The Adventures of a Citizen Scientist
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Harry Truman, the Atomic Bomb and the Apocalyptic Narrative
Volume 5 | Issue 7 | Article ID 2479 | Jul 12, 2007 The Asia-Pacific Journal | Japan Focus The Decision to Risk the Future: Harry Truman, the Atomic Bomb and the Apocalyptic Narrative Peter J. Kuznick The Decision to Risk the Future: Harry stressed that the future of mankind would be Truman, the Atomic Bomb and theshaped by how such bombs were used and Apocalyptic Narrative subsequently controlled or shared.[3] Truman recalled Stimson “gravely” expressing his Peter J. Kuznick uncertainty about whether the U.S. should ever use the bomb, “because he was afraid it was so I powerful that it could end up destroying the whole world.” Truman admitted that, listening In his personal narrative Atomic Quest, Nobel to Stimson and Groves and reading Groves’s Prize-winning physicist Arthur Holly Compton, accompanying memo, he “felt the same who directed atomic research at the University fear.”[4] of Chicago’s Metallurgical Laboratory during the Second World War, tells of receiving an urgent visit from J. Robert Oppenheimer while vacationing in Michigan during the summer of 1942. Oppenheimer and the brain trust he assembled had just calculated the possibility that an atomic explosion could ignite all the hydrogen in the oceans or the nitrogen in the atmosphere. If such a possibility existed, Compton concluded, “these bombs must never be made.” As Compton said, “Better to accept the slavery of the Nazis than to run a chance of drawing the final curtain on mankind.”[1] Certainly, any reasonable human being could be expected to respond similarly. Three years later, with Hitler dead and the Nazis defeated, President Harry Truman faced Truman and Byrnes en route to Potsdam, July a comparably weighty decision. -

No. 40. the System of Lunar Craters, Quadrant Ii Alice P
NO. 40. THE SYSTEM OF LUNAR CRATERS, QUADRANT II by D. W. G. ARTHUR, ALICE P. AGNIERAY, RUTH A. HORVATH ,tl l C.A. WOOD AND C. R. CHAPMAN \_9 (_ /_) March 14, 1964 ABSTRACT The designation, diameter, position, central-peak information, and state of completeness arc listed for each discernible crater in the second lunar quadrant with a diameter exceeding 3.5 km. The catalog contains more than 2,000 items and is illustrated by a map in 11 sections. his Communication is the second part of The However, since we also have suppressed many Greek System of Lunar Craters, which is a catalog in letters used by these authorities, there was need for four parts of all craters recognizable with reasonable some care in the incorporation of new letters to certainty on photographs and having diameters avoid confusion. Accordingly, the Greek letters greater than 3.5 kilometers. Thus it is a continua- added by us are always different from those that tion of Comm. LPL No. 30 of September 1963. The have been suppressed. Observers who wish may use format is the same except for some minor changes the omitted symbols of Blagg and Miiller without to improve clarity and legibility. The information in fear of ambiguity. the text of Comm. LPL No. 30 therefore applies to The photographic coverage of the second quad- this Communication also. rant is by no means uniform in quality, and certain Some of the minor changes mentioned above phases are not well represented. Thus for small cra- have been introduced because of the particular ters in certain longitudes there are no good determi- nature of the second lunar quadrant, most of which nations of the diameters, and our values are little is covered by the dark areas Mare Imbrium and better than rough estimates. -

Glossary Glossary
Glossary Glossary Albedo A measure of an object’s reflectivity. A pure white reflecting surface has an albedo of 1.0 (100%). A pitch-black, nonreflecting surface has an albedo of 0.0. The Moon is a fairly dark object with a combined albedo of 0.07 (reflecting 7% of the sunlight that falls upon it). The albedo range of the lunar maria is between 0.05 and 0.08. The brighter highlands have an albedo range from 0.09 to 0.15. Anorthosite Rocks rich in the mineral feldspar, making up much of the Moon’s bright highland regions. Aperture The diameter of a telescope’s objective lens or primary mirror. Apogee The point in the Moon’s orbit where it is furthest from the Earth. At apogee, the Moon can reach a maximum distance of 406,700 km from the Earth. Apollo The manned lunar program of the United States. Between July 1969 and December 1972, six Apollo missions landed on the Moon, allowing a total of 12 astronauts to explore its surface. Asteroid A minor planet. A large solid body of rock in orbit around the Sun. Banded crater A crater that displays dusky linear tracts on its inner walls and/or floor. 250 Basalt A dark, fine-grained volcanic rock, low in silicon, with a low viscosity. Basaltic material fills many of the Moon’s major basins, especially on the near side. Glossary Basin A very large circular impact structure (usually comprising multiple concentric rings) that usually displays some degree of flooding with lava. The largest and most conspicuous lava- flooded basins on the Moon are found on the near side, and most are filled to their outer edges with mare basalts. -

Atomic Physics & Quantum Effects
KEY CONCEPTS ATOMIC PHYSICS & QUANTUM EFFECTS 1. PHOTONS & THE PHOTOELECTRIC EFFECT Max Planck explained blackbody radiation with his quantum hypothesis, which states that the energy of a thermal oscillator, Eosc, is not continuous, but instead is a discrete quantity given by the equation: Eosc = nhf n = 1, 2, 3,... where f is the frequency and h is a constant now known as Planck’s constant. Albert Einstein extended the idea by adding that all emitted radiation is quantized. He suggested that light is composed of discrete quanta, rather than of waves. According to his theory, each particle of light, known as a photon, has an energy E given by: E = hf Einstein’s theory helped him explain a phenomenon known as the photoelectric effect, in which a photon of light strikes a photosensitive material and causes an electron to be ejected from the material. A photocell constructed from photosensitive material can produce an electrical current when light shines on it. The kinetic energy, K, of a photoelectron displaced by a photon of energy, hf, is given by: K = hf - φ where the work function, φ, is the minimum energy needed to free the electron from the photosensitive material. No photoemission occurs if the frequency of the incident light falls below a certain cutoff frequency – or threshold frequency – given by: φ f0 = h Einstein's theory explained several aspects of the photoelectric effect that could not be explained by classical theory: • The kinetic energy of photoelectrons is dependent on the light’s frequency. • No photoemission occurs for light below a certain threshold frequency. -

Norman Hilberry' Dr
Norman Hilberry' Dr. Norman Hilberry, now retired, was formerly director of Argonne Na tional Laboratory, one of the principal research and development centers in the field of nuclear science and engineering in the United States. The Laboratory is operated by the University of Chicago for the U. S. Atomic Energy Commis sion. Dr. Hilberry's association with the nuclear energy program began late in 1941 when, as personal aide, he joined Dr. A. H. Compton who had just been designated as project director for the Metallurgical Project, later known as the Plutonium Project. In this capacity Dr. Hilberry was associated with the group under Fermi which first achieved a self sustaining, controlled nuclear chain re action on December 2, 1942. As associate project director of the Metallurgical Project, he acted as the project office's representative at Clinton Laboratories during the start-up of the X-10 reactor in the Fall of 1943. Starting in the summer of 1944 he served ( again in his capacity as associate project director) as head of the resident project group which provided the official liaison between the DuPont organization and the Metallurgical Project during the start-up of the production facilities at the Hanford Engineer Works. With the successful operation of Hanford an accomplished fact, Dr. Hilberry returned to the Metallurgical Project office during the winter of 1945. After Dr. Compton's move to St. Louis as Chancellor of Washington University in July, 1945, he took over the closing out of the affairs of the central project office. Dur ing this period he assisted the Manhattan District of the U. -

The Spirit Rover's Athena Science Investigation at Gusev Crater, Mars
SPIRIT AT CUSEV CRATER RESEARCH ARTICLE The Spirit Rover's Athena Science Investigation at Gusev Crater, Mars S. W. Squyres,^* R. E. Arvidson,^ J. F. Bell III/ J. Bruckner,' N. A. Cabrol,'* W. Calvîn,= M. H. Carr,® P. R. Christensen/ B. C. Clark,® L Crumpler,® D. J. Des Marais,^" C. d'Uston,''^ T. Economou,^^ J. Farmer,^ W. Farrand," W. Folkner,^^ M. Colombek,^'* S. Corevan," J. A. Crant,^^ R. Creeley,'^ J. Crotzinger,^'^ L Haskin,^ K. E. Herkenhoff,^® S. Hvîid," J. Johnson,^® C. Klîngelhofer,^" A. Knoll,^^ C. Landis,^^ M. Lemmon,^' R. Li,^* M. B. Madsen,2= M. C. Malin,^^ s. M. McLennan,^^ H. Y. McSween,^« D. W. Ming,^^ J. Moersch,^« R. V. Morris,^^ T. Parker,^'* J. W. Rice Jr.,' L Richter,'" R. Rieder,' M. Sims,^° M. Smitli,'^ P. Smith,'^ L A. Soderblom,^® R. Sullivan,^ H. Wanke,' T. Wdowiak," M. Wolff,'^ A. Yen^'* The Mars Exploration Rover Spirit and its Atliena science payload liave been used to sediment carried by the water that cut investigate a landing site in Gusev crater. Gusev is hypothesized to be the site of a Ma'adim before it overflowed and exited former lake, but no clear evidence for lacustrine sedimentation has been found to through a gap in the northern rim of the date. Instead, the dominant lithology is basalt, and the dominant geologic processes are impact events and eolian transport. I^jany rocks exhibit coatings and other characteristics that may be evidence for minor aqueous alteration. Any lacustrine ^Department of Astronomy, Cornell University, Ithaca, NY 14853, USA. ^Department of Earth and sediments that may exist at this location within Gusev apparently have been buried Planetary Sciences, Washington University, St. -

Arthur Holly Compton
Arthur Holly Compton ALSO LISTED IN Physicists ALSO KNOWN AS Arthur Holly Compton FAMOUS AS Nobel Prize Laureate in Physics NATIONALITY American Famous American Men RELIGION Baptist BORN ON 10 September 1892 AD Famous 10th September Birthdays ZODIAC SIGN Virgo Virgo Men BORN IN Wooster, Ohio, USA DIED ON 15 March 1962 AD PLACE OF DEATH Berkeley, California, USA FATHER Elias Compton MOTHER Otelia Catherine SIBLINGS Karl Taylor Compton, Wilson Martindale Compton SPOUSE: Betty Charity McCloskey CHILDREN Arthur Allen Compton, John Joseph Compton EDUCATION University of Cambridge, The College of Wooster, Princeton University DISCOVERIES / INVENTIONS Compton Effect AWARDS: Nobel Prize for Physics (1927) Matteucci Medal (1930) Franklin Medal (1940) Hughes Medal (1940) Arthur Holly Compton was a renowned American physicist who first rose to fame with his famous revolutionary discovery of the Compton Effect for which he also won the Nobel Prize in Physics. This discovery confirmed the dual nature of electromagnetic radiation as both a wave and a particle. Thomson was initially interested in astronomy before he shifted his focus to the study of quantum physics. He started his research in Cavendish Laboratory of Cambridge University and this research led to the discovery of Compton Effect. Later on, during the Second World War, Compton became head of the Manhattan Project’s Metallurgical Laboratory. Manhattan Project developed the first nuclear weapons of the world and Compton played a key role in it. He also served as Chancellor of Washington University in St. Louis. Under his leadership, the University made remarkable academic progress; the university formally desegregated its undergraduate divisions, named its first female full professor, and enrolled a record number of students. -
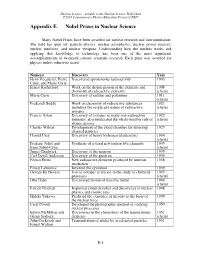
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

Prelim Reading List for Modern Physical Science (Last Updated: February 27, 2006)
Prelim Reading List for Modern Physical Science (last updated: February 27, 2006) Proposed preliminary examination reading list for Dana Freiburger. List of categories: 1 – Overview, Historiography, some ‘Classics’, and Survey Works 2.1 – 19th Century Physics 2.2 – 20th Century Physics 3 – Big Science 4 – Astronomy 5.1 – National Histories 5.2 – Atomic Weapons 6 – Sites of Research 7 – Instruments and Experiments 8 – Biography 9 – Japan Document History: 09/12/05 – First draft submitted to Richard 11/14/05 – Updated based on 10/12/05 meeting with Richard 02/27/06 – Updated to add categories to Endnote records, close to ‘final’ page 1 Prelim Reading List for Modern Physical Science (last updated: February 27, 2006) 1 – Overview, Historiography, some ‘Classics’, and Survey Works 01 Brown, Pais and Pippard, Twentieth century physics, 1995. 02 Cajori, A history of physics in its elementary branches (through 1925): including the evolution of physical laboratories, 1962. 03 Collins and Pinch, The Golem: What You Should Know About Science, 1998. 04 Dear, Revolutionizing the Sciences: European Knowledge and its Ambitions, 1500-1700, 2001. 05 Forman, The environment and practice of atomic physics in Weimar, Germany; a study in the history of science, 1968. 06 Fraser, The particle century, 1998. 07 Galison and Stump, The Disunity of Science: Boundaries, Contexts, and Power, 1996. 08 Kragh, Quantum Generations: a History of Physics in the Twentieth Century, 1999. 09 Kuhn, Black-body theory and the quantum discontinuity, 1894-1912, 1987. 10 Morus, When physics became king, 2005. 11 Nye, Before Big Science: the Pursuit of Modern Chemistry and Physics, 1800-1940, 1996. -

Applications in Solid-State Nuclear Magnetic Resonance and Physics
On Fer and Floquet-Magnus Expansions: Applications in Solid-State Nuclear Magnetic Resonance and Physics Eugene Stephane Mananga The City University of New York New York University International Conference on Physics June 27-29, 2016 New Orleans, LA, USA OUTLINE A. Background of NMR: Solid-State NMR • Principal References B. Commonly Used Methods in Solid-State NMR • Floquet Theory • Average Hamiltonian Theory C. Alternative Expansion Approaches Used Methods in SS-NMR • Fer Expansion • Floquet-Magnus Expansion D. Applications of Fer and Floquet-Magnus expansion in SS-SNMR E. Applications of Fer and Floquet-Magnus expansion in Physics A. Background of NMR: Solid-State NMR • NMR is an extraordinary versatile technique which started in Physics In 1945 and has spread with great success to Chemistry, Biochemistry, Biology, and Medicine, finding applications also in Geophysics, Archeology, Pharmacy, etc... • Hardly any discipline has remained untouched by NMR. • It is practiced in scientific labs everywhere, and no doubt before long will be found on the moon. • NMR has proved useful in elucidating problems in all forms of matter. In this talk we consider applications of NMR to solid state: Solid-State NMR BRIEF HISTORY OF NMR • 1920's Physicists Have Great Success With Quantum Theory • 1921 Stern and Gerlach Carry out Atomic and Molecular Beam Experiments • 1925/27 Schrödinger/ Heisenberg/ Dirac Formulate The New Quantum Mechanics • 1936 Gorter Attempts Experiments Using The Resonance Property of Nuclear Spin • 1937 Rabi Predicts and Observes -

Otto Stern Annalen 4.11.11
(To be published by Annalen der Physik in December 2011) Otto Stern (1888-1969): The founding father of experimental atomic physics J. Peter Toennies,1 Horst Schmidt-Böcking,2 Bretislav Friedrich,3 Julian C.A. Lower2 1Max-Planck-Institut für Dynamik und Selbstorganisation Bunsenstrasse 10, 37073 Göttingen 2Institut für Kernphysik, Goethe Universität Frankfurt Max-von-Laue-Strasse 1, 60438 Frankfurt 3Fritz-Haber-Institut der Max-Planck-Gesellschaft Faradayweg 4-6, 14195 Berlin Keywords History of Science, Atomic Physics, Quantum Physics, Stern- Gerlach experiment, molecular beams, space quantization, magnetic dipole moments of nucleons, diffraction of matter waves, Nobel Prizes, University of Zurich, University of Frankfurt, University of Rostock, University of Hamburg, Carnegie Institute. We review the work and life of Otto Stern who developed the molecular beam technique and with its aid laid the foundations of experimental atomic physics. Among the key results of his research are: the experimental test of the Maxwell-Boltzmann distribution of molecular velocities (1920), experimental demonstration of space quantization of angular momentum (1922), diffraction of matter waves comprised of atoms and molecules by crystals (1931) and the determination of the magnetic dipole moments of the proton and deuteron (1933). 1 Introduction Short lists of the pioneers of quantum mechanics featured in textbooks and historical accounts alike typically include the names of Max Planck, Albert Einstein, Arnold Sommerfeld, Niels Bohr, Max von Laue, Werner Heisenberg, Erwin Schrödinger, Paul Dirac, Max Born, and Wolfgang Pauli on the theory side, and of Wilhelm Conrad Röntgen, Ernest Rutherford, Arthur Compton, and James Franck on the experimental side. However, the records in the Archive of the Nobel Foundation as well as scientific correspondence, oral-history accounts and scientometric evidence suggest that at least one more name should be added to the list: that of the “experimenting theorist” Otto Stern. -

April 9, 2021 PAUL R. BIERMAN Department of Geology and 86 Brookes Avenue Rubenstein School of the Environment Burlington, VT 05
April 9, 2021 PAUL R. BIERMAN Department of Geology and 86 Brookes Avenue Rubenstein School of the Environment Burlington, VT 05401 and Natural Resources (802) 863-3609 (home) University of Vermont (802) 656-0045 (fax) Burlington, VT 05405 (802) 238-6826 (cell) (802) 656-4411 [email protected] http://uvm.edu/~pbierman Research and Teaching Interests Geomorphology, Geohydrology, Isotope and Low-temperature Geochemistry, Environmental Geology, Glacial Geology, Neotectonics, Surface Processes, Geoeducation, Geohazards, Science Writing, Public Outreach for Science Academic Training Ph.D., 1993, Geology, University of Washington, Seattle, WA with A. Gillespie "Cosmogenic Isotopes and the Evolution of Granitic Landforms" MS, 1990, Geology, University of Washington, Seattle, WA with A. Gillespie "Accuracy and Precision of Rock Varnish Cation Ratio Dating" BA, 1985, Geology and Environmental Studies, Williams College, Williamstown, MA "Deglaciation of Northwestern Massachusetts," (cum laude and senior thesis) Professional Experience 2002-present Professor University of Vermont, Geology Department and School of Natural Resources (secondary appointment) – have taught Climate: past, present, future, Geomorphology, Geohydrology, Geohazards, Regional Geology, Interdisciplinary Watershed Fieldcamp; Graduate seminars in Surface Processes, Environmental Geology, Neotectonics, Cosmogenic Isotopes, Critical Writing, First year Earth Hazards Seminar and lecture, and Introduction to Graduate Studies, on-line course in Vermont Landscape Change; advise 2 MS,