Organization of the Catecholaminergic System in the Short-Lived Fish Nothobranchius Furzeri
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Hunting in Archerfish – an Ecological Perspective on a Remarkable Combination of Skills Stefan Schuster*
© 2018. Published by The Company of Biologists Ltd | Journal of Experimental Biology (2018) 221, jeb159723. doi:10.1242/jeb.159723 REVIEW Hunting in archerfish – an ecological perspective on a remarkable combination of skills Stefan Schuster* ABSTRACT and van Leeuwen, 2004) or mantids (Rossel, 1983)] at prey. Thus, Archerfish are well known for using jets of water to dislodge distant this Review may be of interest also to those studying non-spitting aerial prey from twigs or leaves. This Review gives a brief overview of a creatures. It should also be useful for researchers investigating number of skills that the fish need to secure prey with their shooting which properties of brains might be crucial for allowing some technique. Archerfish are opportunistic hunters and, even in the wild, species to use tools, imitate others or show insight into the tasks shoot at artificial objects to determine whether these are rewarding. demanded of them (e.g. Sol et al., 2005; Emery, 2006; Huber et al., They can detect non-moving targets and use efficient search strategies 2009; Morand-Ferron et al., 2016; Cauchoix et al., 2017; Powell with characteristics of human visual search. Their learning of how to et al., 2017; Street et al., 2017). Here, archerfish illustrate how engage targets can be remarkably efficient and can show impressive difficult it is to devise standardized tests that could compare learning ‘ ’ degrees of generalization, including learning from observation. In other and cognition across species. Such tests would require a detailed cases, however, the fish seem unable to learn and it requires some understanding of basic environmental constraints that can understanding of the ecological and biophysical constraints to sometimes be far from obvious. -

Present Status of Fish Biodiversity and Abundance in Shiba River, Bangladesh
Univ. J. zool. Rajshahi. Univ. Vol. 35, 2016, pp. 7-15 ISSN 1023-6104 http://journals.sfu.ca/bd/index.php/UJZRU © Rajshahi University Zoological Society Present status of fish biodiversity and abundance in Shiba river, Bangladesh D.A. Khanom, T Khatun, M.A.S. Jewel*, M.D. Hossain and M.M. Rahman Department of Fisheries, University of Rajshahi, Rajshahi 6205, Bangladesh Abstract: The study was conducted to investigate the abundance and present status of fish biodiversity in the Shiba river at Tanore Upazila of Rajshahi district, Bangladesh. The study was conducted from November, 2016 to February, 2017. A total of 30 species of fishes were recorded belonging to nine orders, 15 families and 26 genera. Cypriniformes and Siluriformes were the most diversified groups in terms of species. Among 30 species, nine species under the order Cypriniformes, nine species of Siluriformes, five species of Perciformes, two species of Channiformes, two species of Mastacembeliformes, one species of Beloniformes, one species of Clupeiformes, one species of Osteoglossiformes and one species of Decapoda, Crustacea were found. Machrobrachium lamarrei of the family Palaemonidae under Decapoda order was the most dominant species contributing 26.29% of the total catch. In the Shiba river only 6.65% threatened fish species were found, and among them 1.57% were endangered and 4.96% were vulnerable. The mean values of Shannon-Weaver diversity (H), Margalef’s richness (D) and Pielou’s (e) evenness were found as 1.86, 2.22 and 0.74, respectively. Relationship between Shannon-Weaver diversity index (H) and pollution indicates the river as light to moderate polluted. -

§4-71-6.5 LIST of CONDITIONALLY APPROVED ANIMALS November
§4-71-6.5 LIST OF CONDITIONALLY APPROVED ANIMALS November 28, 2006 SCIENTIFIC NAME COMMON NAME INVERTEBRATES PHYLUM Annelida CLASS Oligochaeta ORDER Plesiopora FAMILY Tubificidae Tubifex (all species in genus) worm, tubifex PHYLUM Arthropoda CLASS Crustacea ORDER Anostraca FAMILY Artemiidae Artemia (all species in genus) shrimp, brine ORDER Cladocera FAMILY Daphnidae Daphnia (all species in genus) flea, water ORDER Decapoda FAMILY Atelecyclidae Erimacrus isenbeckii crab, horsehair FAMILY Cancridae Cancer antennarius crab, California rock Cancer anthonyi crab, yellowstone Cancer borealis crab, Jonah Cancer magister crab, dungeness Cancer productus crab, rock (red) FAMILY Geryonidae Geryon affinis crab, golden FAMILY Lithodidae Paralithodes camtschatica crab, Alaskan king FAMILY Majidae Chionocetes bairdi crab, snow Chionocetes opilio crab, snow 1 CONDITIONAL ANIMAL LIST §4-71-6.5 SCIENTIFIC NAME COMMON NAME Chionocetes tanneri crab, snow FAMILY Nephropidae Homarus (all species in genus) lobster, true FAMILY Palaemonidae Macrobrachium lar shrimp, freshwater Macrobrachium rosenbergi prawn, giant long-legged FAMILY Palinuridae Jasus (all species in genus) crayfish, saltwater; lobster Panulirus argus lobster, Atlantic spiny Panulirus longipes femoristriga crayfish, saltwater Panulirus pencillatus lobster, spiny FAMILY Portunidae Callinectes sapidus crab, blue Scylla serrata crab, Samoan; serrate, swimming FAMILY Raninidae Ranina ranina crab, spanner; red frog, Hawaiian CLASS Insecta ORDER Coleoptera FAMILY Tenebrionidae Tenebrio molitor mealworm, -

Visual and Electrosensory Circuits of the Diencephalon in Mormyrids: an Evolutionary Perspective
THE JOURNAL OF COMPARATIVE NEUROLOGY 297:537-552 (1990) Visual and Electrosensory Circuits of the Diencephalon in Mormyrids: An Evolutionary Perspective MARIO F. WULLIMANN AND R. GLENN NORTHCUTT Georg-August-Universitat,Zentrum Anatomie, 3400 Gottingen, Federal Republic of Germany (M.F.W.); University of California, San Diego, and Department of Neurosciences A-001, Scripps Institution of Oceanography, La Jolla, California 92093 (R.G.N.) ABSTRACT Mormyrids are one of two groups of teleost fishes known to have evolved electroreception, and the concomitant neuroanatomical changes have confounded the interpretation of many of their brain areas in a comparative context, e.g., the diencephalon, where different sensory systems are processed and relayed. Recently, cerebellar and retinal connections of the diencephalon in mormyrids were reported. The present study reports on the telencephalic and tectal connections, specifically in Gnathonemus petersii, as these data are critical for an accurate interpretation of diencephalic nuclei in teleosts. Injections of horseradish peroxidase into the telencephalon retrogradely labeled neurons ipsilaterally in various thalamic, preglomer- ular, and tuberal nuclei, the nucleus of the locus coeruleus (also contralaterally), the superior raphe, and portions of the nucleus lateralis valvulae. Telencephalic injections anterogradely labeled the dorsal preglomerular and the dorsal tegmental nuclei bilaterally. Injections into the optic tectum retrogradely labeled neurons bilaterally in the central zone of area dorsalis telencephali and ipsilaterally in the torus longitudinalis, various thalamic, pretectal, and tegmental nuclei, some nuclei in the torus semicircularis, the nucleus of the locus coeruleus, the nucleus isthmi and the superior reticular formation, basal cells in the ipsilateral valvula cerebelli, and eurydendroid cells in the contralateral lobe C4 of the corpus cerebelli. -

Deterministic Shifts in Molecular Evolution Correlate with Convergence to Annualism in Killifishes
bioRxiv preprint doi: https://doi.org/10.1101/2021.08.09.455723; this version posted August 10, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Deterministic shifts in molecular evolution correlate with convergence to annualism in killifishes Andrew W. Thompson1,2, Amanda C. Black3, Yu Huang4,5,6 Qiong Shi4,5 Andrew I. Furness7, Ingo, Braasch1,2, Federico G. Hoffmann3, and Guillermo Ortí6 1Department of Integrative Biology, Michigan State University, East Lansing, Michigan 48823, USA. 2Ecology, Evolution & Behavior Program, Michigan State University, East Lansing, MI, USA. 3Department of Biochemistry, Molecular Biology, Entomology, & Plant Pathology, Mississippi State University, Starkville, MS 39759, USA. 4Shenzhen Key Lab of Marine Genomics, Guangdong Provincial Key Lab of Molecular Breeding in Marine Economic Animals, BGI Marine, Shenzhen 518083, China. 5BGI Education Center, University of Chinese Academy of Sciences, Shenzhen 518083, China. 6Department of Biological Sciences, The George Washington University, Washington, DC 20052, USA. 7Department of Biological and Marine Sciences, University of Hull, UK. Corresponding author: Andrew W. Thompson, [email protected] bioRxiv preprint doi: https://doi.org/10.1101/2021.08.09.455723; this version posted August 10, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Abstract: The repeated evolution of novel life histories correlating with ecological variables offer opportunities to test scenarios of convergence and determinism in genetic, developmental, and metabolic features. Here we leverage the diversity of aplocheiloid killifishes, a clade of teleost fishes that contains over 750 species on three continents. -

Marine Fish Conservation Global Evidence for the Effects of Selected Interventions
Marine Fish Conservation Global evidence for the effects of selected interventions Natasha Taylor, Leo J. Clarke, Khatija Alliji, Chris Barrett, Rosslyn McIntyre, Rebecca0 K. Smith & William J. Sutherland CONSERVATION EVIDENCE SERIES SYNOPSES Marine Fish Conservation Global evidence for the effects of selected interventions Natasha Taylor, Leo J. Clarke, Khatija Alliji, Chris Barrett, Rosslyn McIntyre, Rebecca K. Smith and William J. Sutherland Conservation Evidence Series Synopses 1 Copyright © 2021 William J. Sutherland This work is licensed under a Creative Commons Attribution 4.0 International license (CC BY 4.0). This license allows you to share, copy, distribute and transmit the work; to adapt the work and to make commercial use of the work providing attribution is made to the authors (but not in any way that suggests that they endorse you or your use of the work). Attribution should include the following information: Taylor, N., Clarke, L.J., Alliji, K., Barrett, C., McIntyre, R., Smith, R.K., and Sutherland, W.J. (2021) Marine Fish Conservation: Global Evidence for the Effects of Selected Interventions. Synopses of Conservation Evidence Series. University of Cambridge, Cambridge, UK. Further details about CC BY licenses are available at https://creativecommons.org/licenses/by/4.0/ Cover image: Circling fish in the waters of the Halmahera Sea (Pacific Ocean) off the Raja Ampat Islands, Indonesia, by Leslie Burkhalter. Digital material and resources associated with this synopsis are available at https://www.conservationevidence.com/ -

Nothobranchius As a Model for Aging Studies. a Review
Volume 5, Number 2; xxx-xx, April 2014 http://dx.doi.org/10.14336/AD Review Article Nothobranchius as a model for aging studies. A review Alejandro Lucas-Sánchez 1, *, Pedro Francisco Almaida-Pagán 2, Pilar Mendiola 1, Jorge de Costa 1 1 Department of Physiology. Faculty of Biology. University of Murcia. 30100 Murcia, Spain. 2 Institute of Aquaculture, School of Natural Sciences, University of Stirling, Stirling FK9 4LA, UK. [Received October 17, 2013; Revised December 4, 2013; Accepted December 4, 2013] ABSTRACT: In recent decades, the increase in human longevity has made it increasingly important to expand our knowledge on aging. To accomplish this, the use of animal models is essential, with the most common being mouse (phylogenetically similar to humans, and a model with a long life expectancy) and Caenorhabditis elegans (an invertebrate with a short life span, but quite removed from us in evolutionary terms). However, some sort of model is needed to bridge the differences between those mentioned above, achieving a balance between phylogenetic distance and life span. Fish of the genus Nothobranchius were suggested 10 years ago as a possible alternative for the study of the aging process. In the meantime, numerous studies have been conducted at different levels: behavioral (including the study of the rest-activity rhythm), populational, histochemical, biochemical and genetic, among others, with very positive results. This review compiles what we know about Nothobranchius to date, and examines its future prospects as a true alternative to the classic models for studies on aging. Key words: Aging, Fish, killifish, Nothobranchius Aging is a multifactorial process that involves numerous To be considered a model for aging, an animal must mechanisms that operate during the normal life cycle of have a number of features. -

Fishes of Terengganu East Coast of Malay Peninsula, Malaysia Ii Iii
i Fishes of Terengganu East coast of Malay Peninsula, Malaysia ii iii Edited by Mizuki Matsunuma, Hiroyuki Motomura, Keiichi Matsuura, Noor Azhar M. Shazili and Mohd Azmi Ambak Photographed by Masatoshi Meguro and Mizuki Matsunuma iv Copy Right © 2011 by the National Museum of Nature and Science, Universiti Malaysia Terengganu and Kagoshima University Museum All rights reserved. No part of this publication may be reproduced or transmitted in any form or by any means without prior written permission from the publisher. Copyrights of the specimen photographs are held by the Kagoshima Uni- versity Museum. For bibliographic purposes this book should be cited as follows: Matsunuma, M., H. Motomura, K. Matsuura, N. A. M. Shazili and M. A. Ambak (eds.). 2011 (Nov.). Fishes of Terengganu – east coast of Malay Peninsula, Malaysia. National Museum of Nature and Science, Universiti Malaysia Terengganu and Kagoshima University Museum, ix + 251 pages. ISBN 978-4-87803-036-9 Corresponding editor: Hiroyuki Motomura (e-mail: [email protected]) v Preface Tropical seas in Southeast Asian countries are well known for their rich fish diversity found in various environments such as beautiful coral reefs, mud flats, sandy beaches, mangroves, and estuaries around river mouths. The South China Sea is a major water body containing a large and diverse fish fauna. However, many areas of the South China Sea, particularly in Malaysia and Vietnam, have been poorly studied in terms of fish taxonomy and diversity. Local fish scientists and students have frequently faced difficulty when try- ing to identify fishes in their home countries. During the International Training Program of the Japan Society for Promotion of Science (ITP of JSPS), two graduate students of Kagoshima University, Mr. -

Sample Assessment Tasks
SAMPLE ASSESSMENT TASKS MARINE AND MARITIME STUDIES GENERAL YEAR 11 Copyright © School Curriculum and Standards Authority, 2014 This document – apart from any third party copyright material contained in it – may be freely copied, or communicated on an intranet, for non-commercial purposes in educational institutions, provided that the School Curriculum and Standards Authority is acknowledged as the copyright owner, and that the Authority’s moral rights are not infringed. Copying or communication for any other purpose can be done only within the terms of the Copyright Act 1968 or with prior written permission of the School Curriculum and Standards Authority. Copying or communication of any third party copyright material can be done only within the terms of the Copyright Act 1968 or with permission of the copyright owners. Any content in this document that has been derived from the Australian Curriculum may be used under the terms of the Creative Commons Attribution 4.0 International licence. Disclaimer Any resources such as texts, websites and so on that may be referred to in this document are provided as examples of resources that teachers can use to support their learning programs. Their inclusion does not imply that they are mandatory or that they are the only resources relevant to the course. 2014/18740v5 1 Sample assessment task Marine and Maritime Studies – General Year 11 Task 3 — Unit 1 Assessment type: Extended response Conditions Period allowed for completion of the task – three weeks; a combination of in-class and out-of-class time Task weighting: 2.5% of the school mark for this pair of units __________________________________________________________________________________ Marine resource management research (25 marks) Background Commercial fishing is an important contributor to the Western Australian economy. -
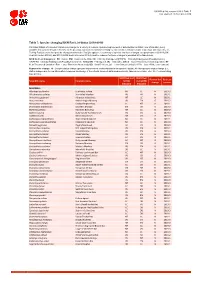
Table 7: Species Changing IUCN Red List Status (2018-2019)
IUCN Red List version 2019-3: Table 7 Last Updated: 10 December 2019 Table 7: Species changing IUCN Red List Status (2018-2019) Published listings of a species' status may change for a variety of reasons (genuine improvement or deterioration in status; new information being available that was not known at the time of the previous assessment; taxonomic changes; corrections to mistakes made in previous assessments, etc. To help Red List users interpret the changes between the Red List updates, a summary of species that have changed category between 2018 (IUCN Red List version 2018-2) and 2019 (IUCN Red List version 2019-3) and the reasons for these changes is provided in the table below. IUCN Red List Categories: EX - Extinct, EW - Extinct in the Wild, CR - Critically Endangered [CR(PE) - Critically Endangered (Possibly Extinct), CR(PEW) - Critically Endangered (Possibly Extinct in the Wild)], EN - Endangered, VU - Vulnerable, LR/cd - Lower Risk/conservation dependent, NT - Near Threatened (includes LR/nt - Lower Risk/near threatened), DD - Data Deficient, LC - Least Concern (includes LR/lc - Lower Risk, least concern). Reasons for change: G - Genuine status change (genuine improvement or deterioration in the species' status); N - Non-genuine status change (i.e., status changes due to new information, improved knowledge of the criteria, incorrect data used previously, taxonomic revision, etc.); E - Previous listing was an Error. IUCN Red List IUCN Red Reason for Red List Scientific name Common name (2018) List (2019) change version Category -

SPC Traditional Marine Resource Management and Knowledge
ISSN 1025-7497 Secretariat of the Pacific Community TRADITIONAL Marine Resource Management and Knowledge Number 18 — August 2005 INFORMATION BULLETIN Group Coordinator and Bulletin Editor: Kenneth Ruddle, Katsuragi 2-24-20, Kita-ku, Kobe-shi, Hyogo-ken 651-1223, Japan; Email: [email protected] — Production: Information Section, Marine Resources Division, SPC, BP D5, 98848 Noumea Cedex, New Caledonia. Fax: +687 263818; Email: [email protected]. The bulletin is also available at: http://www.spc.int/coastfish — Produced with financial assistance from France. Editor’s note We include three articles in this edition. In the first, “Fishing for drummerfish (Kyphosidae) with termites and spider webs Inside on the weather coast of Guadalcanal, Solomon Islands”, William T. Atu describes a unique traditional fishing method this issue known as bulukochi, which was used by his forefathers to catch drummerfish. This fishing method is on the verge of disap- pearing, and the only person who knows about it and the asso- ciated customs is Mr Atu’s elderly uncle. So Mr Atu decided to Fishing for drummerfish preserve some of this information here, because, as he says (Kyphosidae) with termites and “With the passing of my uncle the techniques and intricate spider webs on the weather coast customs associated with this method will be lost forever”. of Guadalcanal, Solomon Islands William T. Atu p. 3 William T. Atu has set a wonderful example. We hope it will stimulate other people to set about documenting “endangered Indigenous ecological knowledge information” in their own communities. This Information (IEK) of the aggregating and Bulletin would be delighted to publish such material. -

The Neotropical Genus Austrolebias: an Emerging Model of Annual Killifishes Nibia Berois1, Maria J
lopmen ve ta e l B D io & l l o l g e y C Cell & Developmental Biology Berois, et al., Cell Dev Biol 2014, 3:2 ISSN: 2168-9296 DOI: 10.4172/2168-9296.1000136 Review Article Open Access The Neotropical Genus Austrolebias: An Emerging Model of Annual Killifishes Nibia Berois1, Maria J. Arezo1 and Rafael O. de Sá2* 1Departamento de Biologia Celular y Molecular, Facultad de Ciencias, Universidad de la República, Montevideo, Uruguay 2Department of Biology, University of Richmond, Richmond, Virginia, USA *Corresponding author: Rafael O. de Sá, Department of Biology, University of Richmond, Richmond, Virginia, USA, Tel: 804-2898542; Fax: 804-289-8233; E-mail: [email protected] Rec date: Apr 17, 2014; Acc date: May 24, 2014; Pub date: May 27, 2014 Copyright: © 2014 Rafael O. de Sá, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Abstract Annual fishes are found in both Africa and South America occupying ephemeral ponds that dried seasonally. Neotropical annual fishes are members of the family Rivulidae that consist of both annual and non-annual fishes. Annual species are characterized by a prolonged embryonic development and a relatively short adult life. Males and females show striking sexual dimorphisms, complex courtship, and mating behaviors. The prolonged embryonic stage has several traits including embryos that are resistant to desiccation and undergo up to three reversible developmental arrests until hatching. These unique developmental adaptations are closely related to the annual fish life cycle and are the key to the survival of the species.