Serbian Journal of Experimental and Clinical Research Vol12
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Effect of Selected Herbal Extracts on Lactic Acid Bacteria Activity
applied sciences Article The Effect of Selected Herbal Extracts on Lactic Acid Bacteria Activity Małgorzata Ziarno 1,* , Mariola Kozłowska 2 , Iwona Scibisz´ 3 , Mariusz Kowalczyk 4 , Sylwia Pawelec 4 , Anna Stochmal 4 and Bartłomiej Szleszy ´nski 5 1 Division of Milk Technology, Department of Food Technology and Assessment, Institute of Food Science, Warsaw University of Life Sciences–SGGW (WULS–SGGW), 02-787 Warsaw, Poland 2 Department of Chemistry, Institute of Food Science, Warsaw University of Life Sciences–SGGW (WULS–SGGW), 02-787 Warsaw, Poland; [email protected] 3 Division of Fruit, Vegetable and Cereal Technology, Department of Food Technology and Assessment, Institute of Food Science, Warsaw University of Life Sciences–SGGW (WULS–SGGW), 02-787 Warsaw, Poland; [email protected] 4 Department of Biochemistry and Crop Quality, Institute of Soil Science and Plant Cultivation, State Research Institute, 24-100 Puławy, Poland; [email protected] (M.K.); [email protected] (S.P.); [email protected] (A.S.) 5 Institute of Horticultural Sciences, Warsaw University of Life Sciences–SGGW (WULS–SGGW), 02-787 Warsaw, Poland; [email protected] * Correspondence: [email protected]; Tel.: +48-225-937-666 Abstract: This study aimed to investigate the effect of plant extracts (valerian Valeriana officinalis L., sage Salvia officinalis L., chamomile Matricaria chamomilla L., cistus Cistus L., linden blossom Tilia L., ribwort plantain Plantago lanceolata L., marshmallow Althaea L.) on the activity and growth of lactic acid bacteria (LAB) during the fermentation and passage of milk through a digestive system model. Citation: Ziarno, M.; Kozłowska, M.; The tested extracts were also characterized in terms of their content of polyphenolic compounds and Scibisz,´ I.; Kowalczyk, M.; Pawelec, S.; antioxidant activity. -

The Benefits of Flavonoids in Diabetic Retinopathy
nutrients Review The Benefits of Flavonoids in Diabetic Retinopathy 1, 1, 2,3,4,5 1,2,3,4, Ana L. Matos y, Diogo F. Bruno y, António F. Ambrósio and Paulo F. Santos * 1 Department of Life Sciences, University of Coimbra, Calçada Martim de Freitas, 3000-456 Coimbra, Portugal; [email protected] (A.L.M.); [email protected] (D.F.B.) 2 Coimbra Institute for Clinical and Biomedical Research (iCBR), Faculty of Medicine, University of Coimbra, 3000-548 Coimbra, Portugal; [email protected] 3 Center for Innovative Biomedicine and Biotechnology (CIBB), University of Coimbra, 3000-548 Coimbra, Portugal 4 Clinical Academic Center of Coimbra (CACC), 3004-561 Coimbra, Portugal 5 Association for Innovation and Biomedical Research on Light and Image (AIBILI), 3000-548 Coimbra, Portugal * Correspondence: [email protected]; Tel.: +351-239-240-762 These authors contributed equally to the work. y Received: 10 September 2020; Accepted: 13 October 2020; Published: 16 October 2020 Abstract: Diabetic retinopathy (DR), one of the most common complications of diabetes, is the leading cause of legal blindness among adults of working age in developed countries. After 20 years of diabetes, almost all patients suffering from type I diabetes mellitus and about 60% of type II diabetics have DR. Several studies have tried to identify drugs and therapies to treat DR though little attention has been given to flavonoids, one type of polyphenols, which can be found in high levels mainly in fruits and vegetables, but also in other foods such as grains, cocoa, green tea or even in red wine. -

Rosemary)-Derived Ingredients As Used in Cosmetics
Safety Assessment of Rosmarinus Officinalis (Rosemary)-Derived Ingredients as Used in Cosmetics Status: Tentative Amended Report for Public Comment Release Date: March 28, 2014 Panel Meeting Date: June 9-10, 2014 All interested persons are provided 60 days from the above release date to comment on this safety assessment and to identify additional published data that should be included or provide unpublished data which can be made public and included. Information may be submitted without identifying the source or the trade name of the cosmetic product containing the ingredient. All unpublished data submitted to CIR will be discussed in open meetings, will be available at the CIR office for review by any interested party and may be cited in a peer-reviewed scientific journal. Please submit data, comments, or requests to the CIR Director, Dr. Lillian J. Gill. The 2014 Cosmetic Ingredient Review Expert Panel members are: Chairman, Wilma F. Bergfeld, M.D., F.A.C.P.; Donald V. Belsito, M.D.; Ronald A. Hill, Ph.D.; Curtis D. Klaassen, Ph.D.; Daniel C. Liebler, Ph.D.; James G. Marks, Jr., M.D.; Ronald C. Shank, Ph.D.; Thomas J. Slaga, Ph.D.; and Paul W. Snyder, D.V.M., Ph.D. The CIR Director is Lillian J. Gill, D.P.A. This safety assessment was prepared by Monice M. Fiume, Assistant Director/Senior Scientific Analyst. © Cosmetic Ingredient Review 1620 L Street, NW, Suite 1200♢ Washington, DC 20036 ♢ ph 202.331.0651 ♢ fax 202.331.0088 ♢ [email protected] TABLE OF CONTENTS Abstract ...................................................................................................................................................................................................................................... -

Supplementary Materials Evodiamine Inhibits Both Stem Cell and Non-Stem
Supplementary materials Evodiamine inhibits both stem cell and non-stem-cell populations in human cancer cells by targeting heat shock protein 70 Seung Yeob Hyun, Huong Thuy Le, Hye-Young Min, Honglan Pei, Yijae Lim, Injae Song, Yen T. K. Nguyen, Suckchang Hong, Byung Woo Han, Ho-Young Lee - 1 - Table S1. Short tandem repeat (STR) DNA profiles for human cancer cell lines used in this study. MDA-MB-231 Marker H1299 H460 A549 HCT116 (MDA231) Amelogenin XX XY XY XX XX D8S1179 10, 13 12 13, 14 10, 14, 15 13 D21S11 32.2 30 29 29, 30 30, 33.2 D7S820 10 9, 12 8, 11 11, 12 8 CSF1PO 12 11, 12 10, 12 7, 10 12, 13 D3S1358 17 15, 18 16 12, 16, 17 16 TH01 6, 9.3 9.3 8, 9.3 8, 9 7, 9.3 D13S317 12 13 11 10, 12 13 D16S539 12, 13 9 11, 12 11, 13 12 D2S1338 23, 24 17, 25 24 16 21 D19S433 14 14 13 11, 12 11, 14 vWA 16, 18 17 14 17, 22 15 TPOX 8 8 8, 11 8, 9 8, 9 D18S51 16 13, 15 14, 17 15, 17 11, 16 D5S818 11 9, 10 11 10, 11 12 FGA 20 21, 23 23 18, 23 22, 23 - 2 - Table S2. Antibodies used in this study. Catalogue Target Vendor Clone Dilution ratio Application1) Number 1:1000 (WB) ADI-SPA- 1:50 (IHC) HSP70 Enzo C92F3A-5 WB, IHC, IF, IP 810-F 1:50 (IF) 1 :1000 (IP) ADI-SPA- HSP90 Enzo 9D2 1:1000 WB 840-F 1:1000 (WB) Oct4 Abcam ab19857 WB, IF 1:100 (IF) Nanog Cell Signaling 4903S D73G4 1:1000 WB Sox2 Abcam ab97959 1:1000 WB ADI-SRA- Hop Enzo DS14F5 1:1000 WB 1500-F HIF-1α BD 610958 54/HIF-1α 1:1000 WB pAkt (S473) Cell Signaling 4060S D9E 1:1000 WB Akt Cell Signaling 9272S 1:1000 WB pMEK Cell Signaling 9121S 1:1000 WB (S217/221) MEK Cell Signaling 9122S 1:1000 -

Sideritis Clandestina (Bory & Chaub.) Hayek; Sideritis Raeseri Boiss
2 February 2016 EMA/HMPC/39455/2015 Committee on Herbal Medicinal Products (HMPC) Assessment report on Sideritis scardica Griseb.; Sideritis clandestina (Bory & Chaub.) Hayek; Sideritis raeseri Boiss. & Heldr.; Sideritis syriaca L., herba Final Based on Article 16d(1), Article 16f and Article 16h of Directive 2001/83/EC as amended (traditional use) Herbal substances (binomial scientific name of Sideritis scardica Griseb.; Sideritis clandestina the plant, including plant part) (Bory & Chaub.) Hayek; Sideritis raeseri Boiss. & Heldr.; Sideritis syriaca L., herba Herbal preparation Comminuted herbal substance Pharmaceutical form Comminuted herbal substance as herbal tea for oral use Rapporteur I. Chinou Peer-reviewer B. Kroes 30 Churchill Place ● Canary Wharf ● London E14 5EU ● United Kingdom Telephone +44 (0)20 3660 6000 Facsimile +44 (0)20 3660 5555 Send a question via our website www.ema.europa.eu/contact An agency of the European Union © European Medicines Agency, 2016. Reproduction is authorised provided the source is acknowledged. Table of contents Table of contents ................................................................................................................... 2 1. Introduction ....................................................................................................................... 4 1.1. Description of the herbal substance(s), herbal preparation(s) or combinations thereof .. 4 1.2. Search and assessment methodology ..................................................................... 8 2. Data on -

Shilin Yang Doctor of Philosophy
PHYTOCHEMICAL STUDIES OF ARTEMISIA ANNUA L. THESIS Presented by SHILIN YANG For the Degree of DOCTOR OF PHILOSOPHY of the UNIVERSITY OF LONDON DEPARTMENT OF PHARMACOGNOSY THE SCHOOL OF PHARMACY THE UNIVERSITY OF LONDON BRUNSWICK SQUARE, LONDON WC1N 1AX ProQuest Number: U063742 All rights reserved INFORMATION TO ALL USERS The quality of this reproduction is dependent upon the quality of the copy submitted. In the unlikely event that the author did not send a com plete manuscript and there are missing pages, these will be noted. Also, if material had to be removed, a note will indicate the deletion. uest ProQuest U063742 Published by ProQuest LLC(2017). Copyright of the Dissertation is held by the Author. All rights reserved. This work is protected against unauthorized copying under Title 17, United States C ode Microform Edition © ProQuest LLC. ProQuest LLC. 789 East Eisenhower Parkway P.O. Box 1346 Ann Arbor, Ml 48106- 1346 ACKNOWLEDGEMENT I wish to express my sincere gratitude to Professor J.D. Phillipson and Dr. M.J.O’Neill for their supervision throughout the course of studies. I would especially like to thank Dr. M.F.Roberts for her great help. I like to thank Dr. K.C.S.C.Liu and B.C.Homeyer for their great help. My sincere thanks to Mrs.J.B.Hallsworth for her help. I am very grateful to the staff of the MS Spectroscopy Unit and NMR Unit of the School of Pharmacy, and the staff of the NMR Unit, King’s College, University of London, for running the MS and NMR spectra. -

Phenolic Profiling of Veronica Spp. Grown in Mountain, Urban and Sand Soil Environments
CORE Metadata, citation and similar papers at core.ac.uk Provided by Biblioteca Digital do IPB Phenolic profiling of Veronica spp. grown in mountain, urban and sand soil environments João C.M. Barreiraa,b,c, Maria Inês Diasa,c, Jelena Živkovićd, Dejan Stojkoviće, Marina Sokoviće, Celestino Santos-Buelgab,*, Isabel C.F.R. Ferreiraa,* aCIMO/Escola Superior Agrária, Instituto Politécnico de Bragança, Apartado 1172, 5301-855 Bragança, Portugal. bGIP-USAL, Facultad de Farmacia, Universidad de Salamanca, Campus Miguel de Unamuno, 37007 Salamanca, Spain. cREQUIMTE/Departamento de Ciências Químicas, Faculdade de Farmácia, Universidade do Porto, Rua Jorge Viterbo Ferreira, nº 228, 4050-313 Porto, Portugal. dInstitute for Medicinal Plant Research “Dr. Josif Pančić”, Tadeuša Košćuška 1, 11000 Belgrade, Serbia. eDepartment of Plant Physiology, Institute for Biological Research “Siniša Stanković”, University of Belgrade, Bulevar Despota Stefana 142, 11000 Belgrade, Serbia. * Authors to whom correspondence should be addressed (Isabel C.F.R. Ferreira; e-mail: [email protected], telephone +351273303219, fax +351273325405; e-mail: Celestino Santos- Buelga: [email protected]; telephone +34923294537; fax +34923294515). 1 Abstract Veronica (Plantaginaceae) genus is widely distributed in different habitats. Phytochemistry studies are increasing because most metabolites with pharmacological interest are obtained from plants. The phenolic compounds of V. montana, V. polita and V. spuria were tentatively identified by HPLC-DAD-ESI/MS. The phenolic profiles showed that flavones were the major compounds (V. montana: 7 phenolic acids, 5 flavones, 4 phenylethanoids and 1 isoflavone; V. polita: 10 flavones, 5 phenolic acids, 2 phenylethanoids, 1 flavonol and 1 isoflavone; V. spuria: 10 phenolic acids, 5 flavones, 2 flavonols, 2 phenylethanoids and 1 isoflavone), despite the overall predominance of flavones. -

Herbal Principles in Cosmetics Properties and Mechanisms of Action Traditional Herbal Medicines for Modern Times
Traditional Herbal Medicines for Modern Times Herbal Principles in Cosmetics Properties and Mechanisms of Action Traditional Herbal Medicines for Modern Times Each volume in this series provides academia, health sciences, and the herbal medicines industry with in-depth coverage of the herbal remedies for infectious diseases, certain medical conditions, or the plant medicines of a particular country. Series Editor: Dr. Roland Hardman Volume 1 Shengmai San, edited by Kam-Ming Ko Volume 2 Rasayana: Ayurvedic Herbs for Rejuvenation and Longevity, by H.S. Puri Volume 3 Sho-Saiko-To: (Xiao-Chai-Hu-Tang) Scientific Evaluation and Clinical Applications, by Yukio Ogihara and Masaki Aburada Volume 4 Traditional Medicinal Plants and Malaria, edited by Merlin Willcox, Gerard Bodeker, and Philippe Rasoanaivo Volume 5 Juzen-taiho-to (Shi-Quan-Da-Bu-Tang): Scientific Evaluation and Clinical Applications, edited by Haruki Yamada and Ikuo Saiki Volume 6 Traditional Medicines for Modern Times: Antidiabetic Plants, edited by Amala Soumyanath Volume 7 Bupleurum Species: Scientific Evaluation and Clinical Applications, edited by Sheng-Li Pan Traditional Herbal Medicines for Modern Times Herbal Principles in Cosmetics Properties and Mechanisms of Action Bruno Burlando, Luisella Verotta, Laura Cornara, and Elisa Bottini-Massa Cover art design by Carlo Del Vecchio. CRC Press Taylor & Francis Group 6000 Broken Sound Parkway NW, Suite 300 Boca Raton, FL 33487-2742 © 2010 by Taylor and Francis Group, LLC CRC Press is an imprint of Taylor & Francis Group, an Informa business No claim to original U.S. Government works Printed in the United States of America on acid-free paper 10 9 8 7 6 5 4 3 2 1 International Standard Book Number-13: 978-1-4398-1214-3 (Ebook-PDF) This book contains information obtained from authentic and highly regarded sources. -

Cushnie TPT, Lamb AJ. Antimicrobial Activity of Flavonoids. International Journal of Antimicrobial Agents, 2005. 26(5):343-356
Cushnie TPT, Lamb AJ. Antimicrobial activity of flavonoids. International Journal of Antimicrobial Agents, 2005. 26(5):343-356. PMID: 16323269 DOI: 10.1016/j.ijantimicag.2005.09.002 The journal article above is freely available from the publishers at: http://www.idpublications.com/journals/PDFs/IJAA/ANTAGE_MostCited_1.pdf and also... http://www.ijaaonline.com/article/S0924-8579(05)00255-4/fulltext Errata for the article (typesetting errors by Elsevier Ireland) are freely available from the publishers at: http://www.ijaaonline.com/article/S0924-8579(05)00352-3/fulltext and also... http://www.sciencedirect.com/science/article/pii/S0924857905003523 International Journal of Antimicrobial Agents 26 (2005) 343–356 Review Antimicrobial activity of flavonoids T.P. Tim Cushnie, Andrew J. Lamb ∗ School of Pharmacy, The Robert Gordon University, Schoolhill, Aberdeen AB10 1FR, UK Abstract Flavonoids are ubiquitous in photosynthesising cells and are commonly found in fruit, vegetables, nuts, seeds, stems, flowers, tea, wine, propolis and honey. For centuries, preparations containing these compounds as the principal physiologically active constituents have been used to treat human diseases. Increasingly, this class of natural products is becoming the subject of anti-infective research, and many groups have isolated and identified the structures of flavonoids possessing antifungal, antiviral and antibacterial activity. Moreover, several groups have demonstrated synergy between active flavonoids as well as between flavonoids and existing chemotherapeutics. Reports of activity in the field of antibacterial flavonoid research are widely conflicting, probably owing to inter- and intra-assay variation in susceptibility testing. However, several high-quality investigations have examined the relationship between flavonoid structure and antibacterial activity and these are in close agreement. -

Distribution of Flavonoids Among Malvaceae Family Members – a Review
Distribution of flavonoids among Malvaceae family members – A review Vellingiri Vadivel, Sridharan Sriram, Pemaiah Brindha Centre for Advanced Research in Indian System of Medicine (CARISM), SASTRA University, Thanjavur, Tamil Nadu, India Abstract Since ancient times, Malvaceae family plant members are distributed worldwide and have been used as a folk remedy for the treatment of skin diseases, as an antifertility agent, antiseptic, and carminative. Some compounds isolated from Malvaceae members such as flavonoids, phenolic acids, and polysaccharides are considered responsible for these activities. Although the flavonoid profiles of several Malvaceae family members are REVIEW REVIEW ARTICLE investigated, the information is scattered. To understand the chemical variability and chemotaxonomic relationship among Malvaceae family members summation of their phytochemical nature is essential. Hence, this review aims to summarize the distribution of flavonoids in species of genera namely Abelmoschus, Abroma, Abutilon, Bombax, Duboscia, Gossypium, Hibiscus, Helicteres, Herissantia, Kitaibelia, Lavatera, Malva, Pavonia, Sida, Theobroma, and Thespesia, Urena, In general, flavonols are represented by glycosides of quercetin, kaempferol, myricetin, herbacetin, gossypetin, and hibiscetin. However, flavonols and flavones with additional OH groups at the C-8 A ring and/or the C-5′ B ring positions are characteristic of this family, demonstrating chemotaxonomic significance. Key words: Flavones, flavonoids, flavonols, glycosides, Malvaceae, phytochemicals INTRODUCTION connate at least at their bases, but often forming a tube around the pistils. The pistils are composed of two to many connate he Malvaceae is a family of flowering carpels. The ovary is superior, with axial placentation, with plants estimated to contain 243 genera capitate or lobed stigma. The flowers have nectaries made with more than 4225 species. -
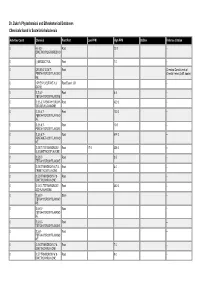
Dr. Duke's Phytochemical and Ethnobotanical Databases Chemicals Found in Scutellaria Baicalensis
Dr. Duke's Phytochemical and Ethnobotanical Databases Chemicals found in Scutellaria baicalensis Activities Count Chemical Plant Part Low PPM High PPM StdDev Refernce Citation 0 (+)-5,5'- Root 22.0 -- DIMETHOXYLARICIRESINO L 0 (-)-ERODICTYOL Root 7.0 -- 0 (2R,3R)-2',3,5,6',7- Root Chemical Constituents of PENTAHYDROXYFLAVONO Oriental Herbs (3 diff. books) NE 0 1-PHENYL-BUTANE-1,3- Root Essent. Oil -- DIONE 0 2',3',5,7- Root 6.4 -- TETRAHYDROXYFLAVONE 0 2',3,5,6',7-PENTAHYDROXY- Root 420.0 -- 2(R),3(R)-FLAVANONE 0 2',3,5,6',7- Root 120.0 -- PENTAHYDROXYFLAVANO NE 0 2',3,5,6',7- Root 10.0 -- PENTAHYDROXYFLAVONE 0 2',3,5,6',7- Root 674.0 -- PENTAMETHOXYFLAVANO NE 0 2',5,5',7-TETRAHYDROXY- Root 17.0 356.0 -- 6',8-DIMETHOXYFLAVONE 0 2',5,5',7- Root 3.5 -- TETRAHYDROXYFLAVONE 0 2',5,5'-TRIHYDROXY-6,7,8- Root 4.0 -- TRIMETHOXYFLAVONE 0 2',5,5'-TRIHYDROXY-7,8- Root -- DIMETHOXYFLAVONE 0 2',5,6',7-TETRAHYDROXY- Root 380.0 -- 2(S)-FLAVANONE 0 2',5,6',7- Stem -- TETRAHYDROXYFLAVANO NE 0 2',5,6',7- Root -- TETRAHYDROXYFLAVANO NE 0 2',5,6',7- Root -- TETRAHYDROXYFLAVONE 0 2',5,6'- Root -- TETRAHYDROXYFLAVANO NE 0 2',5,6'-TRIHYDROXY-7,8- Root 7.0 -- DIMETHOXYFLAVONE 0 2',5,7-TRIHYDROXY-6',8- Root 9.0 -- DIMETHOXYFLAVONE Activities Count Chemical Plant Part Low PPM High PPM StdDev Refernce Citation 0 2',5,7-TRIHYDROXY-6',8- Root 7.0 -- TRIMETHOXYFLAVONE 0 2',5,7-TRIHYDROXY-6'- Root 4.0 -- METHOXYFLAVONE 0 2',5,7-TRIHYDROXY-8- Root 6.0 -- METHOXYFLAVONE 0 2',5,7- Root 6.0 -- TRIHYDROXYFLAVONE 0 2',5,8-TRIHYDROXY-6,7- Root 200.0 -- DIMETHOXYFLAVONE 0 2'- Root -- METHYLSKULLCAPFLAVO NE 0 2(S),2',5,6',7- Root 230.0 -- TETRAHYDROXYFLAVANO NE 0 2,2',4',6-TETRAHYDROXY- Root 5.0 -- 6'-METHOXYCHALCONE 1 2,3-DIHYDROBENZOFURAN Root Essent. -

New Perspectives of Purple Starthistle (Centaurea Calcitrapa) Leaf Extracts
Dimkić et al. AMB Expr (2020) 10:183 https://doi.org/10.1186/s13568-020-01120-5 ORIGINAL ARTICLE Open Access New perspectives of purple starthistle (Centaurea calcitrapa) leaf extracts: phytochemical analysis, cytotoxicity and antimicrobial activity Ivica Dimkić1*† , Marija Petrović1†, Milan Gavrilović1, Uroš Gašić2, Petar Ristivojević3, Slaviša Stanković1 and Peđa Janaćković1 Abstract Ethnobotanical and ethnopharmacological studies of many Centaurea species indicated their potential in folk medi- cine so far. However, investigations of diferent Centaurea calcitrapa L. extracts in terms of cytotoxicity and antimi- crobial activity against phytopathogens are generally scarce. The phenolic profle and broad antimicrobial activity (especially towards bacterial phytopathogens) of methanol (MeOH), 70% ethanol (EtOH), ethyl-acetate (EtOAc), 50% acetone (Me2CO) and dichloromethane: methanol (DCM: MeOH, 1: 1) extracts of C. calcitrapa leaves and their poten- tial toxicity on MRC-5 cell line were investigated for the frst time. A total of 55 phenolic compounds were identifed: 30 phenolic acids and their derivatives, 25 favonoid glycosides and aglycones. This is also the frst report of the pres- ence of centaureidin, jaceidin, kaempferide, nepetin, favonoid glycosides, phenolic acids and their esters in C. calci- trapa extracts. The best results were obtained with EtOAc extract with lowest MIC values expressed in µg/mL ranging from 13 to 25, while methicillin resistant Staphylococcus aureus was the most susceptible strain. The most susceptible phytopathogens were Pseudomonas syringae pv. syringae, Xanthomonas campestris pv. campestris and Agrobacte- rium tumefaciens. The highest cytotoxicity was recorded for EtOAc and Me2CO extracts with the lowest relative and absolute IC50 values between 88 and 102 µg/mL, while EtOH extract was the least toxic with predicted relative IC 50 value of 1578 µg/mL.