Hep28280 Am.Pdf
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Treatment of Herpes Simplex Virus Epithelial Keratitis
THE TREATMENT OF HERPES SIMPLEX VIRUS EPITHELIAL KERATITIS BY Kirk R. Wilhelmus, MD, MPH ABSTRACT Purpose: Epithelial keratitis is the most common presentation of ocular infection by herpes simplex virus (HSV). Quantitative assessment of available therapy is needed to guide evidence-based ophthalmology. This study aimed to compare the efficacy of various treatments for dendritic or geographic HSV epithelial keratitis and to evaluate the role of various clinical characteristics on epithelial healing. Methods: Following a systematic review of the literature, information from clinical trials of HSV dendritic or geographic epithelial keratitis was extracted, and the methodological quality of each study was scored. Methods of epithelial cau- terization and curettage were grouped as relatively equivalent physicochemical therapy, and solution and ointment for- mulations of a given topical antiviral agent were combined. The proportion healed with 1 week of therapy, a scheduled follow-up day that approximated the average time of resolution with antiviral therapy, was selected as the primary out- come based on a masked evaluation of maximum treatment differences in published healing curves. The proportion healed at 14 days was recorded as supplemental information. Fixed-effects and random-effects meta-analysis models were used to obtain summary estimates by pooling results from comparative treatment trials. Hypotheses about which prognostic factors might affect epithelial healing during antiviral therapy were developed by multivariate analysis of the Herpetic Eye Disease Study dataset. Results: After excluding 48 duplicate reports, 14 nonrandomized studies, 15 studies with outdated or similar treatments, and 29 trials lacking sufficient data on healing or accessibility, 76 primary reports were identified. -
(12) United States Patent (10) Patent No.: US 8,993,581 B2 Perrine Et Al
US00899.3581B2 (12) United States Patent (10) Patent No.: US 8,993,581 B2 Perrine et al. (45) Date of Patent: Mar. 31, 2015 (54) METHODS FOR TREATINGVIRAL (58) Field of Classification Search DSORDERS CPC ... A61K 31/00; A61K 31/166; A61K 31/185: A61K 31/233; A61K 31/522: A61K 38/12: (71) Applicant: Trustees of Boston University, Boston, A61K 38/15: A61K 45/06 MA (US) USPC ........... 514/263.38, 21.1, 557, 565, 575, 617; 424/2011 (72) Inventors: Susan Perrine, Weston, MA (US); Douglas Faller, Weston, MA (US) See application file for complete search history. (73) Assignee: Trustees of Boston University, Boston, (56) References Cited MA (US) U.S. PATENT DOCUMENTS (*) Notice: Subject to any disclaimer, the term of this 3,471,513 A 10, 1969 Chinn et al. patent is extended or adjusted under 35 3,904,612 A 9/1975 Nagasawa et al. U.S.C. 154(b) by 0 days. (Continued) (21) Appl. No.: 13/915,092 FOREIGN PATENT DOCUMENTS (22) Filed: Jun. 11, 2013 CA 1209037 A 8, 1986 CA 2303268 A1 4f1995 (65) Prior Publication Data (Continued) US 2014/OO45774 A1 Feb. 13, 2014 OTHER PUBLICATIONS Related U.S. Application Data (63) Continuation of application No. 12/890,042, filed on PCT/US 10/59584 Search Report and Written Opinion mailed Feb. Sep. 24, 2010, now abandoned. 11, 2011. (Continued) (60) Provisional application No. 61/245,529, filed on Sep. 24, 2009, provisional application No. 61/295,663, filed on Jan. 15, 2010. Primary Examiner — Savitha Rao (74) Attorney, Agent, or Firm — Nixon Peabody LLP (51) Int. -

WO 2015/063180 Al 7 May 2015 (07.05.2015) P O P C T
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2015/063180 Al 7 May 2015 (07.05.2015) P O P C T (51) International Patent Classification: (74) Agent: WILLIAMS, Rachel; Novozymes Biopharma UK A61K 39/00 (2006.01) C07K 16/00 (2006.01) Ltd., Castle Court, 59 Castle Boulevard, Nottingham Not A61K 39/395 (2006.01) tinghamshire NG7 1FD (GB). (21) International Application Number: (81) Designated States (unless otherwise indicated, for every PCT/EP2014/073261 kind of national protection available): AE, AG, AL, AM, AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, (22) International Filing Date: BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, 29 October 2014 (29.10.2014) DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (25) Filing Language: English HN, HR, HU, ID, IL, IN, IR, IS, JP, KE, KG, KN, KP, KR, KZ, LA, LC, LK, LR, LS, LU, LY, MA, MD, ME, MG, (26) Publication Language: English MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, (30) Priority Data: PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SA, SC, 13 190750.3 29 October 2013 (29. 10.2013) EP SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, 14166865.7 2 May 2014 (02.05.2014) EP TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (71) Applicant: NOVOZYMES BIOPHARMA DK A S (84) Designated States (unless otherwise indicated, for every [DK/DK]; Krogshoejvej 36, DK-2880 Bagsvaerd (DK). -

“Amenalief® Tab. 200Mg” in Japan
News Release July 3, 2017 Maruho Co., Ltd. Maruho receives manufacturing and marketing approval for Anti-Herpes Virus Agent “Amenalief® Tab. 200mg” in Japan Osaka (Japan), July 3, 2017 – Maruho Co., Ltd (“Maruho”, Head Office: Osaka, Japan, President and CEO: Koichi Takagi) announces that today it has received manufacturing and marketing approval from the Japanese Ministry of Health, Labor and Welfare (MHLW), for anti-herpes virus agent “Amenalief® Tab. 200mg” (INN: amenamevir) (hereinafter referred to as “the product”) for the treatment of herpes zoster (shingles) in Japan. The product is an anti-herpes virus agent with a novel mechanism of action created by Astellas Pharma Inc. (“Astellas”; Head Office: Tokyo, President and CEO: Yoshihiko Hatanaka). The product has been observed to inhibit the proliferation of the varicella-zoster virus (hereinafter VZV) by inhibiting the activity of the helicase-primase complex, which is essential for viral DNA replication. In August 2012, Maruho and Astellas agreed on a license agreement for the development and commercialization of the product in Japan, and Maruho has been progressing its development. Herpes zoster (shingles) is a disease caused by reactivation of the chickenpox VZV in latently-infected nerve ganglia. The main treatment for shingles is anti-herpes virus agents. The product is proven to be effective against VZV when administered once a day after meals. Also, since most of the product is excreted in feces, it is not necessary to adjust the dosage and administration according to creatinine clearance, an indicator of kidney function. Maruho hopes the product will contribute to the expansion of treatment options and improvement in adherence for the treatment of shingles in Japan. -

Infectious Diseases
2013 MEDICINES IN DEVELOPMENT REPORT Infectious Diseases A Report on Diseases Caused by Bacteria, Viruses, Fungi and Parasites PRESENTED BY AMERICA’S BIOPHARMACEUTICAL RESEARCH COMPANIES Biopharmaceutical Research Evolves Against Infectious Diseases with Nearly 400 Medicines and Vaccines in Testing Throughout history, infectious diseases hepatitis C that inhibits the enzyme have taken a devastating toll on the lives essential for viral replication. and well-being of people around the • An anti-malarial drug that has shown Medicines in Development world. Caused when pathogens such activity against Plasmodium falci- For Infectious Diseases as bacteria or viruses enter a body and parum malaria which is resistant to multiply, infectious diseases were the current treatments. Application leading cause of death in the United Submitted States until the 1920s. Today, vaccines • A potential new antibiotic to treat methicillin-resistant Staphylococcus Phase III and infectious disease treatments have proven to be effective treatments in aureus (MRSA). Phase II many cases, but infectious diseases still • A novel treatment that works by Phase I pose a very serious threat to patients. blocking the ability of the smallpox Recently, some infectious pathogens, virus to spread to other cells, thus 226 such as pseudomonas bacteria, have preventing it from causing disease. become resistant to available treatments. Infectious diseases may never be fully Diseases once considered conquered, eradicated. However, new knowledge, such as tuberculosis, have reemerged new technologies, and the continuing as a growing health threat. commitment of America’s biopharma- America’s biopharmaceutical research ceutical research companies can help companies are developing 394 medicines meet the continuing—and ever-changing and vaccines to combat the many threats —threat from infectious diseases. -

Predictive QSAR Tools to Aid in Early Process Development of Monoclonal Antibodies
Predictive QSAR tools to aid in early process development of monoclonal antibodies John Micael Andreas Karlberg Published work submitted to Newcastle University for the degree of Doctor of Philosophy in the School of Engineering November 2019 Abstract Monoclonal antibodies (mAbs) have become one of the fastest growing markets for diagnostic and therapeutic treatments over the last 30 years with a global sales revenue around $89 billion reported in 2017. A popular framework widely used in pharmaceutical industries for designing manufacturing processes for mAbs is Quality by Design (QbD) due to providing a structured and systematic approach in investigation and screening process parameters that might influence the product quality. However, due to the large number of product quality attributes (CQAs) and process parameters that exist in an mAb process platform, extensive investigation is needed to characterise their impact on the product quality which makes the process development costly and time consuming. There is thus an urgent need for methods and tools that can be used for early risk-based selection of critical product properties and process factors to reduce the number of potential factors that have to be investigated, thereby aiding in speeding up the process development and reduce costs. In this study, a framework for predictive model development based on Quantitative Structure- Activity Relationship (QSAR) modelling was developed to link structural features and properties of mAbs to Hydrophobic Interaction Chromatography (HIC) retention times and expressed mAb yield from HEK cells. Model development was based on a structured approach for incremental model refinement and evaluation that aided in increasing model performance until becoming acceptable in accordance to the OECD guidelines for QSAR models. -
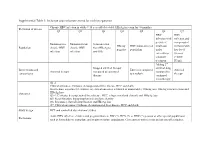
Inclusion and Exclusion Criteria for Each Key Question
Supplemental Table 1: Inclusion and exclusion criteria for each key question Chronic HBV infection in adults ≥ 18 year old (detectable HBsAg in serum for >6 months) Definition of disease Q1 Q2 Q3 Q4 Q5 Q6 Q7 HBV HBV infection with infection and persistent compensated Immunoactive Immunotolerant Seroconverted HBeAg HBV mono-infected viral load cirrhosis with Population chronic HBV chronic HBV from HBeAg to negative population under low level infection infection anti-HBe entecavir or viremia tenofovir (<2000 treatment IU/ml) Adding 2nd Stopped antiviral therapy antiviral drug Interventions and Entecavir compared Antiviral Antiviral therapy compared to continued compared to comparisons to tenofovir therapy therapy continued monotherapy Q1-2: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Intermediate outcomes (if evidence on clinical outcomes is limited or unavailable): HBsAg loss, HBeAg seroconversion and Outcomes HBeAg loss Q3-4: Cirrhosis, decompensated liver disease, HCC, relapse (viral and clinical) and HBsAg loss Q5: Renal function, hypophosphatemia and bone density Q6: Resistance, flare/decompensation and HBeAg loss Q7: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Study design RCT and controlled observational studies Acute HBV infection, children and pregnant women, HIV (+), HCV (+) or HDV (+) persons or other special populations Exclusions such as hemodialysis, transplant, and treatment failure populations. Co treatment with steroids and uncontrolled studies. Supplemental Table 2: Detailed Search Strategy: Ovid Database(s): Embase 1988 to 2014 Week 37, Ovid MEDLINE(R) In-Process & Other Non- Indexed Citations and Ovid MEDLINE(R) 1946 to Present, EBM Reviews - Cochrane Central Register of Controlled Trials August 2014, EBM Reviews - Cochrane Database of Systematic Reviews 2005 to July 2014 Search Strategy: # Searches Results 1 exp Hepatitis B/dt 26410 ("hepatitis B" or "serum hepatitis" or "hippie hepatitis" or "injection hepatitis" or 2 178548 "hepatitis type B").mp. -

Aicuris Granted Fast Track Designation by U.S. FDA for Oral Pritelivir for Treatment of HSV Infections in Immunocompromised Adults
AiCuris Granted Fast Track Designation by U.S. FDA for Oral Pritelivir for Treatment of HSV Infections in Immunocompromised Adults Wuppertal, August 01, 2017 - AiCuris Anti-infective Cures GmbH, a leading company in the discovery and development of drugs against infectious diseases, today announced that the Company has been granted Fast Track designation by the U.S. Food and Drug Administration (FDA) for oral pritelivir, AiCuris’ lead candidate for the treatment of acyclovir-resistant mucocutaneous herpes simplex virus (HSV) infections in immunocompromised adults. Fast track is a process designed to facilitate the development, expedite the review and accelerate the approval process of drugs to treat serious conditions and fill an unmet medical need, with the purpose of getting important new drugs to patients sooner. Oral pritelivir, a small molecule helicase-primase inhibitor with a novel mode of action, is currently in a clinical phase 2 study, called PRIOH-1, in the U.S. to evaluate the product candidate’s efficacy and safety compared to i.v. foscarnet, a virostatic agent which is used mainly for the treatment of herpes viruses resistant to other antiviral drugs. In a prior phase 2 study, oral pritelivir showed to significantly improve the suppression of viral shedding compared to the current standard of care for genital HSV-2 infections, the nucleoside analog valacyclovir. The results of this study were published in the Journal of the American Medical Association (JAMA) earlier this year. “The decision by the FDA to grant fast track designation to oral pritelivir underscores that our product might fill the major need for innovative, more efficacious therapies for immunocompromised patients with HSV infections that have become resistant to standard treatments,” said Dr. -

)&F1y3x PHARMACEUTICAL APPENDIX to THE
)&f1y3X PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE )&f1y3X PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 3 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. Product CAS No. Product CAS No. ABAMECTIN 65195-55-3 ACTODIGIN 36983-69-4 ABANOQUIL 90402-40-7 ADAFENOXATE 82168-26-1 ABCIXIMAB 143653-53-6 ADAMEXINE 54785-02-3 ABECARNIL 111841-85-1 ADAPALENE 106685-40-9 ABITESARTAN 137882-98-5 ADAPROLOL 101479-70-3 ABLUKAST 96566-25-5 ADATANSERIN 127266-56-2 ABUNIDAZOLE 91017-58-2 ADEFOVIR 106941-25-7 ACADESINE 2627-69-2 ADELMIDROL 1675-66-7 ACAMPROSATE 77337-76-9 ADEMETIONINE 17176-17-9 ACAPRAZINE 55485-20-6 ADENOSINE PHOSPHATE 61-19-8 ACARBOSE 56180-94-0 ADIBENDAN 100510-33-6 ACEBROCHOL 514-50-1 ADICILLIN 525-94-0 ACEBURIC ACID 26976-72-7 ADIMOLOL 78459-19-5 ACEBUTOLOL 37517-30-9 ADINAZOLAM 37115-32-5 ACECAINIDE 32795-44-1 ADIPHENINE 64-95-9 ACECARBROMAL 77-66-7 ADIPIODONE 606-17-7 ACECLIDINE 827-61-2 ADITEREN 56066-19-4 ACECLOFENAC 89796-99-6 ADITOPRIM 56066-63-8 ACEDAPSONE 77-46-3 ADOSOPINE 88124-26-9 ACEDIASULFONE SODIUM 127-60-6 ADOZELESIN 110314-48-2 ACEDOBEN 556-08-1 ADRAFINIL 63547-13-7 ACEFLURANOL 80595-73-9 ADRENALONE -

A Screening-Based Approach to Circumvent Tumor Microenvironment
JBXXXX10.1177/1087057113501081Journal of Biomolecular ScreeningSingh et al. 501081research-article2013 Original Research Journal of Biomolecular Screening 2014, Vol 19(1) 158 –167 A Screening-Based Approach to © 2013 Society for Laboratory Automation and Screening DOI: 10.1177/1087057113501081 Circumvent Tumor Microenvironment- jbx.sagepub.com Driven Intrinsic Resistance to BCR-ABL+ Inhibitors in Ph+ Acute Lymphoblastic Leukemia Harpreet Singh1,2, Anang A. Shelat3, Amandeep Singh4, Nidal Boulos1, Richard T. Williams1,2*, and R. Kiplin Guy2,3 Abstract Signaling by the BCR-ABL fusion kinase drives Philadelphia chromosome–positive acute lymphoblastic leukemia (Ph+ ALL) and chronic myelogenous leukemia (CML). Despite their clinical activity in many patients with CML, the BCR-ABL kinase inhibitors (BCR-ABL-KIs) imatinib, dasatinib, and nilotinib provide only transient leukemia reduction in patients with Ph+ ALL. While host-derived growth factors in the leukemia microenvironment have been invoked to explain this drug resistance, their relative contribution remains uncertain. Using genetically defined murine Ph+ ALL cells, we identified interleukin 7 (IL-7) as the dominant host factor that attenuates response to BCR-ABL-KIs. To identify potential combination drugs that could overcome this IL-7–dependent BCR-ABL-KI–resistant phenotype, we screened a small-molecule library including Food and Drug Administration–approved drugs. Among the validated hits, the well-tolerated antimalarial drug dihydroartemisinin (DHA) displayed potent activity in vitro and modest in vivo monotherapy activity against engineered murine BCR-ABL-KI–resistant Ph+ ALL. Strikingly, cotreatment with DHA and dasatinib in vivo strongly reduced primary leukemia burden and improved long-term survival in a murine model that faithfully captures the BCR-ABL-KI–resistant phenotype of human Ph+ ALL. -

Herpes Simplex Virus
HSV Herpes simplex virus HSV (Herpes simplex virus) can be spread when an infected person is producing and shedding the virus. Herpes simplex can be spread through contact with saliva, such as sharing drinks. Symptoms of herpes simplex virus infection include watery blisters in the skin or mucous membranes of the mouth, lips or genitals. Lesions heal with ascab characteristic of herpetic disease. As neurotropic and neuroinvasive viruses, HSV-1 and -2 persist in the body by becoming latent and hiding from the immune system in the cell bodies of neurons. After the initial or primary infection, some infected people experience sporadic episodes of viral reactivation or outbreaks. www.MedChemExpress.com 1 HSV Inhibitors (Z)-Capsaicin 1-Docosanol (Zucapsaicin; Civamide; cis-Capsaicin) Cat. No.: HY-B1583 (Behenyl alcohol) Cat. No.: HY-B0222 (Z)-Capsaicin is the cis isomer of capsaicin, acts 1-Docosanol is a saturated fatty alcohol used as an orally active TRPV1 agonist, and is used in traditionally as an emollient, emulsifier, and the research of neuropathic pain. thickener in cosmetics, and nutritional supplement; inhibitor of lipid-enveloped viruses including herpes simplex. Purity: 99.96% Purity: ≥98.0% Clinical Data: Launched Clinical Data: Launched Size: 10 mM × 1 mL, 10 mg, 50 mg Size: 500 mg 2-Deoxy-D-glucose 20(R)-Ginsenoside Rh2 (2-DG; 2-Deoxy-D-arabino-hexose; D-Arabino-2-deoxyhexose) Cat. No.: HY-13966 Cat. No.: HY-N1401 2-Deoxy-D-glucose is a glucose analog that acts as 20(R)-Ginsenoside Rh2, a matrix a competitive inhibitor of glucose metabolism, metalloproteinase (MMP) inhibitor, acts as a inhibiting glycolysis via its actions on hexokinase. -

Phage Display-Based Strategies for Cloning and Optimization of Monoclonal Antibodies Directed Against Human Pathogens
Int. J. Mol. Sci. 2012, 13, 8273-8292; doi:10.3390/ijms13078273 OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Review Phage Display-based Strategies for Cloning and Optimization of Monoclonal Antibodies Directed against Human Pathogens Nicola Clementi *,†, Nicasio Mancini †, Laura Solforosi, Matteo Castelli, Massimo Clementi and Roberto Burioni Microbiology and Virology Unit, “Vita-Salute” San Raffaele University, Milan 20132, Italy; E-Mails: [email protected] (N.M.); [email protected] (L.S.); [email protected] (M.C.); [email protected] (M.C.); [email protected] (R.B.) † These authors contributed equally to this work. * Author to whom correspondence should be addressed; E-Mail: [email protected]; Tel.: +39-2-2643-5082; Fax: +39-2-2643-4288. Received: 16 March 2012; in revised form: 25 June 2012 / Accepted: 27 June 2012 / Published: 4 July 2012 Abstract: In the last two decades, several phage display-selected monoclonal antibodies (mAbs) have been described in the literature and a few of them have managed to reach the clinics. Among these, the anti-respiratory syncytial virus (RSV) Palivizumab, a phage-display optimized mAb, is the only marketed mAb directed against microbial pathogens. Palivizumab is a clear example of the importance of choosing the most appropriate strategy when selecting or optimizing an anti-infectious mAb. From this perspective, the extreme versatility of phage-display technology makes it a useful tool when setting up different strategies for the selection of mAbs directed against human pathogens, especially when their possible clinical use is considered.