01.MPG THESIS.Pdf
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Basic Histology and Connective Tissue Chapter 5
Basic Histology and Connective Tissue Chapter 5 • Histology, the Study of Tissues • Tissue Types • Connective Tissues Histology is the Study of Tissues • 200 different types of cells in the human body. • A Tissue consist of two or more types of cells that function together. • Four basic types of tissues: – epithelial tissue – connective tissue – muscular tissue – nervous tissue • An Organ is a structure with discrete boundaries that is composed of 2 or more tissue types. • Example: skin is an organ composed of epidermal tissue and dermal tissue. Distinguishing Features of Tissue Types • Types of cells (shapes and functions) • Arrangement of cells • Characteristics of the Extracellular Matrix: – proportion of water – types of fibrous proteins – composition of the ground substance • ground substance is the gelatinous material between cells in addition to the water and fibrous proteins • ground substance consistency may be liquid (plasma), rubbery (cartilage), stony (bone), elastic (tendon) • Amount of space occupied by cells versus extracellular matrix distinguishes connective tissue from other tissues – cells of connective tissues are widely separated by a large amount of extracellular matrix – very little extracellular matrix between the cells of epithelia, nerve, and muscle tissue Embryonic Tissues • An embryo begins as a single cell that divides into many cells that eventually forms 3 Primary Layers: – ectoderm (outer layer) • forms epidermis and nervous system – endoderm (inner layer) • forms digestive glands and the mucous membrane lining digestive tract and respiratory system – mesoderm (middle layer) • Forms muscle, bone, blood and other organs. Histotechnology • Preparation of specimens for histology: – preserve tissue in a fixative to prevent decay (formalin) – dehydrate in solvents like alcohol and xylene – embed in wax or plastic – slice into very thin sections only 1 or 2 cells thick – float slices on water and mount on slides and then add color with stains • Sectioning an organ or tissue reduces a 3-dimensional structure to a 2- dimensional slice. -

The Science of Aging Skin Leslie Baumann, MD; Susan Weinkle, MD
REVIEW Improving Elasticity: The Science of Aging Skin Leslie Baumann, MD; Susan Weinkle, MD Although collagen changes from both chronologic aging and photoaging have been well stud- ied, there is a dearth of research into the elastin component of the dermis. No products have been approved by the US Food and Drug Administration for either replacing elastin or stimulating its syn- thesis. Recently, a zinc complex was shown to increase dermal elastin staining, hypodermal fat, and epidermal thickness in animal studies. Histologic studies of the same zinc complex in humans have revealed increased deposition of functional elastin fibers following 10 days of use. In the clinical trials reported here, there was an increase in functional elastin, as well as improvements in fine lines and wrinkles. COSNo adverse events were noted. DERM ollagen and elastin are the fibers that make collagen bundles and endow the skin with rebounding up the dermal matrix. Cosmetic dermatol- properties. Tropoelastin molecules, which are precur- ogists have long used topical medications, sors to elastin, bind covalently with cross-links to form fillers, and light-based and laser-based elastin. Elastin fibers are assembled on bundles of micro- therapies to improve the loss of collagen fibrils composed of fibrillin. The latter forms a template Cthat occurs in aging skin. However, until recently, no fill- on which elastin is deposited.1 Most elastin production ers orDo topical agents have been availableNot to treat the loss is restricted Copy to a narrow window of development. Elas- of function of elastin, which is also an important feature togenesis increases dramatically during fetal life, peaks of dermal photoaging. -

Connective Tissue • Includes Things Like Bone, Fat, & Blood. All
Connective Tissue • includes things like bone, fat, & blood. All connective tissues include: 1. specialized cells 2.extracellular protein fibers } matrix that surrounds cells. 3. a fluid known as ground substance Functions include: Connective tissues come in 3 major types •Establish a structural framework 1. Connective tissue proper •Transporting fluids from one part of the body to another 2. Fluid Connective Tissue •Protecting delicate organs •Supporting, surrounding and interconnecting 3. Supporting Connective Tissue other tissue types • Other CTP cells are involved in defense and Connective Tissue Proper large repair jobs (these roam from site to site as • Connective tissue with many cell types and needed) extracellular fibers in a syrupy ground substance. A. Macrophages • Some cells of CTP are involved w/repair, B. Mast cells maintenance, and energy storage. C. Lymphocytes a. Fibroblasts D. plasma cells E. Microphages b. Adipocytes • The number of cells and cell types within a tissue at c. Mesenchymal cells any given moment varies depending on local conditions. 1 The Cell Population C. Adipocytes A. Fibroblasts • Fat cells • Most abundant cells in CTP • Typically contain a single enormous lipid droplet • Permanent resident of CTP (always present) • Other organelles squeezed to side of cell wall • Produce proteins to make the ground substance (resemble a class ring) very viscous • Also secret e prot ei ns th at mak e th e fib ers DMD. Mesenc hyma l ce lls • Stem cells B. Macrophages • Large amoeboid cells • Respond to injury by dividing into daughter cells which differentiate into connective tissue cells • Engulf & digest pathogens or damaged cells that enter the tissue • Release chemicals that activate the bodies immune system E. -

Connective Tissue N. Swailes, Ph.D. Department of Anatomy and Cell
Module 1.3: Connective Tissue N. Swailes, Ph.D. Department of Anatomy and Cell Biology Rm: B046A ML Tel: 5-7726 E-mail: [email protected] Required reading Mescher AL, Junqueira’s Basic Histology Text and Atlas, 13th Edition, Chapter 5 (also via AccessMedicine) Learning objectives 1) Name the three major classes of connective tissue and give examples of each. 2) Identify and describe the origin, organization and fate of embryonic connective tissue 3) Identify and discuss the functional properties imparted to tissue by the extracellular matrix: a. fibers (elastin, collagen Type I, II, III, IV and VII) b. ground substance (glycosaminoglycans, proteoglycans, glycoproteins) 4) Distinguish between different connective tissue cells and discuss their roles: a. fibroblasts b. adipocytes c. macrophages d. mast cells e. lymphocytes f. plasma cells g. eosinophils h. neutrophils 5) Classify the different connective tissues proper and compare and contrast their functional roles within an organ. Introduction The human body is made up of only four basic tissues: 1. Epithelial tissue 2. Connective tissue 3. Muscle tissue 4. Nervous tissue By adjusting the organization, composition and special features associated with each of these tissues is is possible to impart a wide variety of functions to the region or organ that they form. During this lecture you will examine the basic histological structure and function of Connective Tissue. 1 | Page: Connective Tissue Swailes a loose meshwork Part A: General characteristics of connective tissues that cushions and allows diffusion A1. There are three major classes of connective tissue i. Connective tissues proper - the most common class of connective tissue in the body. -

Flavio Akira Sakae Distribuição Das Fibras Colágenas E Do Sistema De
Flavio Akira Sakae Distribuição das fibras colágenas e do sistema de fibras elásticas na camada superficial da lâmina própria da prega vocal com edema de Reinke Tese apresentada à Faculdade de Medicina da Universidade de São Paulo para obtenção do título de Doutor em Ciências Área de concentração: Otorrinolaringologia Orientador: Prof. Dr. Domingos Hiroshi Tsuji São Paulo 2008 Dados Internacionais de Catalogação na Publicação (CIP) Preparada pela Biblioteca da Faculdade de Medicina da Universidade de São Paulo Óreprodução autorizada pelo autor Sakae, Flavio Akira Distribuição das fibras colágenas e do sistema de fibras elásticas na camada superficial da lâmina própria da prega vocal com edema de Reinke / Flavio Akira Sakae. -- São Paulo, 2008. Tese(doutorado)--Faculdade de Medicina da Universidade de São Paulo. Departamento de Oftalmologia e Otorrinolaringologia. Área de concentração: Otorrinolaringologia. Orientador: Domingos Hiroshi Tsuji. Descritores: 1.Edema laríngeo 2.Colágeno 3.Tecido elástico 4.Membrana mucosa 5.Cordas vocais USP/FM/SBD-146/08 "O único homem que está isento de erros, é aquele que não arrisca acertar." Albert Einstein Dedicatória Aos meus queridos pais, Masao e Junko, por tudo que fazem por mim, pelo apoio incondicional e amor eterno. São os meus ídolos. A minha esposa, Renata, amor da minha vida, pela alegria de viver, companheirismo e incentivo constante. A minha irmã, Cristiane, por ter contribuído em todos os passos de minha vida. Agradecimentos Ao meu orientador, Prof. Dr. Domingos Hiroshi Tsuji pela oportunidade e apoio na concretização deste sonho. Sua amizade e franqueza foram essenciais na elaboração deste trabalho. É o grande mestre. Ao Prof. Dr. -

Pg 131 Chondroblast -> Chondrocyte (Lacunae) Firm Ground Substance
Figure 4.8g Connective tissues. Chondroblast ‐> Chondrocyte (Lacunae) Firm ground substance (chondroitin sulfate and water) Collagenous and elastic fibers (g) Cartilage: hyaline No BV or nerves Description: Amorphous but firm Perichondrium (dense irregular) matrix; collagen fibers form an imperceptible network; chondroblasts produce the matrix and when mature (chondrocytes) lie in lacunae. Function: Supports and reinforces; has resilient cushioning properties; resists compressive stress. Location: Forms most of the embryonic skeleton; covers the ends Chondrocyte of long bones in joint cavities; forms in lacuna costal cartilages of the ribs; cartilages of the nose, trachea, and larynx. Matrix Costal Photomicrograph: Hyaline cartilage from the cartilages trachea (750x). Thickness? Metabolism? Copyright © 2010 Pearson Education, Inc. Pg 131 Figure 6.1 The bones and cartilages of the human skeleton. Epiglottis Support Thyroid Larynx Smooth Cartilage in Cartilages in cartilage external ear nose surface Cricoid Trachea Articular Lung Cushions cartilage Cartilage of a joint Cartilage in Costal Intervertebral cartilage disc Respiratory tube cartilages in neck and thorax Pubic Bones of skeleton symphysis Meniscus (padlike Axial skeleton cartilage in Appendicular skeleton knee joint) Cartilages Articular cartilage of a joint Hyaline cartilages Elastic cartilages Fibrocartilages Pg 174 Copyright © 2010 Pearson Education, Inc. Figure 4.8g Connective tissues. (g) Cartilage: hyaline Description: Amorphous but firm matrix; collagen fibers form an imperceptible network; chondroblasts produce the matrix and when mature (chondrocytes) lie in lacunae. Function: Supports and reinforces; has resilient cushioning properties; resists compressive stress. Location: Forms most of the embryonic skeleton; covers the ends Chondrocyte of long bones in joint cavities; forms in lacuna costal cartilages of the ribs; cartilages of the nose, trachea, and larynx. -

Differentiation of the Secondary Elastic Cartilage in the External Ear of the Rat
Int..I. Dc\'. Bi,,!. 35: 311-320 (1991) 311 Differentiation of the secondary elastic cartilage in the external ear of the rat ZELIMIR BRADAMANTE*', LJILJANA KOSTOVIC-KNEZEVIC', BOZICA LEVAK-SVAJGER' and ANTON SVAJGER' 'Institute of Histology and Embryology and 2/nstitute of Biology, Faculty of Medicine, University of Zagreb, Republic of Croatia, Yugoslavia ABSTRACT The cartilage in the external ear of the rat belongs to the group of secondary cartilages and it has a unique structural organization. The chondrocytes aretransformed intotypical adipose cells, the proteoglycan cartilage matrix is reduced to thin capsules around the cells and the rest of the extracelullar matrix is occupied by a network of coarse elastic fibers. It appears late in development 116-day fetus) and needs more than one month for final development. The differentiation proceeds in several steps which partly overlap: the appearance of collagen fibrils, elastin fibers, the proteoglycan matrix, and the adipose transformation of chondrocytes. The phenotype of this cartilage and the course of its differentiation are very stable, even in very atypical experimental environmental conditions. The only exceptions are explants in organ culture in vitro and perichondrial regenerates. In these conditions the development of elastic fibers is slow and poor while the production of the proteogycan matrix is abundant. The resulting cartilage then displays structural characteristics of hyaline cartilage rather than those of the initial elastic one. KEY WORDS: elastic mrlilagf, tI/(Wdrogfllt'.\'i.\. plasfogellf'.\is, extenuil Ntr, rat Introduction Structural organization The elastic cartilage belongs to the group of secondary or ac- According to Baecker (1928) and Schaffer (1930) the cartilage cessory cartilages since it is not a part of the cartilagineous in the external ear (external auditory meatus and pinna) of the rat primordium of the body skeleton as is most hyaline cartilage can be defined as: a) cellular or parenchymatous, [jecause of the (Schaffer, 1930). -

Mbios 401: Lecture 10.2 Extracellular Matrix Slide #1 Extracellular Matrix
MBios 401: Lecture 10.2 Extracellular Matrix Slide #1 Extracellular Matrix Shelden, Chapter 19, Basal Lamina. Audio: The extracellular matrix is the only topic in this course where we’ll be discussing something that’s fundamentally not a cell or part of a cell. Instead, the extracellular matrix, or ECM, is mostly made up of a material known as ground substance, along with a variety of types of imbedded protein fibers. Cells are the third and least abundant component of the extracellular matrix. However, since the extracellular matrix is made by cells and plays a significant role in determining the behavior of cells in it, we’ll discuss the cells of the extracellular matrix briefly. Every distinct tissue in an organism expresses characteristic extracellular matrix components, which gives the tissue its physical characteristics and also give the cells imbedded in it signals that they have to interpret correctly. As a consequence, the composition of the extracellular matrix is highly variable, and we’ll be looking at some of the molecular mechanisms that give rise to this variability. However, there are only two major types of extracellular matrix materials, which are shown in the cartoon image at the top of this slide. Most of this picture is filled with the first type, which includes the grey shaded material and the red and green imbedded fibers. This tissue, which is called connective tissue, forms most of the extracellular matrix material in our bodies. It has a variety of roles, including the formation of conduits for nerves and blood cells. It also provides supporting material for cellular organs, such as the liver and kidney and it performs a variety of mechanical roles in our bodies, including the formation of the skeletal system and things like tendons and ligaments. -

Connective Tissue 2
Tissue Physiology Connective tissue is one of the most abundant and widely distributed tissues in the body. It forms our bones, surrounds our organs, allows for the integrity of our neural and vascular system, cushions and lubricates our joints, and connects the muscles to our skeleton. Connective tissue serves multiple functions. It provides the organism with shape and mechanical support. Also, it modulates cell migration, growth, and differentiation. The following are major features of connective tissue: structural and mechanical, defense, nutrition and transport of molecules and storage. Connective tissue supports the shape of cells, tissues, and organs interacting with the cytoskeleton. The most obvious structural connective tissue is bone, comprising the skeleton and supporting the entire organism. It contains cells and metabolites important in immune function, such as inflammation, and in tissue repair after injury. Blood and blood vessels are connective tissue, which transports substances throughout the body. The nervous system is housed within connective tissue. Components in connective tissue regulate movement of nutrients between cells. Adipose tissue is a unique connective tissue, providing storage of energy and insulation. Connective tissue function is mediated by its different components, most of which are macromolecules that interact with one another and with the cells. Varying the proportions and the arrangement of the individual components determines the function of the particular tissue. Nutrient deficiencies may disrupt regulation of tissue synthesis and degradation. Connective tissue is metabolically active and serves many functions. The tissue consists of three basic components: fibers, ground substance, and cells. Outside the cell the fibers and ground substance form the extra cellular matrix. -

Chapter 2 Extracellular Matrix and Cardiac Remodeling
Chapter 2 Extracellular Matrix and Cardiac Remodeling Bodh I. Jugdutt University of Alberta‚ Edmonton‚ Alberta 1. Introduction Cumulative evidence over the last 3 decades suggests that the cardiac interstitium or extracellular matrix (ECM)‚ especially the extracellular collagen matrix (ECCM)‚ plays an important role in cardiac remodeling during various cardiac diseases including myocardial infarction (MI) and heart failure [1-4]. A unique feature of the healthy heart is the ability of the pumping chambers to return to the ideal shape at end-diastole despite repetitive changes during every cardiac cycle throughout life. This physiologic remodeling or property of resuming functional shape depends largely on the integrity of the ECCM‚ the intricate network of fibrillar collagens found in the ECM. In a broad sense‚ pathophysiologic cardiac remodeling refers to changes in cardiac structure‚ shape and function following stress or damage [5-7]. To remodel is defined in the Webster dictionary as “ to alter the structure‚ to remake” and in the Oxford dictionary as “to model again and differently‚ reconstruct and reorganize.” The Oxford dictionary defines “model” as “representation in three dimensions of the proposed structure” and “to model” as “to fashion and shape.” This review will focus on the role of the ECCM in pathologic cardiac remodeling and specifically left ventricular (LV) remodeling. 2. Cardiac extracellular matrix 2.1. Cardiac ECM‚ ECCM and fibroblasts In the heart‚ as in other organs [8‚ 9]‚ the specialized parenchymal cells (cardiomyocytes) are supported by an ECM made up of an intricate macromolecular network of fibers and different cell types of mesenchymal origin including fibroblasts‚ endothelial cells (cardiac and vascular)‚ smooth 24 muscle cells‚ blood-borne cells (macrophages and others)‚ pericytes and neurons‚ bathed in a gel-type ground substance (Table 1). -

Clinical Outcomes of Elastin Fibre Defects
ytology & f C H i o s l t a o n l o r g u y o Hassan et al., J Cytol Histol 2013, 4:1 J Journal of Cytology & Histology DOI: 10.4172/2157-7099.1000166 ISSN: 2157-7099 CommentaryResearch Article OpenOpen Access Access Clinical Outcomes of Elastin Fibre Defects Ashfaq Ul Hassan1*, Ghulam Hassan2, Zahida Rasool3 and Shabinul Hassan3 1Anatomy Department, SKIMS Medical College, Bemina, Srinagar, Kashmir, India 2Department of Anatomy, Al Qassim University, Saudi Arabia 3Islamic University of Science and Technology, Kashmir, India Abstract Elastic fibres are a major class of extracellular matrix fibres that are abundant in dynamic connective tissues such as arteries, lungs, skin and ligaments. Their structural role is to endow tissues with elastic recoil and resilience. They also act as an important adhesion template for cells, and they regulate growth factor availability. Mutations in major structural components of elastic fibres, especially elastin, fibrillins and fibulin-5, cause severe, often life-threatening, heritable connective tissue diseases such as Marfan syndrome, Menkes Syndrome, Cutis laxa. Elastic-fibre function is also frequently compromised in damaged or aged elastic tissues. The ability to regenerate or engineer elastic fibres and tissues remains a significant challenge, requiring improved understanding of the molecular and cellular basis of elastic-fibre biology and pathology. The aim of the article is to present in brief the structural details of elastin fibres, the defects and pleiotrophic effects as a result of damage and various clinical conditions associated with defects in elastin fibres ranging from rare pediatric syndromes to common entities like hypertension. -
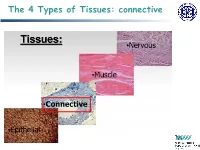
The 4 Types of Tissues: Connective
The 4 Types of Tissues: connective Connective Tissue General structure of CT cells are dispersed in a matrix matrix = a large amount of extracellular material produced by the CT cells and plays a major role in the functioning matrix component = ground substance often crisscrossed by protein fibers ground substance usually fluid, but it can also be mineralized and solid (bones) CTs = vast variety of forms, but typically 3 characteristic components: cells, large amounts of amorphous ground substance, and protein fibers. Connective Tissue GROUND SUBSTANCE In connective tissue, the ground substance is an amorphous gel-like substance surrounding the cells. In a tissue, cells are surrounded and supported by an extracellular matrix. Ground substance traditionally does not include fibers (collagen and elastic fibers), but does include all the other components of the extracellular matrix . The components of the ground substance vary depending on the tissue. Ground substance is primarily composed of water, glycosaminoglycans (most notably hyaluronan ), proteoglycans, and glycoproteins. Usually it is not visible on slides, because it is lost during the preparation process. Connective Tissue Functions of Connective Tissues Support and connect other tissues Protection (fibrous capsules and bones that protect delicate organs and, of course, the skeletal system). Transport of fluid, nutrients, waste, and chemical messengers is ensured by specialized fluid connective tissues, such as blood and lymph. Adipose cells store surplus energy in the form of fat and contribute to the thermal insulation of the body. Embryonic Connective Tissue All connective tissues derive from the mesodermal layer of the embryo . The first connective tissue to develop in the embryo is mesenchyme , the stem cell line from which all connective tissues are later derived.