A Medical Mystery J M Van Der Klooster
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Age-Related Features and Pathology of Blood in Children
MINISTRY OF PUBLIC HEALTH OF UKRAINE HIGHER STATE EDUCATIONAL ESTABLISHMENT OF UKRAINE «UKRAINIAN MEDICAL STOMATOLOGICAL ACADEMY» V.I. POKHYLKO, S.M. TSVIRENKO, YU.V. LYSANETS AGE-RELATED FEATURES AND PATHOLOGY OF BLOOD IN CHILDREN MANUAL FOR STUDENTS OF HIGHER MEDICAL EDUCATIONAL INSTITUTIONS OF THE III-IV ACCREDITATION LEVELS Poltava 2017 МІНІСТЕРСТВО ОХОРОНИ ЗДОРОВ’Я УКРАЇНИ ВИЩИЙ ДЕРЖАВНИЙ НАВЧАЛЬНИЙ ЗАКЛАД УКРАЇНИ «УКРАЇНСЬКА МЕДИЧНА СТОМАТОЛОГІЧНА АКАДЕМІЯ» ПОХИЛЬКО В.І., ЦВІРЕНКО С.М., ЛИСАНЕЦЬ Ю.В. ВІКОВІ ОСОБЛИВОСТІ ТА ПАТОЛОГІЯ КРОВІ У ДІТЕЙ НАВЧАЛЬНИЙ ПОСІБНИК ДЛЯ СТУДЕНТІВ ВИЩИХ МЕДИЧНИХ НАВЧАЛЬНИХ ЗАКЛАДІВ III-IV РІВНІВ АКРЕДИТАЦІЇ Полтава 2017 2 UDC: 616+616.15]-053.2(075.8) ВВС: 57.33я73 The manual highlights the issues of embryogenesis, age-related features, semiotics of lesion, examination methods and diseases of hemic system in children. The manual is intended for students of higher educational institutions of III-IV accreditation levels, and can be used by medical interns and primary care doctors. Authors: Doctor of Medical Sciences, Professor of the Department of Pediatrics No.1 with Propedeutics and Neonatology V.I. Pokhylko Candidate of Medical Sciences, Acting Head of the Department of Pediatrics No.1 with Propedeutics and Neonatology S.M. Tsvirenko Candidate of Philological Sciences, Senior Lecturer of the Department of Foreign Languages with Latin and Medical Terminology Yu.V. Lysanets Reviewers: O.S. Yablon’ ― Doctor of Medical Sciences, Professor, Head of the Department of Pediatrics No.1, Vinnytsya National M.I. Pirogov Memorial Medical University of Ministry of Public Health of Ukraine. M.O. Honchar ― Doctor of Medical Sciences, Professor, Head of the Department of Pediatrics and Neonatology No.1, Kharkiv National Medical University. -

Erythrocytes: Overview, Morphology, Quantity by AH Rebar Et
In: A Guide to Hematology in Dogs and Cats, Rebar A.H., MacWilliams P.S., Feldman B.F., Metzger F.L., Pollock R.V.H. and Roche J. (Eds.). Publisher: Teton NewMedia, Jackson WY (www.veterinarywire.com). Internet Publisher: International Veterinary Information Service, Ithaca NY (www.ivis.org), 8-Feb-2005; A3304.0205 Erythrocytes: Overview, Morphology, Quantity A.H. Rebar1, P.S. MacWilliams2, B.F. Feldman 3, F.L. Metzger 4, R.V.H. Pollock 5 and J. Roche 6 1Dept of Veterinary Pathobiology, School of Veterinary Medicine, Purdue University, IN,USA. 2Dept of Pathobiological Sciences, School of Veterinary Medicine, University of Wisconsin, WI, USA. 3Dept of Biomedical Sciences & Pathobiology, VA-MD - Regional College of Veterinary Medicine, Virginia Tech, VA, USA. 4Metzger Animal Hospital,State College,PA, USA. 5Fort Hill Company, Montchanin, DE, USA. 6 Hematology Systems, IDEXX Laboratories, Westbrook, ME, USA. Overview Production Red blood cells (RBC) are produced in the bone marrow. Numbers of circulating RBCs are affected by changes in plasma volume, rate of RBC destruction or loss, splenic contraction, erythropoietin (EPO) secretion, and the rate of bone marrow production. A normal PCV is maintained by an endocrine loop that involves generation and release of erythropoietin (EPO) from the kidney in response to renal hypoxia. Erythropoietin stimulates platelet production as well as red cell production. However, erythropoietin does not stimulate white blood cell (WBC) production. Erythropoiesis and RBC numbers are also affected by hormones from the adrenal cortex, thyroid, ovary, testis, and anterior pituitary. Destruction Red cells have a finite circulating lifespan. In dogs, the average normal red cell circulates approximately 100 days. -
![Anormal Rbc in Peripheral Blood. [Repaired].Pdf](https://docslib.b-cdn.net/cover/4277/anormal-rbc-in-peripheral-blood-repaired-pdf-544277.webp)
Anormal Rbc in Peripheral Blood. [Repaired].Pdf
1. Acanthocyte 2. Burr-cell 3. Microcyte 1. Basophilic Normoblast 2. Polychromatic Normoblast 3. Pycnotic Normoblast 4. Plasmocyte 5. Eosinophil 6. Promyelocyte 1. Macrocyte 2. Elliptocyte 1. Microcyte 2. Normocyte 1. Polychromatic Erythrocyte 2. Acanthocyte 3. Elliptocyte 1. Polychromatic Normoblast 2. Pycnotic Normoblast 3. Neutrophil Myelocyte 4. Neutrophil Metamyelocyte 1. Schistocyte 2. Microcyte BASOPHILIC ( EARLY ) NORMOBLASTS Basophilic Erythroblast Basophilic Stippling, Blood smear, May-Giemsa stain, (×1000) CABOT'S RINGS Drepanocyte Elliptocyte Erythroblast ERYTHROBLAST in the blood Howell-jolly body Hypo chromic LACRYMOCYTES Leptocyte Malaria, Blood smear, May-Giemsa stain, ×1000 MICROCYTES Orthochromatic erythroblast Pappen heimer Bodies & 1. Schistocyte 2. Elliptocyte 3. Acanthocyte POIKILOCYTOSIS Polychromatic Erythroblast Pro Erytroblast Proerythroblasts Reticulocyte Rouleaux SICKLE CELLS Sickle cell Spherocyte Spherocyte Spherocyte SPHEROCYTES STOMATOCYTES Target Cells Tear Drop Cell, Blood smear, May-Giemsa stain, x1000 Anulocyte 1. Burr-cell 2. Elliptocyte 1. Macrocyte 2. Microcyte 3. Elliptocyte 4. Schistocyte 1. Ovalocyte 2. Lacrymocyte 3. Target cell 1. Polychromatic Erythrocyte 2. Basophilic Stippling 1. Proerythroblast 2. Basophilic Erythroblast 3. Intermediate Erythroblast 4. Late Erythroblast 5. Monocyte 6. Lymphocyte 1. Target-cell 2. Elliptocyte 3. Acanthocyte 4. Stomatocyte 5. Schistocyte 6. Polychromatophilic erythrocyte. 1.Pro erythroblast 2.Basophilic normoblast 3.Polychromatic normoblast 4.Pycnotic normoblast -
Complete Blood Count in Primary Care
Complete Blood Count in Primary Care bpac nz better medicine Editorial Team bpacnz Tony Fraser 10 George Street Professor Murray Tilyard PO Box 6032, Dunedin Clinical Advisory Group phone 03 477 5418 Dr Dave Colquhoun Michele Cray free fax 0800 bpac nz Dr Rosemary Ikram www.bpac.org.nz Dr Peter Jensen Dr Cam Kyle Dr Chris Leathart Dr Lynn McBain Associate Professor Jim Reid Dr David Reith Professor Murray Tilyard Programme Development Team Noni Allison Rachael Clarke Rebecca Didham Terry Ehau Peter Ellison Dr Malcolm Kendall-Smith Dr Anne Marie Tangney Dr Trevor Walker Dr Sharyn Willis Dave Woods Report Development Team Justine Broadley Todd Gillies Lana Johnson Web Gordon Smith Design Michael Crawford Management and Administration Kaye Baldwin Tony Fraser Kyla Letman Professor Murray Tilyard Distribution Zane Lindon Lyn Thomlinson Colleen Witchall All information is intended for use by competent health care professionals and should be utilised in conjunction with © May 2008 pertinent clinical data. Contents Key points/purpose 2 Introduction 2 Background ▪ Haematopoiesis - Cell development 3 ▪ Limitations of reference ranges for the CBC 4 ▪ Borderline abnormal results must be interpreted in clinical context 4 ▪ History and clinical examination 4 White Cells ▪ Neutrophils 5 ▪ Lymphocytes 9 ▪ Monocytes 11 ▪ Basophils 12 ▪ Eosinophils 12 ▪ Platelets 13 Haemoglobin and red cell indices ▪ Low haemoglobin 15 ▪ Microcytic anaemia 15 ▪ Normocytic anaemia 16 ▪ Macrocytic anaemia 17 ▪ High haemoglobin 17 ▪ Other red cell indices 18 Summary Table 19 Glossary 20 This resource is a consensus document, developed with haematology and general practice input. We would like to thank: Dr Liam Fernyhough, Haematologist, Canterbury Health Laboratories Dr Chris Leathart, GP, Christchurch Dr Edward Theakston, Haematologist, Diagnostic Medlab Ltd We would like to acknowledge their advice, expertise and valuable feedback on this document. -

Red Blood Cell Morphology in Patients with Β-Thalassemia Minor
J Lab Med 2017; 41(1): 49–52 Short Communication Carolin Körber, Albert Wölfler, Manfred Neubauer and Christoph Robier* Red blood cell morphology in patients with β-thalassemia minor DOI 10.1515/labmed-2016-0052 Keywords: β-thalassemia minor; erythrocytes; red blood Received July 11, 2016; accepted October 20, 2016; previously published cells; red blood cell morphology. online December 10, 2016 Abstract In β-thalassemias, the examination of a peripheral blood (PB) smear may provide relevant clues to initial diagnosis. Background: A systematic analysis of the occurrence of Complete laboratory investigation consists of the determina- red blood cell (RBC) abnormalities in β-thalassemia minor tion of the complete blood count, assessment of red blood has not been performed to date. This study aimed to iden- cell (RBC) morphology, high performance liquid chroma- tify and quantify the frequency of RBC abnormalities in tography (HPLC), hemoglobin electrophoresis and, where patients with β-thalassemia minor. necessary, DNA analysis [1]. Especially in the clinically Methods: We examined blood smears of 33 patients with severe forms referred to as β-thalassemia major and interme- β-thalassemia minor by light microscopy for the occur- dia, RBC abnormalities are often markedly apparent [2]. In rence of 15 defined RBC abnormalities. In the case of posi- β-thalassemia minor, also called β-thalassemia trait, the car- tivity, the abnormal cells/20 high power fields (HPF) at riers are usually clinically asymptomatic, showing persistent 1000-fold magnification were counted. microcytosis and hypochromia or mild microcytic anemia [1, Results: Anisocytosis, poikilocytosis and target cells 3]. The PB smear may show microcytosis, hypochromia and, (median 42/20 HPF) were observed in all, and ovalocytes infrequently, poikilocytosis [2]. -

Red Blood Disorders Anemia Med.Pdf 14.44MB
Lviv National Medical University Department of pathological physiology PATHOLOGY OF RED BLOOD PhD. Sementsiv N.G. In norm The number of erythrocytes: in female - 3,9-4,7·1012/l in male - 4,5-5,0·1012/l Hemoglobin in female - 120-140g/l in male - 140-160g/l Color index(CI) - 0,85-1,15 Globular value = 3 x Hb / the first 3 figures of erythrocytes. Reticulocytes - 0.5-2%, 0,5-2%0 changes in total blood volume normovolemia hypovolemia hypervolemia simple (Ht - norm), polycitemia (Ht > 0,48), olygocytemia (Ht < 0,36). A) Norm B) acut anaemia б) acute hemorrhage г) hydremia Pathological forms of erythrocytes regenerative degenerative cell pathologic regeneration Regenerative forms reticulocytes залежно від зрілості розрізняють: (Зернисті) Stippling (Сітчасті) Mesh Norm in a blood reticulocytes - 0,2–2,0%. Regenerative forms Basophiles substantial erythrocytes - cytoplasm remains basophilic normo blast. Polychromatophil erythrocytes (polychromasia, polychromatophilia ) – erythrocytes with basophiles substantial ( blue cells) indicates increased RBC production by the marrow Qualitative (degenerative) changes of red blood cell - poikilocytosis - different shape of erythrocytes; - anisocytosis - different size of erythrocytes; - anisochromia - different saturation of red blood cells by hemoglobin Degenerative forms Anisocytosis present in a blood different forms erythrocytes » normocyte (7,01–8,0 мкм) » microcyte(6,9–5,7 мкм) » macrocyte(8,1–9,35 мкм) » megalocyte (10–15 мкм) Degenerative forms Poikilocytosis present in a blood pictures erythrocytes different forms : elongate form , oval, ellipsoid and os. ОVAlOCYTE( ELLIPTOCYTE) – 5% all blood. Pathological cells regenerate Megaloblast mehaloblastyc cell type hematopoiesis Megaloblast oval cells in the diameter of 1,5- 2,0 times larger than normal erythrocytes is the final stage mehaloblastyc hematopoiesis. -

Cambridge University Press 978-0-521-51426-2 — Anemia with Online Resource Edited by Edward J
Cambridge University Press 978-0-521-51426-2 — Anemia with Online Resource Edited by Edward J. Benz, Jr. , Nancy Berliner , Fred J. Schiffman Index More Information Index aAA. See acquired aplastic clinical significance of, 187 through RBC transfusion, anemia of inflammation, 169 anemia dementia, 187 175 anemia of prematurity, 34 abdomen, physical examination epidemiology of, 185–6 with rhEPO, 172, 174–7 anemias. See also clinical of, 32 inflammation and, 186–7 pathogenesis of, 172–3 approaches; specific ABO incompatibility, 36 hepcidin levels, 186 pathophysiology, 198 anemias HSCT and, 181 leptin protein, 186 therapy-related toxicity causes of, xi, 1 acanthocytes, 27 MIF, 186 grading, 172 through blood loss, 24 ACD. See anemia of chronic TNF-α, 186 transfusion, 175 classification of, 24 inflammation vitamin D deficiency, 186–7 anemia of chronic inflammation through MCV, 24 ACI. See anemia of chronic management strategies, 187–8 (ACI) clinical definitions of, xi, 23 inflammation prevalence of, 185 cancer and, 172 comorbidities with, xi acquired aplastic anemia (aAA), aHUS. See atypical hemolytic case study for, 153–4 defined, 172, 185 128–32. See also uremic syndrome clinical presentation, 152–3, diagnostic approach, 23–4 paroxysmal nocturnal AIHA. See autoimmune 194 through bone marrow hemoglobinuria hemolytic anemia critical illness with, 151 aspirate specimens, 24 bone marrow with, 128 alcohol use, non-megaloblastic cytokines, 196 through peripheral blood clinical presentation of, 129 macrocytic anemias diagnostic evaluation, 153, smears, 24–5, 29 clonal hematopoiesis and, 129 from, 63 194–5 future approaches to, 230–3 diagnostic evaluation of, alemtuzumab, 134 epidemiology, 150–1 hemoglobin deficiency in, xi 129–30 ALI. -
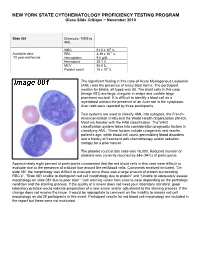
10 11 Cyto Slides 81-85
NEW YORK STATE CYTOHEMATOLOGY PROFICIENCY TESTING PROGRAM Glass Slide Critique ~ November 2010 Slide 081 Diagnosis: MDS to AML 9 WBC 51.0 x 10 /L 12 Available data: RBC 3.39 x 10 /L 72 year-old female Hemoglobin 9.6 g/dL Hematocrit 29.1 % MCV 86.0 fL Platelet count 16 x 109 /L The significant finding in this case of Acute Myelogenous Leukemia (AML) was the presence of many blast forms. The participant median for blasts, all types was 88. The blast cells in this case (Image 081) are large, irregular in shape and contain large prominent nucleoli. It is difficult to identify a blast cell as a myeloblast without the presence of an Auer rod in the cytoplasm. Auer rods were reported by three participants. Two systems are used to classify AML into subtypes, the French- American-British (FAB) and the World Health Organization (WHO). Most are familiar with the FAB classification. The WHO classification system takes into consideration prognostic factors in classifying AML. These factors include cytogenetic test results, patient’s age, white blood cell count, pre-existing blood disorders and a history of treatment with chemotherapy and/or radiation therapy for a prior cancer. The platelet count in this case was 16,000. Reduced number of platelets was correctly reported by 346 (94%) of participants. Approximately eight percent of participants commented that the red blood cells in this case were difficult to evaluate due to the presence of a bluish hue around the red blood cells. Comments received included, “On slide 081 the morphology was difficult to evaluate since there was a large amount of protein surrounding RBC’s”, “Slide 081 unable to distinguish red cell morphology due to protein” and “Unable to adequately assess morphology on slide 081 due to poor stain”. -

Pathological Physiology
PATHOLOGICAL PHYSIOLOGY PRACTICAL PART Student _____________________________ Group number ________________________ Teacher _____________________________ Minsk BSMU 2015 МИНИСТЕРСТВО ЗДРАВООХРАНЕНИЯ РЕСПУБЛИКИ БЕЛАРУСЬ БЕЛОРУССКИЙ ГОСУДАРСТВЕННЫЙ МЕДИЦИНСКИЙ УНИВЕРСИТЕТ КАФЕДРА ПАТОЛОГИЧЕСКОЙ ФИЗИОЛОГИИ ПАТОЛОГИЧЕСКАЯ ФИЗИОЛОГИЯ PATHOLOGICAL PHYSIOLOGY Практикум 4-е издание Минск БГМУ 2015 2 УДК 616-092.18(811.111)-054.6(076.5) (075.8) ББК 52.52 (81.2 Англ-923) П20 Рекомендовано Научно-методическим советом университета в качестве практикума 16.09.2015 г., протокол № 1 Авторы: Ф. И. Висмонт, В. А. Касап, С. А. Жадан, А. А. Кривчик, Е. В. Леонова, Т. В. Короткевич, Л. С. Лемешонок, А. В. Чантурия, Т. А. Афанасьева, В. Ю. Перетятько, О. Г. Шуст, Н. А. Степанова, К. Н. Грищенко, Э. Н. Кучук, Д. М. Попутников, Е. В. Меленчук Рецензенты: член-корр. НАН Беларуси, д-р мед. наук, проф. каф. нормальной физиологии Л. М. Лобанок; д-р мед. наук, проф. каф. патологической анатомии М. К. Недзьведзь Патологическая физиология = Pathological physiology : практикум / Ф. И. Вис- П20 монт [и др.]. – 4-е изд. – Минск : БГМУ, 2015. – 191 с. ISBN 978-985-567-304-1. Содержит описания и протоколы оформления лабораторных работ по основным разделам кур- са патофизиологии. Представлена информация по следующим разделам: патофизиология системы крови, нарушение сердечного ритма, кислотно-основного состояния, наследственности и изменчи- вости организма. Первое издание вышло в 2010 году. Предназначен для студентов 2–3-го курсов медицинского факультета иностранных учащихся для самостоятельной подготовки к занятиям, выполнения и оформления лабораторных работ по предмету. УДК 616-092.18(811.111)-054.6(076.5) (075.8) ББК 52.52 (81.2 Англ-923) ISBN 978-985-567-304-1 ã УО «Белорусский государственный медицинский университет», 2015 3 SECTION I GENERAL NOSOLOGY LESSON 1. -

Research Article
z Available online at http://www.journalcra.com INTERNATIONAL JOURNAL OF CURRENT RESEARCH International Journal of Current Research Vol. 9, Issue, 12, pp.62497-62502, December, 2017 ISSN: 0975-833X RESEARCH ARTICLE INCLUSION BODIES - A REVIEW 1Dr. Mrunalini Mahesh Dadpe, 2, *Dr. Sourab Kumar, 3Dr. Mahesh Dadpe, 4Dr. Payoshnee Bhalinge Jadhav, 5Dr. Abhishek Jadhav and 6Dr. Shilpi Suman 1, 4Department of Oral Pathology, M A Rangoonwala Dental College, Aazam Campus, Camp, Pune 411001, India 2, 5, 6Department of oral Pathology, D.Y. Patil School of Dentistry, Nerul, Navi Mumbai 400706, India 3MIDSR Dental College, Vishwanathpuram, Amba Jogai Road, Latur 413531, India ARTICLE INFO ABSTRACT Article History: The word inclusion means incorporation. Inclusion bodies are nuclear or cytoplasmic aggregates of Received 16th September, 2017 stainable substances which are usually ‘proteins’. They typically represent sites of viral multiplication Received in revised form in bacteria or in a eukaryotic cell and usually consist of viral capsid proteins. Inclusion bodies are 17th October, 2017 typically identified within a cell by their different appearance. Accepted 25th November, 2017 Published online 27th December, 2017 Key words: Proteins, Viral, Types, Structures Copyright © 2017, Mrunalini Mahesh Dadpe et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Citation: Dr. Mrunalini Mahesh Dadpe, Dr. Sourab Kumar, Dr. Mahesh Dadpe, Dr. Payoshnee Bhalinge Jadhav, Dr. Abhishek Jadhav and Dr. Shilpi Suman, 2017. “Inclusion Bodies - A Review”, International Journal of Current Research, 9, (12), 62497-62502. Classification INTRODUCTION Viral inclusion bodies Inclusion bodies (IB) can also be hallmarks of genetic diseases, as in the case of Neuronal Inclusion bodies in neural disorders, A. -
Blood Lab First Week
Blood Lab First Week This is a self propelled powerpoint study atlas of blood cells encountered in examination of the peripheral blood smear. It is a requirement of this course that you begin review of these slides with the first day of class in order to familiarize yourself with terms used in describing peripheral blood morphology. Laboratory final exam questions are derived directly from these slides. The content of these slides may duplicate slides (1-37) listed on the Hematopathology Website. You must familiarize yourself with both sets of slides in order to have the best learning opportunity. Before Beginning This Slide Review • Please read the Laboratory information PDF under Laboratory Resources file to learn about – Preparation of a blood smear – How to select an area of the blood smear for review of morphology and how to perform a white blood cell differential – Platelet estimation – RBC morphology descriptors • Anisocytosis • Poikilocytosis • Hypochromia • Polychromatophilia Normal blood smear. Red blood cells display normal orange pink cytoplasm with central pallor 1/3-1/2 the diameter of the red cell. There is mild variation in size (anisocytosis) but no real variation in shape (poikilocytosis). To the left is a lymphocyte. To the right is a typical neutrophil with the usual 3 segmentations of the nucleus. Med. Utah pathology Normal blood: thin area Ref 2 Normal peripheral blood smear. This field is good for exam of cell morphology, although there are a few minor areas of overlap of red cells. Note that most cells are well dispersed and the normal red blood cell central pallor is noted throughout the smear. -
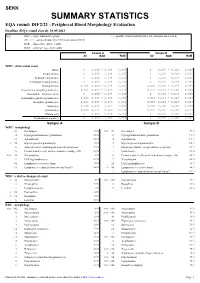
SUMMARY STATISTICS EQA Round: DIF2/21 - Peripheral Blood Morphology Evaluation Deadline (EQA Round Closed): 28.05.2021 Key: ELG
SEKK SUMMARY STATISTICS EQA round: DIF2/21 - Peripheral Blood Morphology Evaluation Deadline (EQA round closed): 28.05.2021 Key: ELG ... expert laboratories group > ... possible result (found by ELG, but consensus not reached) AV, >>> ... assigned value type CVE (consensus of ELG) RAR ... range of acceptable results RoM ... robust average of all results Sample A Sample B AV RAR RoM AV RAR RoM WBC - differential count Blasts 0 0,000 - 0,018 0,000 0 0,000 - 0,018 0,000 Promyelocytes 0 0,000 - 0,018 0,000 0 0,000 - 0,018 0,000 Neutrophil myelocytes 0 0,000 - 0,018 0,000 0 0,000 - 0,018 0,000 Neutrophil metamyelocytes 0 0,000 - 0,018 0,000 0 0,000 - 0,018 0,000 Neutrophil bars 0,0010,000 - 0,018 0,003 0,0020,000 - 0,018 0,004 Segmented neutrophil granulocytes 0,5550,483 - 0,625 0,579 0,5750,503 - 0,644 0,584 Eosinophils - immature forms 0 0,000 - 0,018 0,000 0 0,000 - 0,018 0,000 Eosinophil segmented granulocytes 0,0690,039 - 0,115 0,060 0,0160,003 - 0,043 0,018 Basophilic granulocytes 0,0110,001 - 0,036 0,011 0,0070,000 - 0,028 0,007 Monocytes 0,0660,035 - 0,109 0,067 0,0640,035 - 0,109 0,059 Lymphocytes 0,2980,237 - 0,369 0,278 0,3310,265 - 0,400 0,326 Plasma cells 0 0,000 - 0,018 0,000 0 0,000 - 0,018 0,000 Erythroblasts (number) 0 0,000 - 2,000 0,000 0 0,000 - 3,000 0,002 Sample A Sample B WBC - morphology 23 No changes 15 % >>> 89 No changes 59 % 4 Hypergranulation/toxic granulation 2,6 % 2 Hypergranulation/toxic granulation 1,3 % 10 Agranulation 6,6 % 2 Agranulation 1,3 % > 78 Hypersegmented granulocytes 52 % 3 Hypersegmented granulocytes 2,0 % 6 Atypical/reactive/pathological monocytes/promono.