LA-UR-12-01344 Approved for Public Release; Distribution Is Unlimited
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
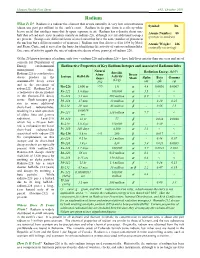
Radium What Is It? Radium Is a Radioactive Element That Occurs Naturally in Very Low Concentrations Symbol: Ra (About One Part Per Trillion) in the Earth’S Crust
Human Health Fact Sheet ANL, October 2001 Radium What Is It? Radium is a radioactive element that occurs naturally in very low concentrations Symbol: Ra (about one part per trillion) in the earth’s crust. Radium in its pure form is a silvery-white heavy metal that oxidizes immediately upon exposure to air. Radium has a density about one- Atomic Number: 88 half that of lead and exists in nature mainly as radium-226, although several additional isotopes (protons in nucleus) are present. (Isotopes are different forms of an element that have the same number of protons in the nucleus but a different number of neutrons.) Radium was first discovered in 1898 by Marie Atomic Weight: 226 and Pierre Curie, and it served as the basis for identifying the activity of various radionuclides. (naturally occurring) One curie of activity equals the rate of radioactive decay of one gram (g) of radium-226. Of the 25 known isotopes of radium, only two – radium-226 and radium-228 – have half-lives greater than one year and are of concern for Department of Energy environmental Radioactive Properties of Key Radium Isotopes and Associated Radionuclides management sites. Natural Specific Radiation Energy (MeV) Radium-226 is a radioactive Abun- Decay Isotope Half-Life Activity decay product in the dance Mode Alpha Beta Gamma (Ci/g) uranium-238 decay series (%) (α) (β) (γ) and is the precursor of Ra-226 1,600 yr >99 1.0 α 4.8 0.0036 0.0067 radon-222. Radium-228 is a radioactive decay product Rn-222 3.8 days 160,000 α 5.5 < < in the thorium-232 decay Po-218 3.1 min 290 million α 6.0 < < series. -

22Sra 226Ra and 222Rn in ' SOUTH CAROLINA GROUND WATER
Report No. 95 WRRI 22sRa 226Ra AND 222Rn IN ' SOUTH CAROLINA GROUND WATER: MEASUR~MEN-T TECHNIQUES AND ISOTOPE RELATIONSHIPS GlANNll\li FCUN~On o;: t,GR!CUL.TU~~tcoNOMlC2' ~~J.i\2Y J\li·iU ('lJ ~C 4· 1982 WATER RESOURCES RESEARCH INSTITUTE CLEMSON UNIVERSITY Clemson, .South Carolina JANUARY 1982 Water Resources Research Institute Clemson University Clemson, South Carolina 29631 ! 228Ra, 226Ra AND 222 Rn IN SOUTH CAROLINA GROUND WATER: MEASUREMENT TECHNIQUES AND ISOTOPE RELATIONSHIPS by Jacqueline Michel, Philip T. King and Willard S. Moore Department of Geology University of South Carolina Columbia, South Carolina 29208 The work upon which this puhlication is based was supported in part by funds provided by the Office of Water Research and Technology, project !!' No. OWRT-B-127-SC, U.S. Department of the Interior, fvashington, D. C., as authorized by the Water Research and Development Act of 1978 (PL95-467). r Project agreement No. 14-34-0001-9158 Period of Investigation: October 1979 - September 1980 Clemson University Water Resources Research Institute Technical Report No. 95 Contents of this publication do not necessarily reflect the views and policies of the Office of Water Research and Technology, U.S. Department of Interior, nor does mention of trade names or cormnercial products con stitute their endorsement or recommendation for use by the U.S. Goverrtment. ACKNOWLEDGEMENTS Much appreciation goes to Lewis Shaw, Rossie Stephens, Jim Ferguson and a large number of district personnel at the South Carolina Department of Health and Environmental Control who assisted us in field collection and provided much helpful information. Dr. -

Measurements of Fission Cross Sections for the Isotopes Relevant to the Thorium Fuel Cycle
EUROPEAN ORGANIZATION FOR NUCLEAR RESEARCH CERN/INTC 2001-025 INTC/P-145 08 Aug 2001 Measurements of Fission Cross Sections for the Isotopes relevant to the Thorium Fuel Cycle The n_TOF Collaboration ABBONDANNO U.20, ANDRIAMONJE S.10, ANDRZEJEWSKI J.24, ANGELOPOULOS A.12, ASSIMAKOPOULOS P.15, BACRI C-O.8, BADUREK G.1, BAUMANN P.9, BEER H.11, BENLLIURE J.32, BERTHIER B.8, BONDARENKO I.26, BOS A.J.J.36, BUSTREO N.19, CALVINO F.33, CANO-OTT D.28, CAPOTE R.31, CARLSON P.34, CHARPAK G.5, CHAUVIN N.8, CENNINI P.5, CHEPEL V.25, COLONNA N.20, CORTES G.33, CORTINA D.32, CORVI F.23, DAMIANOGLOU D.16, DAVID S.8, DIMOVASILI E.12, DOMINGO C.29, DOROSHENKO A.27, DURAN ESCRIBANO I.32, ELEFTHERIADIS C.16, EMBID M.28, FERRANT L.8, FERRARI A.5, FERREIRA–MARQUES R.25, FRAIS–KOELBL H.3, FURMAN W.26, FURSOV B.27, GARZON J.A.32, GIOMATARIS I.10, GLEDENOV Y.26, GONZALEZ–ROMERO E.28, GOVERDOVSKI A.27, GRAMEGNA F.19, GRIESMAYER E.3, GUNSING F.10, HAIGHT R.37, HEIL M.11, HOLLANDER P.36, IOANNIDES K.G.15, IOANNOU P.12, ISAEV S.27, JERICHA E.1, KADI Y.5, KAEPPELER F.11, KARADIMOS D.17, KARAMANIS D.15, KAYUKOV A.26, KAZAKOV L.27, KELIC A.9, KETLEROV V.27, KITIS G.16, KOEHLER P.E.38., KOPACH Y.26, KOSSIONIDES E.14, KROSHKINA I.27, LACOSTE V.5, LAMBOUDIS C.16, LEEB H.1, LEPRETRE A.10, LOPES M.25, LOZANO M.31, MARRONE S.20, MARTINEZ-VAL J. -

Experimental Γ Ray Spectroscopy and Investigations of Environmental Radioactivity
Experimental γ Ray Spectroscopy and Investigations of Environmental Radioactivity BY RANDOLPH S. PETERSON 216 α Po 84 10.64h. 212 Pb 1- 415 82 0- 239 β- 01- 0 60.6m 212 1+ 1630 Bi 2+ 1513 83 α β- 2+ 787 304ns 0+ 0 212 α Po 84 Experimental γ Ray Spectroscopy and Investigations of Environmental Radioactivity Randolph S. Peterson Physics Department The University of the South Sewanee, Tennessee Published by Spectrum Techniques All Rights Reserved Copyright 1996 TABLE OF CONTENTS Page Introduction ....................................................................................................................4 Basic Gamma Spectroscopy 1. Energy Calibration ................................................................................................... 7 2. Gamma Spectra from Common Commercial Sources ........................................ 10 3. Detector Energy Resolution .................................................................................. 12 Interaction of Radiation with Matter 4. Compton Scattering............................................................................................... 14 5. Pair Production and Annihilation ........................................................................ 17 6. Absorption of Gammas by Materials ..................................................................... 19 7. X Rays ..................................................................................................................... 21 Radioactive Decay 8. Multichannel Scaling and Half-life ..................................................................... -

THE NATURAL RADIOACTIVITY of the BIOSPHERE (Prirodnaya Radioaktivnost' Iosfery)
XA04N2887 INIS-XA-N--259 L.A. Pertsov TRANSLATED FROM RUSSIAN Published for the U.S. Atomic Energy Commission and the National Science Foundation, Washington, D.C. by the Israel Program for Scientific Translations L. A. PERTSOV THE NATURAL RADIOACTIVITY OF THE BIOSPHERE (Prirodnaya Radioaktivnost' iosfery) Atomizdat NMoskva 1964 Translated from Russian Israel Program for Scientific Translations Jerusalem 1967 18 02 AEC-tr- 6714 Published Pursuant to an Agreement with THE U. S. ATOMIC ENERGY COMMISSION and THE NATIONAL SCIENCE FOUNDATION, WASHINGTON, D. C. Copyright (D 1967 Israel Program for scientific Translations Ltd. IPST Cat. No. 1802 Translated and Edited by IPST Staff Printed in Jerusalem by S. Monison Available from the U.S. DEPARTMENT OF COMMERCE Clearinghouse for Federal Scientific and Technical Information Springfield, Va. 22151 VI/ Table of Contents Introduction .1..................... Bibliography ...................................... 5 Chapter 1. GENESIS OF THE NATURAL RADIOACTIVITY OF THE BIOSPHERE ......................... 6 § Some historical problems...................... 6 § 2. Formation of natural radioactive isotopes of the earth ..... 7 §3. Radioactive isotope creation by cosmic radiation. ....... 11 §4. Distribution of radioactive isotopes in the earth ........ 12 § 5. The spread of radioactive isotopes over the earth's surface. ................................. 16 § 6. The cycle of natural radioactive isotopes in the biosphere. ................................ 18 Bibliography ................ .................. 22 Chapter 2. PHYSICAL AND BIOCHEMICAL PROPERTIES OF NATURAL RADIOACTIVE ISOTOPES. ........... 24 § 1. The contribution of individual radioactive isotopes to the total radioactivity of the biosphere. ............... 24 § 2. Properties of radioactive isotopes not belonging to radio- active families . ............ I............ 27 § 3. Properties of radioactive isotopes of the radioactive families. ................................ 38 § 4. Properties of radioactive isotopes of rare-earth elements . -

Analysis of Isotopes of Radium, Uranium and Thorium. 210Pb-210Po
Analysis of isotopes of radium, uranium and thorium. 210Pb-210Po Per Roos Risø National Laboratory for Sustainable Energy, Technical university of Denmark Radium isotopes Adsorption of Ra on MnO2 & direct alpha spectrometry Coating of polyamid (Nylon) with MnO2 (KMnO4 solution) ……half-life of Ra-224 is 88h Very thin MnO2 layer, poor yield but excellent resolution Electrodeposited sources Direct 20 days 6 months Analysis of 226Ra in water • Weigh up a suitable amount of water (10 litre is mostly sufficient). • Add HNO3 to pH 1-2. • Add 133Ba tracer (ca 10 Bq). • Add about 13 ml of a 0.2M KMnO4. • Adjust pH to about 8-9 with ammonia. • Add 15 ml 0.3M MnCl2 and stir for some 5 minutes. • Adjust with KMnO4 or MnCl2 to make the sample water phase slightly pink. • Collect the precipitate and centrifuge • Dissolve precipitate in 1M HCl-1%H2O2. • Add about 2g K2SO4+1ml H2SO4 and dilute to 100 ml with water. • Heat the sample to dissolve the K2SO4 completely. • Add 30 mg Pb2+. Heat for 20 minutes and stir. • Centrifuge the precipitate and discard supernate. • Wash precipitate with 100 ml of a solution of 0.2M H2SO4 and 0.1 M K2SO4 • Centrifuge the precipitate and discard supernate • Dissolve the PbSO4-precipitate with 3ml 0.1M EDTA at pH 10. • Transfer the solution to the scintillation vial (low diffusion PE-vial). • Wash centrifuge tube with 2 ml 0.1M EDTA and transfer to Scintillation vial. • Add 10ml OptiFluor 0 and fill with deionised water to total 20 ml. • Allow for ingrowth of 222Rn during 3 weeks and measure by LSQ. -

A Note on Radioactive Materials and Their Measurements Elements Are
A NOTE ON RADIOACTIVE MATERIALS AND THEIR MEASUREMENTS Elements are placed in the periodic table of elements according to atomic number or proton number often referred to as Z. Their atomic weights are also usually indicated and can be fractional if the element in nature contains more than one isotope. Isotopes of an element all have the same atomic number, Z, but have different numbers of neu- trons. Some of these isotopes, even in naturally occurring elements, may be radioactive, and are usually given the atomic weight of the longest known isotope. Radioactive decay occurs when an unstable atom loses energy by emitting particles and gamma radiation or by spontaneously fission- ing. (Spontaneous fission is one of the natural modes of radioactive decay in which fission occurs without the addition of external energy or particles.) The activity of a radioactive sample is simply the rate of radioactive decay, expressed as the number of decays per unit time. Traditionally, the curie (Ci) was used to quantify the decay rate and was defined as the radioactivity measured for one gram of pure radium, or ~3.7 × 1010 decays per second (d/s), but the value changed as measurement techniques improved. To avoid this problem of a “chang- ing” constant, the Curie is now defined as exactly 3.7 × 1010 d/s. The becquerel (Bq), equal to one d/s, is the official SI unit (International System of Units). The roentgen R( ) is commonly used as a unit of radiation exposure to X-ray or gamma radiation, and can be roughly defined as “that quan- tity of X-ray or gamma radiation needed to produce one electrostatic unit of ionic charge in one cm3 of air under standard temperature and pressure (STP) “normal conditions”. -

12 Natural Isotopes of Elements Other Than H, C, O
12 NATURAL ISOTOPES OF ELEMENTS OTHER THAN H, C, O In this chapter we are dealing with the less common applications of natural isotopes. Our discussions will be restricted to their origin and isotopic abundances and the main characteristics. Only brief indications are given about possible applications. More details are presented in the other volumes of this series. A few isotopes are mentioned only briefly, as they are of little relevance to water studies. Based on their half-life, the isotopes concerned can be subdivided: 1) stable isotopes of some elements (He, Li, B, N, S, Cl), of which the abundance variations point to certain geochemical and hydrogeological processes, and which can be applied as tracers in the hydrological systems, 2) radioactive isotopes with half-lives exceeding the age of the universe (232Th, 235U, 238U), 3) radioactive isotopes with shorter half-lives, mainly daughter nuclides of the previous catagory of isotopes, 4) radioactive isotopes with shorter half-lives that are of cosmogenic origin, i.e. that are being produced in the atmosphere by interactions of cosmic radiation particles with atmospheric molecules (7Be, 10Be, 26Al, 32Si, 36Cl, 36Ar, 39Ar, 81Kr, 85Kr, 129I) (Lal and Peters, 1967). The isotopes can also be distinguished by their chemical characteristics: 1) the isotopes of noble gases (He, Ar, Kr) play an important role, because of their solubility in water and because of their chemically inert and thus conservative character. Table 12.1 gives the solubility values in water (data from Benson and Krause, 1976); the table also contains the atmospheric concentrations (Andrews, 1992: error in his Eq.4, where Ti/(T1) should read (Ti/T)1); 2) another category consists of the isotopes of elements that are only slightly soluble and have very low concentrations in water under moderate conditions (Be, Al). -

Naturally Occurring Radioactive Material (NORM VI) (NORM Material Radioactive Occurring Naturally Naturally Occurring Radioactive Material
Naturally Occurring Radioactive Material (NORM VI) Naturally Occurring Radioactive Material All minerals and raw materials contain radionuclides of natural origin. For (NORM VI) most human activities involving minerals and raw materials, the levels of exposure to these radionuclides are not significantly greater than normal background levels. However, certain work activities can give rise to signif- Proceedings icantly enhanced exposures that may need to be controlled by regulation. Material giving rise to these enhanced exposures has become known as of an International Symposium naturally occurring radioactive material (NORM). Historically, most regula- tory attention has been focused on the mining and processing of uranium Marrakesh, Morocco, 22–26 March 2010 ore, because such activities are a direct consequence of the radioactivity in the ore and form part of the nuclear fuel cycle. Over the past decade or two, however, more and more countries have introduced measures to reg- 1 ulate exposures arising from a wider range of natural sources. The NORM VI symposium, which was held in Marrakesh, Morocco, from 22–26 March 2010, provided an important opportunity to review the developments that had taken place during the three year period since the NORM V sym- posium in 2007. This period, which began with the publication of new radiation protection recommendations by the International Commission on Radiological Protection, was characterized by ongoing activities to revise international standards on radiation protection and safety. These Proceedings contain all 38 papers accepted for oral presentation and four rapporteur reports, as well as a summary that concludes with the main findings of the symposium. Text versions of 43 poster presentations are provided on a CD-ROM which accompanies these Proceedings. -

Experimental Aspects of Geoneutrino Detection: Status and Perspectives
Experimental Aspects of Geoneutrino Detection: Status and Perspectives O. Smirnov,1 1JINR, Joint Institute for Nuclear Research, Dubna, Russian Federation October 22, 2019 Abstract Neutrino geophysics, the study of the Earth's interior by measuring the fluxes of geologically produced neutrino at its surface, is a new interdisciplinary field of science, rapidly developing as a synergy between geology, geophysics and particle physics. Geoneutrinos, antineutrinos from long- lived natural isotopes responsible for the radiogenic heat flux, provide valuable information for the chemical composition models of the Earth. The calculations of the expected geoneutrino signal are discussed, together with experimental aspects of geoneutrino detection, including the description of possible backgrounds and methods for their suppression. At present, only two detectors, Borexino and KamLAND, have reached sensitivity to the geoneutrino. The experiments accumulated a set of ∼190 geoneutrino events and continue the data acquisition. The detailed description of the experiments, their results on geoneutrino detection, and impact on geophysics are presented. The start of operation of other detectors sensitive to geoneutrinos is planned for the near future: the SNO+ detector is being filled with liquid scintillator, and the biggest ever 20 kt JUNO detector is under construction. A review of the physics potential of these experiments with respect to the geoneutrino studies, along with other proposals, is presented. New ideas and methods for geoneutrino detection are reviewed. Contents 1 Introduction 2 2 Geoneutrinos and the Earth's heat 4 2.1 Long-lived radiogenic elements . 4 2.2 Radiogenic heat and geoneutrino luminosity of the Earth . 9 arXiv:1910.09321v1 [physics.geo-ph] 21 Oct 2019 3 Geoneutrino flux calculation 11 3.1 Neutrino oscillations . -

Radionuclides Notice of Data Availability Technical Support Document
Radionuclides Notice of Data Availability Technical Support Document March 2000 Targeting and Analysis Branch Standards and Risk Management Division Office of Ground Water and Drinking Water United States Environmental Protection Agency Prepared by: USEPA Office of Ground Water and Drinking Water in collaboration with USEPA Office of Indoor Air and Radiation United States Geological Survey Preface Under the authority of the Safe Drinking Water Act (SDWA) and its amendments, EPA sets drinking water standards for contaminants, which potentially pose a threat to public health via a public drinking water source. In 1976, EPA promulgated National Interim Primary Drinking Water Regulations (NIPDWR) for three categories of radionuclides: gross alpha emitters, radium 226 and 228 combined, and gross beta and photon emitters. The 1976 NIPDWRs for these radionuclides regulated: 1. gross alpha at 15 picoCuries per liter (pCi/L)(excluding radon and uranium); 2. radium-226 and radium-228 combined at 5 pCi/L; 3. and beta/photon emitters at a 4 millirem dose of radioactivity. The 1986 reauthorization of the Safe Drinking Water Act (SDWA) declared the interim standards for these radionuclides to be final National Primary Drinking Water Regulations (NPDWR). The 1986 amendment also required standards to be set as close to the maximum contaminant level goal (MCLG; the health goal) as possible. At that time, radionuclides did not have an MCLG. In 1991 (56 FR 33050), EPA proposed: < A Maximum Contaminant Level Goal (MCLG) of zero for all radionuclides; < A -

A Global Scavenging and Circulation Ocean Model of Thorium-230 and Protactinium-231 with Improved Particle Dynamics (NEMO–Prothorp 0.1)
Geosci. Model Dev., 11, 3537–3556, 2018 https://doi.org/10.5194/gmd-11-3537-2018 © Author(s) 2018. This work is distributed under the Creative Commons Attribution 4.0 License. A global scavenging and circulation ocean model of thorium-230 and protactinium-231 with improved particle dynamics (NEMO–ProThorP 0.1) Marco van Hulten1,2, Jean-Claude Dutay1, and Matthieu Roy-Barman1 1Laboratoire des Sciences du Climat et de l’Environnement, IPSL, CEA–Orme des Merisiers, 91191 Gif-sur-Yvette, France 2Geophysical Institute, University of Bergen, Bergen, Norway Correspondence: Marco van Hulten ([email protected]) Received: 1 November 2017 – Discussion started: 8 December 2017 Revised: 16 August 2018 – Accepted: 20 August 2018 – Published: 31 August 2018 Abstract. In this paper we set forth a 3-D ocean model of the can be done based on our model as its source code is readily radioactive trace isotopes 230Th and 231Pa. The interest arises available. from the fact that these isotopes are extensively used for in- vestigating particle transport in the ocean and reconstructing past ocean circulation. The tracers are reversibly scavenged 1 Introduction by biogenic and lithogenic particles. Our simulations of 230Th and 231Pa are based on the Oceanic circulation and the carbon cycle play a major role NEMO–PISCES ocean biogeochemistry general circulation in the regulation of the past and present climate. Heat and model, which includes biogenic particles, namely small and carbon dioxide in the atmosphere tend to equilibrate with the big particulate organic carbon, calcium carbonate and bio- ocean surface and are transported down into the deep ocean genic silica.