Chapter 12: Oxidation and Reduction
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Redox Models in Chemistry
Redox models in chemistry A depiction of the conceptions held by secondary school students of redox reactions Lise-Lotte Österlund Department of Chemistry 901 87 Umeå Umeå 2010 Copyright©Lise-Lotte Österlund ISBN: 978-91-7459-053-1 ISSN: 1652-5051 Cover picture: Photographer, Lise-Lotte Österlund Printed by: VMC, KBC, Umeå University Umeå, Sweden 2010 T0 my family Abstract According to previous research, students show difficulties in learning redox reactions. By the historical development different redox models exist to explain redox reactions, the oxygen model, the hydrogen model, the electron model and the oxidation number model. This thesis reports about three studies concerning conceptions held by secondary school students of redox reactions. A textbook analysis is also included in the thesis. The first study was an investigation of the students’ use of redox models in inorganic contexts, their use of the activity series of metals, and the students’ ability to transfer redox knowledge. Then the students’ work with an open- ended biochemical task, where the students had access of the textbook was studied. The students talk about redox reactions, the questions raised by the students, what resources used to answer the questions and what kind of talk developed were investigated. A textbook analysis based on chemistry books from Sweden and one book from England was performed. The redox models used as well as the dealing with redox related learning difficulties was studied. Finally, the students’ conceptions about redox in inorganic, organic and biochemistry after completed chemistry courses were studied. The results show that the students were able to use the electron model as a tool to explain inorganic redox reactions and the mutuality of oxidation and reduction was fundamental. -

Thèse En Co-Tutelle
UNIVERSITÉ DE REIMS CHAMPAGNE-ARDENNE ÉCOLE DOCTORALE SCIENCES FONDAMENTALES - SANTÉ N619 FACULTÉ DES SCIENCES DE TUNIS (FST) ÉCOLE DOCTORALE MATHÉMATIQUES, INFORMATIQUE, SCIENCES ET TECHNOLOGIES DES MATÉRIAUX THÈSE EN CO-TUTELLE Pour obtenir le grade de Docteur de l’Université de Reims Champagne-Ardenne Discipline : Physique Spécialité : Physique moléculaire ET Docteur de la Faculté des Sciences de Tunis Discipline : Physique Présentée et soutenue publiquement par Olfa FERCHICHI Le 28 Septembre 2020 Étude des propriétés structurales et spectroscopiques de peroxydes aux niveaux DFT et ab initio JURY Manef ABDERRABBA Professeur à l’Université de Carthage Rapporteur Jean Christophe TREMBLAY Professeur à l’Université de Lorraine Rapporteur et Président du Jury Halima MOUHIB Maître de conférences à l’Université Gustave EiUel Examinatrice Hassen GHALILA Professeur à l’Université de Tunis El Manar Examinateur Alexander ALIJAH Professeur à l’Université de Reims Directeur de thèse Najoua DERBEL Professeur à la Faculté des Sciences de Bizerte Directrice de thèse Thibaud COURS Maître de conférences à l’Université de Reims Membre invité Dédicaces Je dédie cette thèse : À mes très chers parents Mohammed et Latifa, Loin de vous, votre soutien et votre encouragement m'ont toujours donné de la force pour persévérer et pour prospérer dans la vie. À mes sœurs et mon frère, Je vous remercie énormément pour tous les efforts que vous avez fait pour moi et les soutiens moraux dont j’ai pu bénéficier. À mon cher mari Johan TARAPEY Merci pour tes encouragements, tu as toujours su trouver les mots qui conviennent pour me remonter la morale dans les moments pénibles, grâce à toi j’ai pu surmonter toutes les difficultés. -

Promotion Effects and Mechanism of Alkali Metals and Alkaline Earth
Subscriber access provided by RES CENTER OF ECO ENVIR SCI Article Promotion Effects and Mechanism of Alkali Metals and Alkaline Earth Metals on Cobalt#Cerium Composite Oxide Catalysts for N2O Decomposition Li Xue, Hong He, Chang Liu, Changbin Zhang, and Bo Zhang Environ. Sci. Technol., 2009, 43 (3), 890-895 • DOI: 10.1021/es801867y • Publication Date (Web): 05 January 2009 Downloaded from http://pubs.acs.org on January 31, 2009 More About This Article Additional resources and features associated with this article are available within the HTML version: • Supporting Information • Access to high resolution figures • Links to articles and content related to this article • Copyright permission to reproduce figures and/or text from this article Environmental Science & Technology is published by the American Chemical Society. 1155 Sixteenth Street N.W., Washington, DC 20036 Environ. Sci. Technol. 2009, 43, 890–895 Promotion Effects and Mechanism such as Fe-ZSM-5 are more active in the selective catalytic reduction (SCR) of N2O by hydrocarbons than in the - ° of Alkali Metals and Alkaline Earth decomposition of N2O in a temperature range of 300 400 C (3). In recent years, it has been found that various mixed Metals on Cobalt-Cerium oxide catalysts, such as calcined hydrotalcite and spinel oxide, showed relatively high activities. Composite Oxide Catalysts for N2O One of the most active oxide catalysts is a mixed oxide containing cobalt spinel. Calcined hydrotalcites containing Decomposition cobalt, such as Co-Al-HT (9-12) and Co-Rh-Al-HT (9, 11), have been reported to be very efficient for the decomposition 2+ LI XUE, HONG HE,* CHANG LIU, of N2O. -

UNITED STATES PATENT Office SVEND S
Patented May 24, 1932 1,859,998 UNITED STATES PATENT oFFICE SVEND S. SVENDSEN, OF CHICAGO, ILLINOIS, ASSIGNOR TO CLAY REDUCTION comi PANY, OF CHICAGO, ILLINOIS, A CORPORATION OF ILLINOIs . HYDRATED SILICA No Drawing. Application filed December 23, 1927. Serial No. 242,291. This invention relates to the production of silicofluoride is also volatilized and collected hydrated silica from a silicious material such in aqueous animonia. The volatilization oc as silica and silicates generally. - - - - curs about 300 C. The following reactions According to the invention the silicious take place in the aqueous ammonia at tem 5 material is treated with ammonium fluoride peratures below 34 C.: 5 5 or bifluoride and silicon ammonia fluorine compounds are produced. These compounds 2SiF(NH) +2H.O= are volatilized and converted into hydrated (NH4)2SiFs--SiO, (hydrated) + silica by the action of water and ammonia. 2NH.F. (IV) O Metallic silicates or substances containing (NH)SiF+4NH,+2HO = them can be subjected to this treatment di SiO, (hydrated) + 6NH.F. (V) rectly. In treating silica in the form of quartz it is found to be necessary to subject It is thus apparent that O heating the it to a preliminary treatment in order to fa silicious material with the ammonium-flu cilitate the action of the ammonium fluoride oride, ammonia-silicon-fluorine compounds a or bifluoride thereon. A suitable preliminary are formed, and by employing suitable tem treatment is to heat the quartz to a bright red peratures are volatilized from the reaction heat and suddenly cool it by immersion in mixture. These compounds may be silicon water. -

Prebiological Evolution and the Metabolic Origins of Life
Prebiological Evolution and the Andrew J. Pratt* Metabolic Origins of Life University of Canterbury Keywords Abiogenesis, origin of life, metabolism, hydrothermal, iron Abstract The chemoton model of cells posits three subsystems: metabolism, compartmentalization, and information. A specific model for the prebiological evolution of a reproducing system with rudimentary versions of these three interdependent subsystems is presented. This is based on the initial emergence and reproduction of autocatalytic networks in hydrothermal microcompartments containing iron sulfide. The driving force for life was catalysis of the dissipation of the intrinsic redox gradient of the planet. The codependence of life on iron and phosphate provides chemical constraints on the ordering of prebiological evolution. The initial protometabolism was based on positive feedback loops associated with in situ carbon fixation in which the initial protometabolites modified the catalytic capacity and mobility of metal-based catalysts, especially iron-sulfur centers. A number of selection mechanisms, including catalytic efficiency and specificity, hydrolytic stability, and selective solubilization, are proposed as key determinants for autocatalytic reproduction exploited in protometabolic evolution. This evolutionary process led from autocatalytic networks within preexisting compartments to discrete, reproducing, mobile vesicular protocells with the capacity to use soluble sugar phosphates and hence the opportunity to develop nucleic acids. Fidelity of information transfer in the reproduction of these increasingly complex autocatalytic networks is a key selection pressure in prebiological evolution that eventually leads to the selection of nucleic acids as a digital information subsystem and hence the emergence of fully functional chemotons capable of Darwinian evolution. 1 Introduction: Chemoton Subsystems and Evolutionary Pathways Living cells are autocatalytic entities that harness redox energy via the selective catalysis of biochemical transformations. -

Group 17 (Halogens)
Sodium, Na Gallium, Ga CHEMISTRY 1000 Topic #2: The Chemical Alphabet Fall 2020 Dr. Susan Findlay See Exercises 11.1 to 11.4 Forms of Carbon The Halogens (Group 17) What is a halogen? Any element in Group 17 (the only group containing Cl2 solids, liquids and gases at room temperature) Exists as diatomic molecules ( , , , ) Melting Boiling 2State2 2 2Density Point Point (at 20 °C) (at 20 °C) Fluorine -220 °C -188 °C Gas 0.0017 g/cm3 Chlorine -101 °C -34 °C Gas 0.0032 g/cm3 Br2 Bromine -7.25 °C 58.8 °C Liquid 3.123 g/cm3 Iodine 114 °C 185 °C Solid 4.93 g/cm3 A nonmetal I2 Volatile (evaporates easily) with corrosive fumes Does not occur in nature as a pure element. Electronegative; , and are strong acids; is one of the stronger weak acids 2 The Halogens (Group 17) What is a halogen? Only forms one monoatomic anion (-1) and no free cations Has seven valence electrons (valence electron configuration . ) and a large electron affinity 2 5 A good oxidizing agent (good at gaining electrons so that other elements can be oxidized) First Ionization Electron Affinity Standard Reduction Energy (kJ/mol) Potential (kJ/mol) (V = J/C) Fluorine 1681 328.0 +2.866 Chlorine 1251 349.0 +1.358 Bromine 1140 324.6 +1.065 Iodine 1008 295.2 +0.535 3 The Halogens (Group 17) Fluorine, chlorine and bromine are strong enough oxidizing agents that they can oxidize the oxygen in water! When fluorine is bubbled through water, hydrogen fluoride and oxygen gas are produced. -

Process for Purification of Ethylene Compound Having Fluorine-Containing Organic Group
Office europeen des brevets (fi) Publication number : 0 506 374 A1 @ EUROPEAN PATENT APPLICATION @ Application number: 92302586.0 @ Int. CI.5: C07C 17/38, C07C 21/18 (g) Date of filing : 25.03.92 (30) Priority : 26.03.91 JP 86090/91 (72) Inventor : Kishita, Hirofumi 3-19-1, Isobe, Annaka-shi Gunma-ken (JP) (43) Date of publication of application : Inventor : Sato, Shinichi 30.09.92 Bulletin 92/40 3-5-5, Isobe, Annaka-shi Gunma-ken (JP) Inventor : Fujii, Hideki (84) Designated Contracting States : 3-12-37, Isobe, Annaka-shi DE FR GB Gunma-ken (JP) Inventor : Matsuda, Takashi 791 ~4 YdndSG Anri3k3~shi @ Applicant : SHIN-ETSU CHEMICAL CO., LTD. Gunma-ken (JP) 6-1, Ohtemachi 2-chome Chiyoda-ku Tokyo (JP) @) Representative : Votier, Sidney David et al CARPMAELS & RANSFORD 43, Bloomsbury Square London WC1A 2RA (GB) (54) Process for purification of ethylene compound having fluorine-containing organic group. (57) A process for purifying an ethylene compound having a fluorine-containing organic group (fluorine- containing ethylene compound) by mixing the fluorine-containing ethylene compound with an alkali metal or alkaline earth metal reducing agent, and subjecting the resulting mixture to irradiation with ultraviolet radiation, followed by washing with water. The purification process ensures effective removal of iodides which are sources of molecular iodine, from the fluorine-containing ethylene compound. < h- CO CO o 10 o Q_ LU Jouve, 18, rue Saint-Denis, 75001 PARIS EP 0 506 374 A1 BACKGROUND OF THE INVENTION 1. Field of the Invention 5 The present invention relates to a process for purifying an ethylene compound having a fluorine-containing organic group, and more particularly to a purification process by which iodine contained as impurity in an ethylene compound having a fluorine-containing organic group can be removed effectively. -
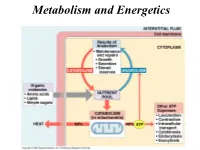
Metabolism and Energetics Oxidation of Carbon Atoms of Glucose Is the Major Source of Energy in Aerobic Metabolism
Metabolism and Energetics Oxidation of carbon atoms of glucose is the major source of energy in aerobic metabolism C6H1206 + 6O2 yields 6 CO2 + H20 + energy Energy released ΔG = - 686 kcal/mol Glucose oxidation requires over 25 discrete steps, with production of 36 ATP. Energy Transformations The mitochondrial synthesis of ATP is not stochiometric. Electron–motive force Proton-motive force Phosphoryl-transfer potential in the form of ATP. Substrate level phosphorylation The formation of ATP by substrate-level phosphorylation ADP ATP P CH O P CH2 O P 2 is used to represent HC OH HC OH a phosphate ester: phosphoglycerate CH OH OH CH2 O P kinase 2 P O bisphosphoglycerate 3-phosphoglycerate OH ADP ATP CH2 CH2 CH3 C O P C OH C O pyruvate kinase non-enzymic COOH COOH COOH phosphoenolpyruvate enolpyruvate pyruvate Why ATP? The reaction of ATP hydrolysis is very favorable ΔGo = - 30.5 kJ/mol = - 7.3 kCal/mol 1. Charge separation of closely packed phosphate groups provides electrostatic relief. Mg2+ 2. Inorganic Pi, the product of the reaction, is immediately resonance-stabilized (electron density spreads equally to all oxygens). 3. ADP immediately ionizes giving H+ into a low [H+] environment (pH~7). 4. Both Pi and ADP are more favorably solvated by water than one ATP molecule. 5. ATP is water soluble. The total body content of ATP and ADP is under 350 mmol – about 10 g, BUT … the amount of ATP synthesized and used each day is about 150 mol – about 110 kg. ATP Production - stage 1 - Glycolysis Glycolysis When rapid production of ATP is needed. -

Elemental Fluorine Product Information (Pdf)
Elemental Fluorine Contents 1 Introduction ............................................................................................................... 4 2.1 Technical Application of Fluorine ............................................................................. 5 2.2 Electronic Application of Fluorine ........................................................................... 7 2.3 Fluorine On-Site Plant ............................................................................................ 8 3 Specifications ............................................................................................................ 9 4 Safety ...................................................................................................................... 10 4.1 Maintenance of the F2 system .............................................................................. 12 4.2 First Aid ................................................................................................................ 13 5.1 Chemical Properties ............................................................................................. 14 5.2 Physical Data ....................................................................................................... 15 6 Toxicity .................................................................................................................... 18 7 Shipping and Transport ........................................................................................... 20 8 Environment ........................................................................................................... -

Reductions and Reducing Agents
REDUCTIONS AND REDUCING AGENTS 1 Reductions and Reducing Agents • Basic definition of reduction: Addition of hydrogen or removal of oxygen • Addition of electrons 9:45 AM 2 Reducible Functional Groups 9:45 AM 3 Categories of Common Reducing Agents 9:45 AM 4 Relative Reactivity of Nucleophiles at the Reducible Functional Groups In the absence of any secondary interactions, the carbonyl compounds exhibit the following order of reactivity at the carbonyl This order may however be reversed in the presence of unique secondary interactions inherent in the molecule; interactions that may 9:45 AM be activated by some property of the reacting partner 5 Common Reducing Agents (Borohydrides) Reduction of Amides to Amines 9:45 AM 6 Common Reducing Agents (Borohydrides) Reduction of Carboxylic Acids to Primary Alcohols O 3 R CO2H + BH3 R O B + 3 H 3 2 Acyloxyborane 9:45 AM 7 Common Reducing Agents (Sodium Borohydride) The reductions with NaBH4 are commonly carried out in EtOH (Serving as a protic solvent) Note that nucleophilic attack occurs from the least hindered face of the 8 carbonyl Common Reducing Agents (Lithium Borohydride) The reductions with LiBH4 are commonly carried out in THF or ether Note that nucleophilic attack occurs from the least hindered face of the 9:45 AM 9 carbonyl. Common Reducing Agents (Borohydrides) The Influence of Metal Cations on Reactivity As a result of the differences in reactivity between sodium borohydride and lithium borohydride, chemoselectivity of reduction can be achieved by a judicious choice of reducing agent. 9:45 AM 10 Common Reducing Agents (Sodium Cyanoborohydride) 9:45 AM 11 Common Reducing Agents (Reductive Amination with Sodium Cyanoborohydride) 9:45 AM 12 Lithium Aluminium Hydride Lithium aluminiumhydride reacts the same way as lithium borohydride. -

Chapter 20 Electrochemistry
Chapter 20 Electrochemistry Learning goals and key skills: Identify oxidation, reduction, oxidizing agent, and reducing agent in a chemical equation Complete and balance redox equations using the method of half-reactions. Sketch a voltaic cell and identify its cathode, anode, and the directions in which electrons and ions move. o Calculate standard emfs (cell potentials), E cell, from standard reduction potentials. Use reduction potentials to predict whether a redox reaction is spontaneous. o o Relate E cell to DG and equilibrium constants. Calculate emf under nonstandard conditions. Identify the components of common batteries. Describe the construction of a lithium-ion battery and explain how it works. Describe the construction of a fuel cell and explain how it generates electrical energy. Explain how corrosion occurs and how it is prevented by cathodic protection. Describe the reactions in electrolytic cells. Relate the amounts of products and reactants in redox reactions to electrical charge. Electrochemistry Electrochemistry is the study of the relationships between electricity and chemical reactions. • It includes the study of both spontaneous and nonspontaneous processes. 1 Redox reactions: assigning oxidation numbers Oxidation numbers help keep track of what species loses electrons and what species gains them. • An element is oxidized when the oxidation number increases • An element is reduced when the oxidation number decreases • an oxidizing agent causes another element to be oxidized • a reducing agent causes another element to be reduced. Assigning oxidation numbers (sect. 4.4) 1. Elemental form, each atom has ox. # = 0. Zn O2 O3 I2 S8 P4 2. Simple ions, = charge on ion. -1 for Cl-, +2 for Mg2+ 3. -

Redox Potential Measurements M.J
Redox Potential Measurements M.J. Vepraskas NC State University December 2002 Redox potential is an electrical measurement that shows the tendency of a soil solution to transfer electrons to or from a reference electrode. From this measurement we can estimate whether the soil is aerobic, anaerobic, and whether chemical compounds such as Fe oxides or nitrate have been chemically reduced or are present in their oxidized forms. Making these measurements requires three basic pieces of equipment: 1. Platinum electrode 2. Voltmeter 3. Reference electrode The basic set-up is shown below: Voltmeter 456 mv Soil surface Reference Pt wire electrode Fig. 1. The Pt wire is buried into the soil to be in contact with the soil solution. The reference electrode must also be in contact with the soil solution. It has a ceramic tip, which can be placed in the soil, or the electrode can be placed in a salt bridge, which is itself placed in the soil. Wires from both the Pt electrode and reference electrode are connected to the voltmeter. Pt Electrodes Platinum electrodes consist of a small piece of platinum wire that is soldered or fused to wire made another metal. Platinum conducts electrons from the soil solution to the wire to which it is attached. Platinum is used because it is assumed to be an inert metal. This means it does not give up its own electrons (does not oxidize) to the wire or soil solution. Iron containing materials such as steel will oxidize themselves and send their own electrons to the voltmeter. As a result the voltage we measure will not result solely from electrons being transferred to or from the soil.