Global Dynamics of a Vaccination Model for Infectious Diseases with Asymptomatic Carriers
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

An Atlas of Potentially Water-Related Diseases in South Africa
AN ATLAS OF POTENTIALLY WATER-RELATED DISEASES IN SOUTH AFRICA Volume 2 Bibliography Report to the WATER RESEARCH COMMISSION by N Coetzee and D E Bourne Department of Community Health University of Cape Town Medical School WRC Report no. 584/2/96 ISBN 1 86845 245 X ISBN Set No 1 86845 246 8 1 CONTENTS VOLUME 2 BIBLIOGRAPHY 1 Introduction 2 2 Amoebiasis 3 3 Arthropod-borne viral diseases 7 4 Cholera 10 5 Diarrhoeal diseases in childhood 14 6 Viral hepatitis A and E 17 7 Leptospirosis 20 3 Pediculosis 22 9 Malaria 23 10 Poliomyelitis 29 11 Scabies 33 12 Schistosomiasis 35 13 Trachoma 39 14 Typhoid Fever 42 15 Intestinal helminthiasis 46 1. INTRODUCTION As many professionals involved in the management of water resources do not have a medical or public health background, it was thought that an explanatory bibliography of the major water related diseases in South Africa would be of use. Each section of the bibliography has (where appropriate) the following sections: 1. Aetiology 2. Transmission 3. Preventive and curative steps 4. South African data 5. References The MEDLINE data base of the national Library of Medicine, Washington DC, and the WATERLIT data base of the CSIR were searched. These were supplemented by South African journals and reports not appearing in the MEDLINE data base. 3 2. AMOEBIASIS 2.1. Aetiology The protozoan parasite Entamoeba histolytica which causes amoebiasis may exist as a hardy infective cyst or a more fragile and potentially pathogenic trophozoite (the "amoebic" form). Amoebiasis most commonly affects the colon and rectum as primary sitas of infection, with extraintestinal (most commonly liver abscesses), local or systemic, spread possible if not treated early. -

Estimating the Instantaneous Asymptomatic Proportion with a Simple Approach: Exemplified with the Publicly Available COVID-19 Surveillance Data in Hong Kong
ORIGINAL RESEARCH published: 03 May 2021 doi: 10.3389/fpubh.2021.604455 Estimating the Instantaneous Asymptomatic Proportion With a Simple Approach: Exemplified With the Publicly Available COVID-19 Surveillance Data in Hong Kong Chunyu Li 1†, Shi Zhao 2,3*†, Biao Tang 4,5, Yuchen Zhu 1, Jinjun Ran 6, Xiujun Li 1*‡ and Daihai He 7*‡ 1 2 Edited by: Department of Biostatistics, School of Public Health, Cheeloo College of Medicine, Shandong University, Jinan, China, JC 3 Catherine Ropert, School of Public Health and Primary Care, Chinese University of Hong Kong, Hong Kong, China, Chinese University of 4 Federal University of Minas Hong Kong (CUHK) Shenzhen Research Institute, Shenzhen, China, School of Mathematics and Statistics, Xi’an Jiaotong 5 Gerais, Brazil University, Xi’an, China, Laboratory for Industrial and Applied Mathematics, Department of Mathematics and Statistics, York University, Toronto, ON, Canada, 6 School of Public Health, Li Ka Shing Faculty of Medicine, University of Hong Kong, Reviewed by: Hong Kong, China, 7 Department of Applied Mathematics, Hong Kong Polytechnic University, Hong Kong, China Mohammad Javanbakht, Baqiyatallah University of Medical Sciences, Iran Background: The asymptomatic proportion is a critical epidemiological characteristic Changjing Zhuge, that modulates the pandemic potential of emerging respiratory virus, which may vary Beijing University of Technology, China *Correspondence: depending on the nature of the disease source, population characteristics, source–host Daihai He interaction, and environmental factors. [email protected] Xiujun Li Methods: We developed a simple likelihood-based framework to estimate the [email protected] instantaneous asymptomatic proportion of infectious diseases. Taking the COVID-19 Shi Zhao epidemics in Hong Kong as a case study, we applied the estimation framework [email protected] to estimate the reported asymptomatic proportion (rAP) using the publicly available † These authors share first authorship surveillance data. -

Vaccination Certificate Information Version: 21 July 2021
معلومات عن شهادة التطعيم، بالعربية 中文疫苗接种凭证信息 Informations sur le certificat de vaccination en FRANÇAIS Информация о сертификате вакцинации на РУССКОМ ЯЗЫКЕ Información sobre el Certificado de Vacunación en ESPAÑOL UN SYSTEM-WIDE COVID-19 VACCINATION PROGRAMME VACCINATION CERTIFICATE INFORMATION VERSION: 21 JULY 2021 ABOUT THE UN SYSTEM-WIDE COVID-19 VACCINATION PROGRAMME The UN is committed to ensuring the protection of its personnel. Tasked by the Secretary-General, the Department of Operational Support (DOS) is leading a coordinated, UN-system-wide effort to ensure the availability of vaccine to UN personnel, their dependents and implementing partners. The roll-out of the UN System-wide COVID-19 Vaccination Programme (the “Programme”) for UN personnel will provide a significant boost to the ability of personnel to stay and deliver on the Organization's mandates, to support beneficiaries in the communities they serve, and to contribute to our on-going work to recover better together from the pandemic. Information about the Programme is available at: https://www.un.org/en/coronavirus/vaccination ABOUT THE VACCINATION CERTIFICATE Upon administration of the requisite vaccine dosage, a certificate of vaccination is generated through the Programme’s registration platform (the “Platform”). The certificate generated by the Platform uses a standardized format, similar to the one used by the WHO in its “International Certificate of Vaccination or Prophylaxis” (Yellow) vaccination booklet. Note: This certificate may not dispense with travel restrictions put in place by the country(ies) of destination. As the nature of the pandemic continues to rapidly evolve, it is highly advisable to check the latest travel regulations. -

COVID-19 Scientific Advisory Group Rapid Response Report
COVID-19 Scientific Advisory Group Rapid Response Report Key Research Question: What is the evidence supporting the possibility of asymptomatic transmission of SARS-CoV-2? [Updated April 13, 2020] Context • Asymptomatic transmission (including pre-symptomatic transmission) of SARS-CoV-2 could reduce the effectiveness of control measures that are related to symptom onset (isolation, face masks and enhanced hygiene for symptomatic persons, and parameters of contact tracing), particularly if routine practices, including the use of diligent hand hygiene and environmental disinfection after patient encounters are not adhered to. • Concerns regarding asymptomatic transmission are driven by observations in various populations that suggested 18% – 50.5% of people with positive RT-PCR in various settings were asymptomatic at testing, and epidemiologic modelling suggesting that asymptomatic or presymptomatic cases may be responsible for potentially significant transmission, with a wide range of estimates from 0% to 44%. However, this is discrepant from epidemiologic descriptions from the initial epidemic in China and elsewhere which do not suggest that asymptomatic transmission is a major driver for the COVID19 epidemic. • The Public Health Agency of Canada (PHAC) has indicated that wearing a non-medical (cloth) mask in the community has not been proven to protect the person wearing it, however, it can be an additional measure to protect others around you, and might be useful in situations where physical distancing is not possible (e.g., grocery stores and public transit). (Tasker, 2020; Public Health Agency of Canada, 2020) Key Messages from the Evidence Summary • It is biologically plausible that SARS-CoV-2 can be transmitted when patients are asymptomatic, pre-symptomatic, or mildly symptomatic (potentially from 2.5 days prior to onset of symptoms), based on the finding that RT-PCR levels are high early in infection. -

Zoonotic Diseases Fact Sheet
ZOONOTIC DISEASES FACT SHEET s e ion ecie s n t n p is ms n e e s tio s g s m to a a o u t Rang s p t tme to e th n s n m c a s a ra y a re ho Di P Ge Ho T S Incub F T P Brucella (B. Infected animals Skin or mucous membrane High and protracted (extended) fever. 1-15 weeks Most commonly Antibiotic melitensis, B. (swine, cattle, goats, contact with infected Infection affects bone, heart, reported U.S. combination: abortus, B. suis, B. sheep, dogs) animals, their blood, tissue, gallbladder, kidney, spleen, and laboratory-associated streptomycina, Brucellosis* Bacteria canis ) and other body fluids causes highly disseminated lesions bacterial infection in tetracycline, and and abscess man sulfonamides Salmonella (S. Domestic (dogs, cats, Direct contact as well as Mild gastroenteritiis (diarrhea) to high 6 hours to 3 Fatality rate of 5-10% Antibiotic cholera-suis, S. monkeys, rodents, indirect consumption fever, severe headache, and spleen days combination: enteriditis, S. labor-atory rodents, (eggs, food vehicles using enlargement. May lead to focal chloramphenicol, typhymurium, S. rep-tiles [especially eggs, etc.). Human to infection in any organ or tissue of the neomycin, ampicillin Salmonellosis Bacteria typhi) turtles], chickens and human transmission also body) fish) and herd animals possible (cattle, chickens, pigs) All Shigella species Captive non-human Oral-fecal route Ranges from asymptomatic carrier to Varies by Highly infective. Low Intravenous fluids primates severe bacillary dysentery with high species. 16 number of organisms and electrolytes, fevers, weakness, severe abdominal hours to 7 capable of causing Antibiotics: ampicillin, cramps, prostration, edema of the days. -

Possible Modes of Transmission of Novel Coronavirus SARS-Cov-2
Acta Biomed 2020; Vol. 91, N. 3: e2020036 DOI: 10.23750/abm.v91i3.10039 © Mattioli 1885 Reviews / Focus on Possible modes of transmission of Novel Coronavirus SARS-CoV-2: a review Richa Mukhra1, Kewal Krishan1, Tanuj Kanchan2 1 Department of Anthropology (UGC Centre of Advanced Study), Panjab University, Sector-14, Chandigarh, India 2 Department of Forensic Medicine and Toxicology, All India Institute of Medical Sciences, Jodhpur, India. Abstract: Introduction: The widespread outbreak of the novel SARS-CoV-2 has raised numerous questions about the origin and transmission of the virus. Knowledge about the mode of transmission as well as assess- ing the effectiveness of the preventive measures would aid in containing the outbreak of the coronavirus. Presently, respiratory droplets, physical contact and aerosols/air-borne have been reported as the modes of SARS-CoV-2 transmission of the virus. Besides, some of the other possible modes of transmission are being explored by the researchers, with some studies suggesting the viral spread through fecal-oral, conjunctival secretions, flatulence (farts), sexual and vertical transmission from mother to the fetus, and through asymp- tomatic carriers, etc. Aim: The primary objective was to review the present understanding and knowledge about the transmission of SARS-CoV-2 and also to suggest recommendations in containing and preventing the novel coronavirus. Methods: A review of possible modes of transmission of the novel SARS-CoV-2 was conducted based on the reports and articles available in PubMed and ScienceDirect.com that were searched using keywords, ‘transmission’, ‘modes of transmission’, ‘SARS-CoV-2’, ‘novel coronavirus’, and ‘COVID-19’. Articles refer- ring to air-borne, conjunctiva, fecal-oral, maternal-fetal, flatulence (farts), and breast milk transmission were included, while the remaining were excluded. -

Epidemiology Annual Report
2018 Epidemiology Annual Report 535 South Loop 288 Denton, TX 76205-4502 www.DentonCounty.gov/Health Denton County Public Health Director Matt Richardson, DrPH, MPH Epidemiology Division Chief Epidemiologist Juan Rodriguez, MPH Epidemiologist Lily Metzler, MPH Epidemiologist Taylor Keplin, MPH Epidemiologist Abby Hoffman, M.S. Acknowledgments This report was prepared by the Epidemiology Division staff. We wish to acknowledge the contribution of our health care providers for reporting notifiable conditions and their cooperation in diseases investigations. In addition, we wish to thank Denton County Public Health staff and Medical Reserve Corp for their assistance in disease surveillance and investigations along with our colleagues at the Texas Department of State Health Services for providing data used in this report. Methodology The total population estimates used in this report were collected from the Texas Demographic Center1 and represent the population estimates for Denton County as of July 1, 2018. Population estimates may vary from previous Epidemiology Annual Reports due to changes of these estimates and are not directly associated to population estimates produced by the U.S. Census Bureau. The Sexually Transmitted Disease (STD) and Human Immunodeficiency Virus (HIV) case numbers and rates for Denton County were obtained from the Texas Department of State Health Services STD Surveillance Program Dashboard2. STD and HIV rates were calculated by the Texas Department of State Health Services based on population projections from the U.S. Census Bureau3. Data for Denton County notifiable conditions was collected from the Texas Department of State Health Services National Electronic Disease Surveillance System (NEDSS)4. This system delivers public health data to both state and local agencies, allowing data sharing and the ability to conduct disease surveillance. -

Vaccine Hesitancy
WHY CHILDREN WORKSHOP ON IMMUNIZATIONS ARE NOT VACCINATED? VACCINE HESITANCY José Esparza MD, PhD - Adjunct Professor, Institute of Human Virology, University of Maryland School of Medicine, Baltimore, MD, USA - Robert Koch Fellow, Robert Koch Institute, Berlin, Germany - Senior Advisor, Global Virus Network, Baltimore, MD, USA. Formerly: - Bill & Melinda Gates Foundation, Seattle, WA, USA - World Health Organization, Geneva, Switzerland The value of vaccination “The impact of vaccination on the health of the world’s people is hard to exaggerate. With the exception of safe water, no other modality has had such a major effect on mortality reduction and population growth” Stanley Plotkin (2013) VACCINES VAILABLE TO PROTECT AGAINST MORE DISEASES (US) BASIC VACCINES RECOMMENDED BY WHO For all: BCG, hepatitis B, polio, DTP, Hib, Pneumococcal (conjugated), rotavirus, measles, rubella, HPV. For certain regions: Japanese encephalitis, yellow fever, tick-borne encephalitis. For some high-risk populations: typhoid, cholera, meningococcal, hepatitis A, rabies. For certain immunization programs: mumps, influenza Vaccines save millions of lives annually, worldwide WHAT THE WORLD HAS ACHIEVED: 40 YEARS OF INCREASING REACH OF BASIC VACCINES “Bill Gates Chart” 17 M GAVI 5.6 M 4.2 M Today (ca 2015): <5% of children in GAVI countries fully immunised with the 11 WHO- recommended vaccines Seth Berkley (GAVI) The goal: 50% of children in GAVI countries fully immunised by 2020 Seth Berkley (GAVI) The current world immunization efforts are achieving: • Equity between high and low-income countries • Bringing the power of vaccines to even the world’s poorest countries • Reducing morbidity and mortality in developing countries • Eliminating and eradicating disease WHY CHILDREN ARE NOT VACCINATED? •Vaccines are not available •Deficient health care systems •Poverty •Vaccine hesitancy (reticencia a la vacunacion) VACCINE HESITANCE: WHO DEFINITION “Vaccine hesitancy refers to delay in acceptance or refusal of vaccines despite availability of vaccination services. -
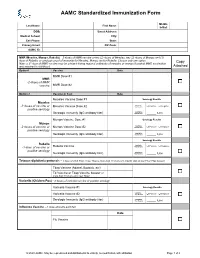
AAMC Standardized Immunization Form
AAMC Standardized Immunization Form Middle Last Name: First Name: Initial: DOB: Street Address: Medical School: City: Cell Phone: State: Primary Email: ZIP Code: AAMC ID: MMR (Measles, Mumps, Rubella) – 2 doses of MMR vaccine or two (2) doses of Measles, two (2) doses of Mumps and (1) dose of Rubella; or serologic proof of immunity for Measles, Mumps and/or Rubella. Choose only one option. Copy Note: a 3rd dose of MMR vaccine may be advised during regional outbreaks of measles or mumps if original MMR vaccination was received in childhood. Attached Option1 Vaccine Date MMR Dose #1 MMR -2 doses of MMR vaccine MMR Dose #2 Option 2 Vaccine or Test Date Measles Vaccine Dose #1 Serology Results Measles Qualitative -2 doses of vaccine or Measles Vaccine Dose #2 Titer Results: Positive Negative positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Mumps Vaccine Dose #1 Serology Results Mumps Qualitative -2 doses of vaccine or Mumps Vaccine Dose #2 Titer Results: Positive Negative positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Serology Results Rubella Qualitative Positive Negative -1 dose of vaccine or Rubella Vaccine Titer Results: positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Tetanus-diphtheria-pertussis – 1 dose of adult Tdap; if last Tdap is more than 10 years old, provide date of last Td or Tdap booster Tdap Vaccine (Adacel, Boostrix, etc) Td Vaccine or Tdap Vaccine booster (if more than 10 years since last Tdap) Varicella (Chicken Pox) - 2 doses of varicella vaccine or positive serology Varicella Vaccine #1 Serology Results Qualitative Varicella Vaccine #2 Titer Results: Positive Negative Serologic Immunity (IgG antibody titer) Quantitative Titer Results: _____ IU/ml Influenza Vaccine --1 dose annually each fall Date Flu Vaccine © 2020 AAMC. -

Statements Contained in This Release As the Result of New Information Or Future Events Or Developments
Pfizer and BioNTech Provide Update on Booster Program in Light of the Delta-Variant NEW YORK and MAINZ, GERMANY, July 8, 2021 — As part of Pfizer’s and BioNTech’s continued efforts to stay ahead of the virus causing COVID-19 and circulating mutations, the companies are providing an update on their comprehensive booster strategy. Pfizer and BioNTech have seen encouraging data in the ongoing booster trial of a third dose of the current BNT162b2 vaccine. Initial data from the study demonstrate that a booster dose given 6 months after the second dose has a consistent tolerability profile while eliciting high neutralization titers against the wild type and the Beta variant, which are 5 to 10 times higher than after two primary doses. The companies expect to publish more definitive data soon as well as in a peer-reviewed journal and plan to submit the data to the FDA, EMA and other regulatory authorities in the coming weeks. In addition, data from a recent Nature paper demonstrate that immune sera obtained shortly after dose 2 of the primary two dose series of BNT162b2 have strong neutralization titers against the Delta variant (B.1.617.2 lineage) in laboratory tests. The companies anticipate that a third dose will boost those antibody titers even higher, similar to how the third dose performs for the Beta variant (B.1.351). Pfizer and BioNTech are conducting preclinical and clinical tests to confirm this hypothesis. While Pfizer and BioNTech believe a third dose of BNT162b2 has the potential to preserve the highest levels of protective efficacy against all currently known variants including Delta, the companies are remaining vigilant and are developing an updated version of the Pfizer-BioNTech COVID-19 vaccine that targets the full spike protein of the Delta variant. -

COVID-19 Vaccines Frequently Asked Questions
Page 1 of 12 COVID-19 Vaccines 2020a Frequently Asked Questions Michigan.gov/Coronavirus The information in this document will change frequently as we learn more about COVID-19 vaccines. There is a lot we are learning as the pandemic and COVID-19 vaccines evolve. The approach in Michigan will adapt as we learn more. September 29, 2021. Quick Links What’s new | Why COVID-19 vaccination is important | Booster and additional doses | What to expect when you get vaccinated | Safety of the vaccine | Vaccine distribution/prioritization | Additional vaccine information | Protecting your privacy | Where can I get more information? What’s new − Pfizer booster doses recommended for some people to boost waning immunity six months after completing the Pfizer vaccine. Why COVID-19 vaccination is important − If you are fully vaccinated, you don’t have to quarantine after being exposed to COVID-19, as long as you don’t have symptoms. This means missing less work, school, sports and other activities. − COVID-19 vaccination is the safest way to build protection. COVID-19 is still a threat, especially to people who are unvaccinated. Some people who get COVID-19 can become severely ill, which could result in hospitalization, and some people have ongoing health problems several weeks or even longer after getting infected. Even people who did not have symptoms when they were infected can have these ongoing health problems. − After you are fully vaccinated for COVID-19, you can resume many activities that you did before the pandemic. CDC recommends that fully vaccinated people wear a mask in public indoor settings if they are in an area of substantial or high transmission. -

Recommended Adult Immunization Schedule
Recommended Adult Immunization Schedule UNITED STATES for ages 19 years or older 2021 Recommended by the Advisory Committee on Immunization Practices How to use the adult immunization schedule (www.cdc.gov/vaccines/acip) and approved by the Centers for Disease Determine recommended Assess need for additional Review vaccine types, Control and Prevention (www.cdc.gov), American College of Physicians 1 vaccinations by age 2 recommended vaccinations 3 frequencies, and intervals (www.acponline.org), American Academy of Family Physicians (www.aafp. (Table 1) by medical condition and and considerations for org), American College of Obstetricians and Gynecologists (www.acog.org), other indications (Table 2) special situations (Notes) American College of Nurse-Midwives (www.midwife.org), and American Academy of Physician Assistants (www.aapa.org). Vaccines in the Adult Immunization Schedule* Report y Vaccines Abbreviations Trade names Suspected cases of reportable vaccine-preventable diseases or outbreaks to the local or state health department Haemophilus influenzae type b vaccine Hib ActHIB® y Clinically significant postvaccination reactions to the Vaccine Adverse Event Hiberix® Reporting System at www.vaers.hhs.gov or 800-822-7967 PedvaxHIB® Hepatitis A vaccine HepA Havrix® Injury claims Vaqta® All vaccines included in the adult immunization schedule except pneumococcal 23-valent polysaccharide (PPSV23) and zoster (RZV) vaccines are covered by the Hepatitis A and hepatitis B vaccine HepA-HepB Twinrix® Vaccine Injury Compensation Program. Information on how to file a vaccine injury Hepatitis B vaccine HepB Engerix-B® claim is available at www.hrsa.gov/vaccinecompensation. Recombivax HB® Heplisav-B® Questions or comments Contact www.cdc.gov/cdc-info or 800-CDC-INFO (800-232-4636), in English or Human papillomavirus vaccine HPV Gardasil 9® Spanish, 8 a.m.–8 p.m.