Which Predators Are Responsible for Faunal
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

African Mammals (Tracks)
L Gi'. M MM S C A POCKET NATURALISru GUIDE HOOFED MAMMALS Dik-Dik Madoqua spp . To 17 in. (43 cm) H Small antelope has a long, flexible snout. .,9-10 in. Common Hippopotamus Klipspringer Hippopotamus amphibius Oreatragus oreotroqus To 5 ft. (1.5 m) H To 2 ft. (60 cm) H Has dark 'tear stains' at the corner of the eyes. Downward-pointing hooves give the impression it walks on 'tiptoe'. Found in rocky habitats. White Black Steenbok Raphieerus eampestris 1 in. White Rhinoceros To 2 ft. (60 cm) H Large ears are striped inside. Ceratotherium simum Muzzle has a dark stripe. To 6 ft. (l.B m) H 9-10 in. t Has a square upper lip. The similar black rhinoceros has a , pointed, prehensile upper lip. t . eWhite '~'rBlack . Common Duiker " . Sylvieapra grimmia ". To 28 in. (70 cm) t1 t Has a prominent black 1 in. 'a: stripe on its snout. Inhabits woodlands Hyena lion and shrubby areas. t Forefoot , e+ Oribi 24-28 in. Ourebia ourebi African Elephant To 2 ft. (60 em) H Note short tail and black 1.5 in. Loxodonto africana Hind foot spot below ears. Inhabits To 14 ft. (4.2 m) H grassland savannas. Hind print is oval-shaped. t This guide provides simplified field reference to familiar animal tracks. It is important to note that tracks change depending on their age, the surface Hippopotamus they are made on, and the animal's gait (e.g., toes are often splayed when Springbok running). Track illustrations are ordered by size in each section and are not Antidorcas marsupialis To 30 in. -

Karoo Large Mammals Check List Note
Karoo Large Mammals Check List Note: Many of the mammals included on this list can only be seen in formal conservation areas such as the National Parks and Provincial Nature Reserves. Mammal Scientific Name Location Date South African hedgehog Erinaceus frontalis Vervet monkey Cercopithecus pygerythrus Chacma baboon Papio ursinus Aardwolf Proteles cristatus Brown hyena Parahyaena brunnea Cape wild cat (African wild cat) Felis lybica Black-footed cat (Small spotted cat) Felis nigripes Caracal Caracal caracal Cheetah Acinonyx jubatus Leopard Panthera pardus Lion Panthera leo Bat-eared fox Otocyon megalotis Cape fox Vulpes chama Blackbacked jackal Canis mesomelas Cape clawless otter Aonyx capensis Striped polecat Ictonyx striatus African striped weasel Poecilogale albinucha Honey badger Mellivora capensis Small-spotted genet Genetta genetta Large-spotted genet Genetta tigrina Water mongoose Atilax paludinosus Cape grey mongoose Galerella pulverulenta Yellow mongoose Cynctis penicillata Small grey mongoose Galerella pulverulenta Suricate (Meerkat) Suricata suricatta Rock dassie (hyrax) Procavia capensis www.karoo-southafrica.com Mammal Scientific Name Location Date Porcupine Hystrix africaeaustralis Ground squirrel Xerus inauris Springhare Pedetes capensis Scrub hare Lepus saxatilis Cape hare Lepus capensis Natal red rock rabbit Pronolagus crassicaudatis Smith’s red rock rabbit Pronolagus rupestris Riverine rabbit Bunolagus monticularis Antbear Orycteropus afer Bushpig Potamochoerus larvatus Cape mountain zebra Equus zebra zebra Burchell’s -

GREVY's ZEBRA Equus Grevyi Swahili Name
Porini Camps Mammal Guide By Rustom Framjee Preface This mammal guide provides some interesting facts about the mammals that are seen by guests staying at Porini Camps. In addition, there are many species of birds and reptiles which are listed separately from this guide. Many visitors are surprised at the wealth of wildlife and how close you can get to the animals without disturbing them. Because the camps operate on a low tourist density basis (one tent per 700 acres) the wildlife is not ‘crowded’ by many vehicles and you can see them in a natural state - hunting, socialising, playing, giving birth and fighting to defend their territories. Some are more difficult to see than others, and some can only be seen when you go on a night drive. All Porini camps are unfenced and located in game rich areas and you will see much wildlife even in and around the camps. The Maasai guides who accompany you on all game drives and walks are very well trained and qualified professional guides. They are passionate and enthusiastic about their land and its wildlife and really want to show you as much as they can. They have a wealth of knowledge and you are encouraged to ask them more about what you see. They know many of the animals individually and can tell you stories about them. If you are particularly interested in something, let them know and they will try to help you see it. While some facts and figures are from some of the references listed, the bulk of information in this guide has come from the knowledge of guides and camp staff. -

Animals Tested for Rabies Virus — Washington State, 1988–2018
Animals Tested for Rabies Virus — Washington State, 1988–2018 Year Bat Cat Dog Ferret Raccoon Skunk Rodent Lagomorph Other Wild Other Domestic Total Total Positive Total Positive Total Positive Total Positive Total Positive Total Positive Total Positive Total Positive Total Positive Total Positive Total Positive 1988 69 4 165 110 15 16 3 12 2 5 3 400 4 1989 102 9 124 91 20 9 4 8 1 9 4 372 9 1990 63 4 104 82 5 7 5 5 1 14 4 290 4 1991 90 9 105 96 13 8 3 13 0 19 2 349 9 1992 73 6 132 90 16 14 2 12 0 14 6 1* 359 7 1993 68 1 122 95 8 4 8 16 2 10 13 346 1 1994 58 14 105 90 7 4 3 15 0 16 14 1^ 312 15 1995 263 15 140 114 12 8 1 23 3 15 18 597 15 1996 257 13 104 101 8 9 2 14 3 20 12 530 13 1997 780 51 155 118 7 17 4 15 2 18 11 1,127 51 1998 447 27 126 109 8 11 1 6 0 19 16 743 27 1999 334 25 103 71 3 11 3 8 1 14 13 561 25 2000 330 23 105 60 1 2 4 6 1 9 4 522 23 2001 263 22 111 93 2 3 1 8 0 4 5 490 22 2002 186 12 99 1 53 7 2 2 9 1 8 9 376 13 2003 229 23 137 72 0 11 1 4 1 9 10 474 23 2004 311 20 141 70 3 13 6 11 0 6 10 571 20 2005 245 15 132 66 3 12 2 5 1 10 4 480 15 2006 273 15 105 70 4 13 1 2 1 8 5 482 15 2007 315 22 132 97 1 16 3 5 0 9 3 581 22 2008 337 17 143 76 1 10 2 5 1 9 11 595 17 2009 311 14 133 90 1 12 5 4 1 7 9 573 14 2010 200 14 103 63 0 14 1 6 1 9 10 407 14 2011 204 11 87 51 1 9 1 2 0 8 5 368 11 2012 221 9 98 54 2 7 0 4 0 7 9 402 9 2013 284 12 80 65 0 13 0 3 0 5 9 459 12 2014 276 15 75 53 0 12 0 1 1 6 11 435 15 2015 305 9 95 1 49 0 8 2 8 0 11 7 485 10 2016 298 20 108 44 0 5 0 4 1 3 3 466 20 2017 376 22 81 48 0 8 1 4 0 2 5 525 22 2018 531 40 84 44 0 4 0 2 0 2 8 675 40 2019 255 9 65 23 0 2 0 2 0 3 7 357 9 Total 8,354 522 3,599 2 2,408 0 148 0 294 0 71 0 242 0 25 0 308 0 260 2 15,709 526 Notes Lagomorphs include: rabbit, hare, and pika. -

University of Florida Thesis Or Dissertation Formatting
UNDERSTANDING CARNIVORAN ECOMORPHOLOGY THROUGH DEEP TIME, WITH A CASE STUDY DURING THE CAT-GAP OF FLORIDA By SHARON ELIZABETH HOLTE A DISSERTATION PRESENTED TO THE GRADUATE SCHOOL OF THE UNIVERSITY OF FLORIDA IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY UNIVERSITY OF FLORIDA 2018 © 2018 Sharon Elizabeth Holte To Dr. Larry, thank you ACKNOWLEDGMENTS I would like to thank my family for encouraging me to pursue my interests. They have always believed in me and never doubted that I would reach my goals. I am eternally grateful to my mentors, Dr. Jim Mead and the late Dr. Larry Agenbroad, who have shaped me as a paleontologist and have provided me to the strength and knowledge to continue to grow as a scientist. I would like to thank my colleagues from the Florida Museum of Natural History who provided insight and open discussion on my research. In particular, I would like to thank Dr. Aldo Rincon for his help in researching procyonids. I am so grateful to Dr. Anne-Claire Fabre; without her understanding of R and knowledge of 3D morphometrics this project would have been an immense struggle. I would also to thank Rachel Short for the late-night work sessions and discussions. I am extremely grateful to my advisor Dr. David Steadman for his comments, feedback, and guidance through my time here at the University of Florida. I also thank my committee, Dr. Bruce MacFadden, Dr. Jon Bloch, Dr. Elizabeth Screaton, for their feedback and encouragement. I am grateful to the geosciences department at East Tennessee State University, the American Museum of Natural History, and the Museum of Comparative Zoology at Harvard for the loans of specimens. -
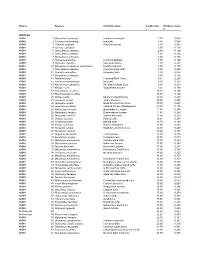
PDF File Containing Table of Lengths and Thicknesses of Turtle Shells And
Source Species Common name length (cm) thickness (cm) L t TURTLES AMNH 1 Sternotherus odoratus common musk turtle 2.30 0.089 AMNH 2 Clemmys muhlenbergi bug turtle 3.80 0.069 AMNH 3 Chersina angulata Angulate tortoise 3.90 0.050 AMNH 4 Testudo carbonera 6.97 0.130 AMNH 5 Sternotherus oderatus 6.99 0.160 AMNH 6 Sternotherus oderatus 7.00 0.165 AMNH 7 Sternotherus oderatus 7.00 0.165 AMNH 8 Homopus areolatus Common padloper 7.95 0.100 AMNH 9 Homopus signatus Speckled tortoise 7.98 0.231 AMNH 10 Kinosternon subrabum steinochneri Florida mud turtle 8.90 0.178 AMNH 11 Sternotherus oderatus Common musk turtle 8.98 0.290 AMNH 12 Chelydra serpentina Snapping turtle 8.98 0.076 AMNH 13 Sternotherus oderatus 9.00 0.168 AMNH 14 Hardella thurgi Crowned River Turtle 9.04 0.263 AMNH 15 Clemmys muhlenbergii Bog turtle 9.09 0.231 AMNH 16 Kinosternon subrubrum The Eastern Mud Turtle 9.10 0.253 AMNH 17 Kinixys crosa hinged-back tortoise 9.34 0.160 AMNH 18 Peamobates oculifers 10.17 0.140 AMNH 19 Peammobates oculifera 10.27 0.140 AMNH 20 Kinixys spekii Speke's hinged tortoise 10.30 0.201 AMNH 21 Terrapene ornata ornate box turtle 10.30 0.406 AMNH 22 Terrapene ornata North American box turtle 10.76 0.257 AMNH 23 Geochelone radiata radiated tortoise (Madagascar) 10.80 0.155 AMNH 24 Malaclemys terrapin diamondback terrapin 11.40 0.295 AMNH 25 Malaclemys terrapin Diamondback terrapin 11.58 0.264 AMNH 26 Terrapene carolina eastern box turtle 11.80 0.259 AMNH 27 Chrysemys picta Painted turtle 12.21 0.267 AMNH 28 Chrysemys picta painted turtle 12.70 0.168 AMNH 29 -

20 April 09 FEIR Bravo 3 MM Printing
May 2009 84 10637 Sensitivities The ratings mentioned above in the site description are derived from the level of conservation of that specific vegetation type. This is useful for an overview perspective, but for detailed sensitivities the focus moves to red/orange data species. Due to the endangered status of the plants, their specific occurrence is kept confidential by the GDACE. For guidance, the department has issued the Conservation Plan (CPlan) data which provides an indication as to the locality of red/orange data fauna or flora. Figure 52 illustrates the areas identified by CPlan as being sensitive. These areas have been investigated in detail for sensitive flora. It was found that the areas highlighted in Figure 52 are mostly wetlands, the Bronkhorstspruit Dam and ridges. All of these areas could provide habitat to sensitive species. 7.1.9 Fauna Data Collection A literature review of the faunal species that could occur in the area was conducted. C-Plan data provided from the Mpumalanga provincial department was used to conduct a desktop study of the area. This data consists of terrestrial and aquatic components, ratings provide an indication as to the importance of the area with respect to biodiversity. Additionally, all fauna were noted during the site visit conducted on the 10th-14th March and 18th – 20th November 2008. Regional Description As a consequence of mining and farming in the area, it appears that only small animals are to be found at the site. Small mammals known to occur in the area include hedgehog, rabbits, polecat, meerkat and the ubiquitous rats and mice. -

Prey Selection by Caracal in the Kgalagadi Transfrontier Park
Prey selection by caracal in the Kgalagadi Transfrontier Park H.I.A.S. Melville1*, J. du P. Bothma1 & M.G.L. Mills2 1Centre for Wildlife Management, University of Pretoria, Pretoria, 0002 South Africa 2SAN Parks, Endangered Wildlife Trust and Mammal Research Institute, University of Pretoria, Private Bag X402, Skukuza, 1350 South Africa Received 29 August 2003. Accepted 4 March 2004 In the Kgalagadi Transfrontier Park, 116 caracal (Caracal caracal) scat samples were col- lected and 327 attempted hunts were reconstructed from spoor-tracking. The data were analysed to establish the prey use of caracals in the Kgalagadi Transfrontier Park and to study the extent to which caracals use small stock by moving into the adjacent farmland in Namibia. It was found that the primary prey resource was small mammals, the vast majority of which were rodents, including springhare (Pedetes capensis). Larger prey animals included steenbok (Raphicerus campestris) and smaller carnivores up to the size of a black-backed jackal (Canis mesomelas). Birds were an abundant prey resource, especially the larger ground-roosting species. Invertebrate remains were found in a large proportion of the scats, indicating that they were commonly used as a source of food. Domestic livestock remains were identified in eight of the scat samples and the temporal distribution of these indicated an increased use of domestic livestock by caracals in the cold season. Key words: caracal, Felidae, livestock, scat analysis, small mammals, spoor-tracking. INTRODUCTION where stock farms border on the Kgalagadi The survival of any predator is directly related Transfrontier Park. Although widely condemned to the quality and quantity of its diet. -

Small Carnivore Conservation Action Plan
Durant, S. M., Foley, C., Foley, L., Kazaeli, C., Keyyu, J., Konzo, E., Lobora, A., Magoma, N., Mduma, S., Meing'ataki, G. E. O., Midala, B. D. V. M., Minushi, L., Mpunga, N., Mpuya, P. M., Rwiza, M., and Tibyenda, R. The Tanzania Small Carnivore Conservation Action Plan. Durant, S. M., De Luca, D., Davenport, T. R. B., Mduma, S., Konzo, S., and Lobora, A. Report: 162-269. 2009. Arusha, Tanzania Wildlife Research Institute. Keywords: 1TZ/abundance/action plan/caracal/Caracal caracal/conservation/conservation action plan/distribution/ecology/Felis silvestris/Leptailurus serval/serval/wildcat Abstract: This report covers the proceedings of the First Tanzania Small Carnivore Conservation Action Plan Workshop held at TAWIRI on 19th-21st April 2006. The workshop brought together key stakeholders to assess existing information and establish a consensus on priorities for research and conservation for 28 species of small to medium carnivore in Tanzania (excluding cheetah, wild dogs, aardwolf, spotted hyaena, striped hyaena, leopard and lion, all of which were covered in other workshops). Recent records were used to confirm the presence of 27 of these species in Tanzania. These were three species of cats or felids: serval (Leptailurus serval); caracal (Caracal caracal) and wild cat (Felis silvestris). Five mustelids: Cape clawless otter (Aonyx capensis); spotted-necked otter (Hydrictis maculicollis); honey badger (Mellivora capensis); striped weasel (Poecilogale albinucha); and zorilla (Ictonyx striatus). Four canids: bat-eared fox (Otocyon megalotis); black-backed jackal (Canis mesomelas); golden jackal (Canis aureus); side-striped jackal (Canis adustus). Four viverrids: common genet (Genetta genetta); large-spotted genet (Genetta maculata); servaline genet (Genetta servalina); and African civet (Viverra civettina). -

Standards for Caniform Sanctuaries
Global Federation of Animal Sanctuaries Standards For Caniform Sanctuaries Version: February, 2018 ©2012 Global Federation of Animal Sanctuaries Global Federation of Animal Sanctuaries – Standards for Caniform Sanctuaries Table of Contents INTRODUCTION...................................................................................................................................... 1 GFAS PRINCIPLES ................................................................................................................................................... 1 ANIMALS COVERED BY THESE STANDARDS ............................................................................................................ 1 STANDARDS UPDATES……………………………………………………………………………………………………………………………………….. 4 CANIFORM STANDARDS ........................................................................................................................................ 4 CANIFORM HOUSING ............................................................................................................................ 5 H-1. Types of Space and Size ..................................................................................................................................... 5 H-2. Containment ...................................................................................................................................................... 7 H-3. Ground and Plantings ........................................................................................................................................ -

Ebook Download Seals and Sea Lions Ebook
SEALS AND SEA LIONS PDF, EPUB, EBOOK John Crossingham,Bobbie Kalman | 32 pages | 28 Feb 2006 | Crabtree Publishing Co,Canada | 9780778713234 | English | New York, Canada Seals and sea lions - CodyCross Answers Cheats and Solutions In one legend, seals, whales and other marine mammals were formed from her severed fingers. The Greeks associated them with both the sea and sun and were considered to be under the protection of the gods Poseidon and Apollo. Pinnipeds can be found in facilities around the world, as their large size and playfulness make them popular attractions. Zoologist Georges Cuvier noted during the 19th century that wild seals show considerable fondness for humans and stated that they are second only to some monkeys among wild animals in their easily tamability. Francis Galton noted in his landmark paper on domestication that seals were a spectacular example of an animal that would most likely never be domesticated despite their friendliness and desire for comfort due to the fact that they serve no practical use for humans. Some modern exhibits have rocky backgrounds with artificial haul-out sites and a pool, while others have pens with small rocky, elevated shelters where the animals can dive into their pools. More elaborate exhibits contain deep pools that can be viewed underwater with rock-mimicking cement as haul-out areas. The most common pinniped species kept in captivity is the California sea lion as it is both easy to train and adaptable. Other species popularly kept include the grey seal and harbor seal. Larger animals like walruses and Steller sea lions are much less common. -

List of Taxa for Which MIL Has Images
LIST OF 27 ORDERS, 163 FAMILIES, 887 GENERA, AND 2064 SPECIES IN MAMMAL IMAGES LIBRARY 31 JULY 2021 AFROSORICIDA (9 genera, 12 species) CHRYSOCHLORIDAE - golden moles 1. Amblysomus hottentotus - Hottentot Golden Mole 2. Chrysospalax villosus - Rough-haired Golden Mole 3. Eremitalpa granti - Grant’s Golden Mole TENRECIDAE - tenrecs 1. Echinops telfairi - Lesser Hedgehog Tenrec 2. Hemicentetes semispinosus - Lowland Streaked Tenrec 3. Microgale cf. longicaudata - Lesser Long-tailed Shrew Tenrec 4. Microgale cowani - Cowan’s Shrew Tenrec 5. Microgale mergulus - Web-footed Tenrec 6. Nesogale cf. talazaci - Talazac’s Shrew Tenrec 7. Nesogale dobsoni - Dobson’s Shrew Tenrec 8. Setifer setosus - Greater Hedgehog Tenrec 9. Tenrec ecaudatus - Tailless Tenrec ARTIODACTYLA (127 genera, 308 species) ANTILOCAPRIDAE - pronghorns Antilocapra americana - Pronghorn BALAENIDAE - bowheads and right whales 1. Balaena mysticetus – Bowhead Whale 2. Eubalaena australis - Southern Right Whale 3. Eubalaena glacialis – North Atlantic Right Whale 4. Eubalaena japonica - North Pacific Right Whale BALAENOPTERIDAE -rorqual whales 1. Balaenoptera acutorostrata – Common Minke Whale 2. Balaenoptera borealis - Sei Whale 3. Balaenoptera brydei – Bryde’s Whale 4. Balaenoptera musculus - Blue Whale 5. Balaenoptera physalus - Fin Whale 6. Balaenoptera ricei - Rice’s Whale 7. Eschrichtius robustus - Gray Whale 8. Megaptera novaeangliae - Humpback Whale BOVIDAE (54 genera) - cattle, sheep, goats, and antelopes 1. Addax nasomaculatus - Addax 2. Aepyceros melampus - Common Impala 3. Aepyceros petersi - Black-faced Impala 4. Alcelaphus caama - Red Hartebeest 5. Alcelaphus cokii - Kongoni (Coke’s Hartebeest) 6. Alcelaphus lelwel - Lelwel Hartebeest 7. Alcelaphus swaynei - Swayne’s Hartebeest 8. Ammelaphus australis - Southern Lesser Kudu 9. Ammelaphus imberbis - Northern Lesser Kudu 10. Ammodorcas clarkei - Dibatag 11. Ammotragus lervia - Aoudad (Barbary Sheep) 12.