogenides. 1.2 Pnictides and chalcogenides based on lanthanides.] 1.2.1 Lanthanide monopnictides. In: Landolt–Börnstein Numerical Data and Functional Relationship in Science and Technology, New Series, ed. by W.Martienssen, Group III: Condensed Matter, Vol. 27: Magnetic Properties of Non-Metallic Inorganic Compounds Based on Transition Elements, Subvol. B1: Pnictides and Chalcogenides I, ed. by H.P.J. Wijn (Berlin: Springer-Vg 1998) pp. 1–428. 6. W. Suski, T.Palewski, [1 Magnetic and Related Properties of Pnictides and Chalcogenides. -

Recycling of Hazardous Waste from Tertiary Aluminium Industry in A
CORE Metadata, citation and similar papers at core.ac.uk Provided by Digital.CSIC 1 Recycling of hazardous waste from tertiary aluminium 2 industry in a value-added material 3 4 Laura Gonzalo-Delgado1, Aurora López-Delgado1*, Félix Antonio López1, Francisco 5 José Alguacil1 and Sol López-Andrés2 6 7 1Nacional Centre for Metallurgical Research, CSIC. Avda. Gregorio del Amo, 8. 28040. 8 Madrid. Spain. 9 2Dpt. Crystallography and Mineralogy. Fac. of Geology. University Complutense of 10 Madrid. Spain. 11 *Corresponding author e-mail: [email protected] 12 13 Abstract 14 15 The recent European Directive on waste, 2008/98/EC seeks to reduce the 16 exploitation of natural resources through the use of secondary resource management. 17 Thus the main objective of this paper is to explore how a waste could cease to be 18 considered as waste and could be utilized for a specific purpose. In this way, a 19 hazardous waste from the tertiary aluminium industry was studied for its use as a raw 20 material in the synthesis of an added value product, boehmite. This waste is classified as 21 a hazardous residue, principally because in the presence of water or humidity, it releases 22 toxic gases such as hydrogen, ammonia, methane and hydrogen sulphide. The low 23 temperature hydrothermal method developed permits the recovery of 90% of the 24 aluminium content in the residue in the form of a high purity (96%) AlOOH (boehmite). 25 The method of synthesis consists of an initial HCl digestion followed by a gel 26 precipitation. In the first stage a 10% HCl solution is used to yield a 12.63 g.l-1 Al3+ 27 solution. -

Tutorial 2 FORMULAS, PERCENTAGE COMPOSITION
T-6 Tutorial 2 FORMULAS, PERCENTAGE COMPOSITION, AND THE MOLE FORMULAS: A chemical formula shows the elemental composition of a substance: the chemical symbols show what elements are present and the numerical subscripts show how many atoms of each element there are in a formula unit. Examples: NaCl: one sodium atom, one chlorine atom in a formula unit CaCl2: one calcium atom, two chlorine atoms in a formula unit Mg3N2: three magnesium atoms, two nitrogen atoms in a formula unit The presence of a metal in a chemical formula indicates an ionic compound, which is composed of positive ions (cations) and negative ions (anions). A formula with only nonmetals indicates a + molecular compound (unless it is an ammonium, NH4 , compound). Only ionic compounds are considered in this Tutorial. There are tables of common ions in your lecture text, p 56 (cations) and p 57 (anions). A combined table of these same ions can be found on the inside back cover of the lecture text. A similar list is on the next page; all formulas needed in this and subsequent Tutorial problems can be written with ions from this list. Writing formulas for ionic compounds is very straightforward: TOTAL POSITIVE CHARGES MUST BE THE SAME AS TOTAL NEGATIVE CHARGES. The formula must be neutral. The positive ion is written first in the formula and the name of the compound is the two ion names. EXAMPLE: Write the formula for potassium chloride. The name tells you there are potassium, K+, and chloride, Cl–, ions. Each potassium ion is +1 and each chloride ion is -1: one of each is needed, and the formula for potassium chloride is KCl. -

A Guide to Export Controls
Foreign Affairs, Trade and Affaires étrangères, Commerce et Development Canada Développment Canada A Guide To CANADA’S EXPORT CONTROLS December 2012 Introduction The issuance of export permits is administered by the Export Controls Division (TIE) of Foreign Affairs, Trade and Development Canada (DFATD). TIE provides assistance to exporters in determining if export permits are required. It also publishes brochures and Notices to Exporters that are freely available on request and on our website www.exportcontrols.gc.ca. How to contact us: Export Controls Division (TIE) Foreign Affairs, Trade and Development Canada 111 Sussex Drive Ottawa, Ontario K1A 0G2 Telephone: (613) 996-2387 Facsimile: (613) 996-9933 Email: [email protected] For information on how to apply for an export permit and additional information on export controls please refer to our website. To enquire on the status of an export permit application: Recognized EXCOL users can check the status of an export permit application on-line. Non-recognized users can call (613) 996-2387 or email [email protected] and quote your export permit application identification (ref ID) number. Export Controls Division website: www.exportcontrols.gc.ca This Guide, at time of publication, encompasses the list of items enumerated on the Export Control List (ECL) that are controlled for export in accordance with Canadian foreign policy, including Canada’s participation in multilateral export control regimes and bilateral agreements. Unless otherwise specified, the export controls contained in this Guide apply to all destinations except the United States. Canada’s Export Control List can be found at the Department of Justice website at http://canada.justice.gc.ca/. -

(12) Patent Application Publication (10) Pub. No.: US 2011/0027386 A1 Kurihara Et Al
US 20110027386A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2011/0027386 A1 Kurihara et al. (43) Pub. Date: Feb. 3, 2011 (54) ANTMICROBAL. ZEOLITE AND (30) Foreign Application Priority Data ANTMICROBAL COMPOSITION Feb. 22, 2006 (JP) ................................. 2006-045241 (75) Inventors: Yasuo Kurihara, Nagoya-shi (JP); Kumiko Miyake, Nagoya-shi (JP); Publication Classification Masashi Uchida, Nagoya-shi (JP) (51) Int. Cl. Correspondence Address: AOIN 59/6 (2006.01) NIXON & VANDERHYE, PC COB 39/02 (2006.01) 901 NORTH GLEBE ROAD, 11TH FLOOR AOIP I/00 (2006.01) ARLINGTON, VA 22203 (US) (52) U.S. Cl. .......................... 424/618; 423/701; 423/700 (73) Assignee: Sinanen Zeomic Co., Ltd., (57) ABSTRACT Nagoya-Shi (JP) The present invention relates to antimicrobial zeolite which comprises zeolite whereina hardly soluble zinc salt is formed (21) Appl. No.: 12/923,854 within fine pores present therein and an antimicrobial com position which comprises the foregoing antimicrobial Zeolite (22) Filed: Oct. 12, 2010 in an amount ranging from 0.05 to 80% by mass. The antimi crobial Zeolite according to the present invention can widely Related U.S. Application Data be applied, without causing any color change, even to the (63) Continuation of application No. 1 1/705,460, filed on goods which undergo color changes with the elapse of time Feb. 13, 2007. when the conventional antimicrobial zeolite is added. US 2011/002738.6 A1 Feb. 3, 2011 ANTMICROBAL. ZEOLITE AND 3. An antimicrobial composition comprising the foregoing ANTMICROBAL COMPOSITION antimicrobial zeolite as set forth in the foregoing item 1 or 2 in an amount ranging from 0.05 to 80% by mass. -
Chapter 13.Pptx
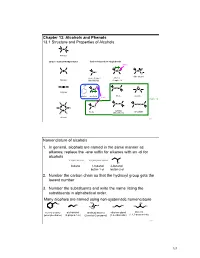
Chapter 13: Alcohols and Phenols 13.1 Structure and Properties of Alcohols C C Alkanes Carbon - Carbon Multiple Bonds Carbon-heteroatom single bonds basic O C C C N C N C X O nitro alkane X= F, Cl, Br, I amines Alkenes Alkyl Halide Chapter 23 OH C C H O C O C C O C C Alkynes phenol alcohols ethers epoxide acidic Chapter 14 H H H C S C C C C S S C C S C C H C C sulfides thiols disulfide H H (thioethers) Arenes 253 Nomenclature of alcohols 1. In general, alcohols are named in the same manner as alkanes; replace the -ane suffix for alkanes with an -ol for alcohols CH3CH2CH2CH3 CH3CH2CH2CH2OH OH butane 1-butanol 2-butanol butan-1-ol butan-2-ol 2. Number the carbon chain so that the hydroxyl group gets the lowest number 3. Number the substituents and write the name listing the substituents in alphabetical order. Many alcohols are named using non-systematic nomenclature H C OH 3 OH OH C OH OH HO OH H3C HO H3C benzyl alcohol allyl alcohol tert-butyl alcohol ethylene glycol glycerol (phenylmethanol) (2-propen-1-ol) (2-methyl-2-propanol) (1,2-ethanediol) (1,2,3-propanetriol) 254 127 Alcohols are classified according to the H R C OH C OH H H degree of substitution of the carbon bearing H H 1° carbon the -OH group methanol primary alcohol primary (1°) : one alkyl substituent R R C OH C OH R R secondary (2°) : two alkyl substituents H R 2° carbon 3° carbon tertiary (3°) : three alkyl substituents secondary alcohol tertiary alcohol Physical properties of alcohols – the C-OH bond of alcohols has a significant dipole moment. -

Department of Chemistry
ST.PHILOMENA’S COLLEGE (AUTONOMOUS), MYSURU (AFFILIATED TO UNIVERSITY OF MYSORE) REACCREDITED BY NAAC WITH A GRADE COURSE – B.Sc Three-year six semesters Choice Based Credit System (CBCS) and Continuous Assessment & Grading Pattern (CAGP) Under Graduate Programme under Autonomous Structure Academic year 2018-19 onwards DEPARTMENT OF CHEMISTRY St. Philomena’s College (Autonomous) Mysuru. B.Sc., Chemistry Syllabus - CBCS Scheme 2018-19 onwards. Page 1 PREAMBLE For the development of any Society, Science education plays an important role. Chemistry, being a major component of Science, is one of the increasingly important disciplinary areas of Science. Chemistry, which is also studied at the Bachelor’s degree programme for years, has been witnessing a slow transition from an analog to a much- needed one. It is an experimental science and students need to be trained both in the theoretical & practical aspects to get expertise. Moreover, the topics prescribed should provide in-depth knowledge of the subject and also the relevant basic allied subjects. Under this context, in order to make the U. G. teaching more effective and meaningful, revamping the syllabus is the need of the hour. It is certain that systematic and planned curricula from first to the third year shall motivate and encourage students for pursuing higher studies in various disciplines of chemistry such as Inorganic, organic, Physical, Analytical and Bio-Chemistry. This curriculum also enables the students to shoulder the responsibility as chemists in chemical industry. Thus, an updated and content revision of UG Chemistry syllabus is essential to improve its quality at the National and International level and also to meet the present-day challenges of PG and research oriented work after the PG programme. -

SAFETY DATA SHEET Revision Date 28.07.2021 According to Regulation (EC) No
Version 7.0 SAFETY DATA SHEET Revision Date 28.07.2021 according to Regulation (EC) No. 1907/2006 Print Date 02.10.2021 GENERIC EU MSDS - NO COUNTRY SPECIFIC DATA - NO OEL DATA SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1 Product identifiers Product name : Aluminum iodide Product Number : 208493 Brand : Aldrich REACH No. : A registration number is not available for this substance as the substance or its uses are exempted from registration, the annual tonnage does not require a registration or the registration is envisaged for a later registration deadline. CAS-No. : 7784-23-8 1.2 Relevant identified uses of the substance or mixture and uses advised against Identified uses : Laboratory chemicals, Manufacture of substances 1.3 Details of the supplier of the safety data sheet Company : Sigma-Aldrich Inc. 3050 SPRUCE ST ST. LOUIS MO 63103 UNITED STATES Telephone : +1 314 771-5765 Fax : +1 800 325-5052 1.4 Emergency telephone Emergency Phone # : 800-424-9300 CHEMTREC (USA) +1-703- 527-3887 CHEMTREC (International) 24 Hours/day; 7 Days/week SECTION 2: Hazards identification 2.1 Classification of the substance or mixture Classification according to Regulation (EC) No 1272/2008 Skin corrosion (Sub-category 1B), H314 Serious eye damage (Category 1), H318 For the full text of the H-Statements mentioned in this Section, see Section 16. 2.2 Label elements Labelling according Regulation (EC) No 1272/2008 Aldrich- 208493 Page 1 of 8 The life science business of Merck operates as MilliporeSigma in the US and Canada Pictogram Signal word Danger Hazard statement(s) H314 Causes severe skin burns and eye damage. -

Determination of Aluminium As Oxide
DETERMINATION OF ALUMINIUM AS OXIDE By William Blum CONTENTS Page I. Introduction 515 II. General principles 516 III. Historical 516 IV. Precipitation of aluminium hydroxide. 518 1. Hydrogen electrode studies 518 (a) The method 518 (b) Apparatus and solutions employed 518 (c) Results of hydrogen electrode experiments 519 (d) Conclusions from hydrogen electrode experiments 520 2. Selection of an indicator for denning the conditions of precipita- '. tion . 522 3. Factors affecting the form of the precipitate 524 4. Precipitation in the presence of iron 525 V. Washing the precipitate . 525 VI. Separation from other elements 526 VII. Ignition and weighing of the precipitate 528 1. Hygroscopicity of aluminium oxide 529 2. Temperature and time of ignition 529 3. Effect of ammonium chloride upon the ignition 531 VIII. Procedure recommended 532 IX. Confirmatory experiments 532 X. Conclusions '534 I. INTRODUCTION Although a considerable number of precipitants have been pro- posed for the determination of aluminium, direct precipitation of aluminium hydroxide by means of ammonium hydroxide, fol- lowed by ignition to oxide, is most commonly used, especially if no separation from iron is desired, in which latter case special methods must be employed. While the general principles involved in this determination are extremely simple, it has long been recog- nized that certain precautions in the precipitation, washing, and ignition are necessary if accurate results are to be obtained. While, however, most of these details have been studied and dis- cussed by numerous authors, it is noteworthy that few publica- tions or textbooks have taken account of all the factors. In the 515 ; 516 Bulletin of the Bureau of Standards [Voi.i3 present paper it seems desirable, therefore, to assemble the various recommendations and to consider their basis and their accuracy. -

Radical Approaches to Alangium and Mitragyna Alkaloids
Radical Approaches to Alangium and Mitragyna Alkaloids A Thesis Submitted for a PhD University of York Department of Chemistry 2010 Matthew James Palframan Abstract The work presented in this thesis has focused on the development of novel and concise syntheses of Alangium and Mitragyna alkaloids, and especial approaches towards (±)-protoemetinol (a), which is a key precursor of a range of Alangium alkaloids such as psychotrine (b) and deoxytubulosine (c). The approaches include the use of a key radical cyclisation to form the tri-cyclic core. O O O N N N O O O H H H H H H O N NH N Protoemetinol OH HO a Psychotrine Deoxytubulosine b c Chapter 1 gives a general overview of radical chemistry and it focuses on the application of radical intermolecular and intramolecular reactions in synthesis. Consideration is given to the mediator of radical reactions from the classic organotin reagents, to more recently developed alternative hydrides. An overview of previous synthetic approaches to a range of Alangium and Mitragyna alkaloids is then explored. Chapter 2 follows on from previous work within our group, involving the use of phosphorus hydride radical addition reactions, to alkenes or dienes, followed by a subsequent Horner-Wadsworth-Emmons reaction. It was expected that the tri-cyclic core of the Alangium alkaloids could be prepared by cyclisation of a 1,7-diene, using a phosphorus hydride to afford the phosphonate or phosphonothioate, however this approach was unsuccessful and it highlighted some limitations of the methodology. Chapter 3 explores the radical and ionic chemistry of a range of silanes. -

Ammonia Snow-Lines and Ammonium Salts Desorption ? F
Astronomy & Astrophysics manuscript no. aanda_ammonium-salts ©ESO 2021 April 22, 2021 Ammonia snow-lines and ammonium salts desorption ? F. Kruczkiewicz1; 2; 3, J. Vitorino2, E. Congiu2, P. Theulé1, and F. Dulieu2 1 Aix Marseille Univ, CNRS, CNES, LAM, Marseille, France e-mail: [email protected] 2 CY Cergy Paris Université, Observatoire de Paris, PSL University, Sorbonne Université, CNRS, LERMA, F-95000, Cergy, France 3 Max-Planck-Institut für extraterrestrische Physik, Gießenbachstraße 1, Garching, 85748, Germany Received –; accepted – ABSTRACT Context. The nitrogen reservoir in planetary systems is a long standing problem. Part of the N-bearing molecules is probably incor- porated into the ice bulk during the cold phases of the stellar evolution, and may be gradually released into the gas phase when the ice is heated, such as in active comets. The chemical nature of the N-reservoir should greatly influence how, when and in what form N returns to the gas phase, or is incorporated into the refractory material forming planetary bodies. Aims. We present the study the thermal desorption of two ammonium salts: ammonium formate and ammonium acetate from a gold surface and from a water ice substrate. Methods. Temperature-programmed desorption experiments and Fourier transform infrared reflection spectroscopy were conducted to investigate the desorption behavior of ammonium salts. Results. Ammonium salts are semi-volatile species releasing neutral species as major components upon desorption, that is ammonia and the corresponding organic acid (HCOOH and CH3COOH), at temperatures higher than the temperature of thermal desorption of water ice. Their desorption follows a first-order Wigner-Polanyi law. We find the first order kinetic parameters A = 7.7 ± 0.6 × 1015 s−1 −1 20 −1 −1 and Ebind = 68.9 ± 0.1 kJ mol for ammonium formate and A = 3.0 ± 0.4 × 10 s and Ebind = 83.0 ± 0.2 kJ mol for ammonium acetate. -

Chemistry Workbook
Bridging the gap 2020 Ripon Grammar School 1 2 Introduction Advanced level Chemistry is a demanding and exciting course. In order to be prepared for your start in September a number of areas from GCSE chemistry are needed to be ‘known’ thoroughly. To help you make the transition as smoothly as possible we have put together this series of exercises. When you start in September you will be expected to have completed the exercises within this booklet and know the material within. It is by no means ALL you need to know but the very foundations of the exciting journey you are about to start. If you have difficulties or confusions there are a number of suggested online resources you could try. There will be opportunities to discuss concerns with staff at the beginning of the year but you should have made significant headway independently. This booklet contains some notes to act as a reminder. If you struggle with a particular area you should investigate the suggested support resources including your GCSE notes. There are exercises for you to complete, the answers are at the end. Contents Periodic Table 2 Topics Writing formulae 4 Equations 7 Moles 10 Atomic Structure 15 Identifying Bonding and Structure types 19 Answers to exercises 20 3 Writing formulae Objectives: • Know the common ions • Be able to construct formulae for common ionic substances • Know the formulae of some common covalent substances Common Ions (you need to know these): Putting together an ionic formula: The charges must balance. Molecular ions will need to be contained in brackets.