10 Mg/Ml Solution for Injection/Infusion Atracurium
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Use of Chemical Chelators As Reversal Agents for Drug
(19) TZZ_ _ZZZ_T (11) EP 1 210 090 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: A61K 31/724 (2006.01) A61K 31/194 (2006.01) 18.06.2014 Bulletin 2014/25 A61P 39/04 (2006.01) (21) Application number: 00964006.1 (86) International application number: PCT/EP2000/007694 (22) Date of filing: 07.08.2000 (87) International publication number: WO 2001/012202 (22.02.2001 Gazette 2001/08) (54) USE OF CHEMICAL CHELATORS AS REVERSAL AGENTS FOR DRUG- INDUCED NEUROMUSCULAR BLOCK VERWENDUNG VON CHEMISCHEN CHELATOREN ZUR UMKEHRUNG VON PHARMAKOLOGISCH-INDUZIERTER NEUROMUSKULÄRER BLOCKIERUNG UTILISATION D’AGENTS CHIMIQUES CHELATANTS COMME AGENTS DE NEUTRALISATION DU BLOCAGE NEUROMUSCULAIRE PROVOQUE PAR DES MEDICAMENTS (84) Designated Contracting States: (56) References cited: AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU AU-A- 3 662 895 MC NL PT SE • B DESIRE: "Inactivaton of sarin and soman by (30) Priority: 13.08.1999 EP 99306411 cyclodextrins in vitro" EXPERIENTIA, vol. 43, no. 4, 1987, pages 395-397, XP000907287 (43) Date of publication of application: • B. DESIRE: "Interaction of soman with beta- 05.06.2002 Bulletin 2002/23 cyclodextrin" FUNDAMENTAL AND APPLIED TOXICOLOGY, vol. 7, no. 4, 1986, pages 647-657, (73) Proprietor: Merck Sharp & Dohme B.V. XP000911170 2031 BN Haarlem (NL) • C. MAY: "Development of a toxin-binding agent as a treatment for tunicamycinuracil toxicity: (72) Inventors: protection against tunicamycin poisoning of • BOM, Antonius, Helena, Adolf sheep" AUSTRALIAN VETERINARY JOURNAL, Ratho, Midlothian EH28 8NY (GB) vol. 76, no. -

Download Document
FAM001159-0001 intervals of 4-7 days to usual dose of 75-100 mg Dolmatil® (Sanofi-Synthelabo) ~ 100 rag/5 mL, thioridazine 100 rag/ per course and max. 4 injections; max. duration of daily according to response; CHILD not recom- Tablets, both scored, sulpiride 200 rag, net price ’ net price 300 mL = £7.14. Label: 2 treatment 2 weeks---if maintenance treatment mended 100-tabpack=£13.85;400mg(f/c), 100-tab ! fNote. These suspensions should not be diluted but the necessary change to an oral antipsychotic 2-3 Short-term adjunctive management of severe pack = £36¯29. Label: 2 .: :~t~a preparations may be mixed with each other to days after last injection, or to a longer acting anti- anxiety, 15-20rag daily in divided doses; max. Sulpltil® (Pharmacia) I~ ~0iovid¢ intermediate strengths psychotic depot injection given concomitantly 40 mg daily; CHILD not recommended Tablets, scored, sulpiride 200 rag. Net price 28-tab "ff~-~p, brown, thioridazine (as hydrochloride) with last injection of zuclopenthixol acetate; By deep intramuscular injection, psychoses, mania, pack = £4.29; 112-tab pack = £12.85. Label: 2 ~ff.~5"~mg/5 mL, net price 300 mL = £1.98. Label: 2 CHILD not recommended prochlorperazine mesilate 12.5-25 mg 2-3 times Sulpor® (Rosemont) IPoMI Clopi~ol Acuphase® (Lundbeck) daily; CHILD not recommended Oral solution, sugar-free, lemon- and aniseed-fla. LUOPERAZINE Injection (oily), zuclopenthixol acetate 50 mg/mL. By rectum in suppositories, psychoses, mania, the voured, sulpiride 200 mg/5 mL, net price 150 mL (’~’n~ications: see under Dose; anti-emetic (section Net price I-mL amp = £5.20; 2-mL amp = £10.03 equivalent of prochlorperazine maleate 25 mg 2- = £27.00. -
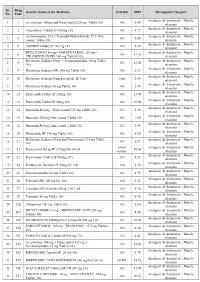
PMBJP Product.Pdf
Sr. Drug Generic Name of the Medicine Unit Size MRP Therapeutic Category No. Code Analgesic & Antipyretic / Muscle 1 1 Aceclofenac 100mg and Paracetamol 325 mg Tablet 10's 10's 8.00 relaxants Analgesic & Antipyretic / Muscle 2 2 Aceclofenac Tablets IP 100mg 10's 10's 4.37 relaxants Acetaminophen 325 + Tramadol Hydrochloride 37.5 film Analgesic & Antipyretic / Muscle 3 4 10's 8.00 coated Tablet 10's relaxants Analgesic & Antipyretic / Muscle 4 5 ASPIRIN Tablets IP 150 mg 14's 14's 2.70 relaxants DICLOFENAC 50 mg+ PARACETAMOL 325 mg+ Analgesic & Antipyretic / Muscle 5 6 10's 11.30 CHLORZOXAZONE 500 mg Tablets 10's relaxants Diclofenac Sodium 50mg + Serratiopeptidase 10mg Tablet Analgesic & Antipyretic / Muscle 6 8 10's 12.00 10's relaxants Analgesic & Antipyretic / Muscle 7 9 Diclofenac Sodium (SR) 100 mg Tablet 10's 10's 6.12 relaxants Analgesic & Antipyretic / Muscle 8 10 Diclofenac Sodium 25mg per ml Inj. IP 3 ml 3 ml 2.00 relaxants Analgesic & Antipyretic / Muscle 9 11 Diclofenac Sodium 50 mg Tablet 10's 10's 2.90 relaxants Analgesic & Antipyretic / Muscle 10 12 Etoricoxilb Tablets IP 120mg 10's 10's 33.00 relaxants Analgesic & Antipyretic / Muscle 11 13 Etoricoxilb Tablets IP 90mg 10's 10's 25.00 relaxants Analgesic & Antipyretic / Muscle 12 14 Ibuprofen 400 mg + Paracetamol 325 mg Tablet 10's 15's 5.50 relaxants Analgesic & Antipyretic / Muscle 13 15 Ibuprofen 200 mg film coated Tablet 10's 10's 1.80 relaxants Analgesic & Antipyretic / Muscle 14 16 Ibuprofen 400 mg film coated Tablet 10's 15's 3.50 relaxants Analgesic & Antipyretic -

(12) United States Patent (10) Patent N0.: US 7,265,099 B1 Born Et A1
US007265099B1 (12) United States Patent (10) Patent N0.: US 7,265,099 B1 Born et a1. (45) Date of Patent: *Sep. 4, 2007 (54) USE OF CHEMICAL CHELATORS AS Tarver, G. et al “2-O-Substituted cyclodextrins as reversal REVERSAL AGENTS FOR DRUG-INDUCED agents . ” Bioorg. Med. Chem. (2002) vol. 10, pp 1819-1827.* NEUROMUSCULAR BLOCK Zhang, M. “Drug-speci?c cyclodextrins . ” Drugs of the Future (2003) vol. 28, no 4, pp 347-354.* (75) Inventors: Antonius Helena Adolf Bom, Lee, C. “Structure, conformation, and action of neuromuscular Midlothian (GB); Alan William Muir, blocking drugs” Brit. J. Anesth. (2001) vol. 87, no 5, pp 755-769.* Lanark (GB); David Rees, Gothenburg B Desire: “Inactivation of sarin and soman by cyclodextrins in (SE) vitro” EXPERIENTIA, vol. 43, No. 4, 1987, pp. 395-397. B. Desire: “Interaction of soman with beta-cyclodextrin” Funda (73) Assignee: Organon N.V., Oss (NL) mental and Applied Toxicology, vol. 7, No. 4, 1986, pp. 647-657. ( * ) Notice: Subject to any disclaimer, the term of this C. May: “Development of a toxin-bindng agent as a treatment for patent is extended or adjusted under 35 tunicamycinuracil toxicity: protection against tunicamycin poison U.S.C. 154(b) by 0 days. ing of sheep” Australian Veterinary Journal, vol. 76, No. 11, 1998 pp. 752-756. This patent is subject to a terminal dis K. Uekama: “Effects of cyclodextrins on chlorpromaZine-induced claimer. haemolysis and nervous systems responses” J. Pharm. Pharmacol., vol. 33, No. 11, 1981, pp. 707-710. (21) Appl. No.: 10/049,393 T. Irie: “Protective mechanism of beta-cyclodextrin for the hemolysis induced With phenothiazine neuroleptics in vitro” J. -

New Zealand Data Sheet
NEW ZEALAND DATA SHEET 1. TRACRIUM (atracurium besilate 10 mg/mL injections (2.5 mL and 5.0 mL)) 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each 2.5 mL ampoule contains 25 mg atracurium besilate, each 5 mL ampoule contains 50 mg atracurium besilate. TRACRIUM 2.5 mL and 5.0 mL injections contain no preservative. 3. PHARMACEUTICAL FORM TRACRIUM injection is a clear, faintly yellow, sterile, aqueous solution in a glass ampoule containing 10 mg/mL atracurium besilate. 4. CLINICAL PARTICULARS 4.1 Therapeutic indications TRACRIUM is a highly selective, competitive or non-depolarising neuromuscular blocking agent which is used as an adjunct to general anaesthesia to enable tracheal intubation to be performed and to relax skeletal muscles during surgery or controlled ventilation, and to facilitate mechanical ventilation in Intensive Care Unit (ICU) patients. 4.2 Dose and method of administration Use in adults Injection TRACRIUM is administered by intravenous injection. The dosage range for adults is 0.3 to 0.6 mg/kg (depending on the duration of full block required) and will provide adequate relaxation for 15 to 35 minutes. Endotracheal intubation can usually be accomplished within 90 seconds from the intravenous injection of 0.5 to 0.6 mg/kg. Full block can be prolonged with supplementary doses of 0.1 to 0.2 mg/kg as required. Successive supplementary dosing does not give rise to accumulation of neuromuscular blocking effect. Spontaneous recovery from the end of full block occurs in about 35 minutes as measured by the restoration of the tetanic response to 95% of normal neuromuscular function. -

Pharmaceutical Appendix to the Harmonized Tariff Schedule
Harmonized Tariff Schedule of the United States Basic Revision 3 (2021) Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE Harmonized Tariff Schedule of the United States Basic Revision 3 (2021) Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 2 Table 1. This table enumerates products described by International Non-proprietary Names INN which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service CAS registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. -

Texts Adopted by the European Phamacopoeia Commission
© Pharmeuropa | Useful information | June 2015 1 List of texts adopted at the March 2015 session of the European Pharmacopoeia Commission NEW TEXTS GENERAL CHAPTERS 2.5.41. Methyl, ethyl and isopropyl benzenesulfonate in active substances 2.7.34. Assay of human C1-esterase inhibitor 5.19. Extemporaneous preparation of radiopharmaceuticals 5.21. Chemometric methods applied to analytical data MONOGRAPHS Vaccines for human use Haemophilus type b and meningococcal group C conjugate vaccine (2622) Radiopharmaceutical preparations and starting materials for radiopharmaceutical preparations Technetium (99mTc) oxidronate injection (2376) Herbal drugs and herbal drug preparations Aucklandia root (1797) Homoeopathic preparations Acidum picrinicum for homoeopathic preparations (2695) Histaminum for homoeopathic preparations (2671) Monographs Cisatracurium besilate (2763) Dienogest (2732) Entecavir monohydrate (2815) Gadobutrol monohydrate (2735) Gefitinib (2866) Human C1-esterase inhibitor (2818) Hydroxypropylcellulose, low-substituted (2083) Pregabalin (2777) Rabeprazole sodium (2868) 2 © Pharmeuropa | Useful information | June 2015 Rabeprazole sodium hydrate (2331) Ropinirole hydrochloride (2604) Sitagliptin phosphate monohydrate (2778) Sitagliptin tablets (2927) Sodium lauroylsarcosinate for external use (2542) Thiocolchicoside crystallised from ethanol (2896) Thiocolchicoside hydrate (2814) REVISED TEXTS GENERAL CHAPTERS 2.2.31. Electrophoresis 2.2.48. Raman spectroscopy 2.5.38. Methyl, ethyl and isopropyl methanesulfonate in active substances -

Troikaa Pharmaceuticals Ltd's Drugs List Anaesthetics
Troikaa Pharmaceuticals Ltd's Drugs List Anaesthetics Sr.No Product Name /Brand Name Strength/ Composition Presentation Dosage Form Choline Salicylate Solution equivalent to Choline Salicylate 8.7% w/w + 1 ALCINIL 10GM TUBE 1 TUBE TUBE BenzalkoniumChloride solution 0.01%w/w + Lignocaine Hcl 2% w/w Bupivacaine HCl Preservative 2 Free 0.5% Inj. 5 mg / ml 20 ml Vial Injection BUPIPTROY PF Bupivacaine HCl 0.5% Inj. 3 5 mg / ml 20 ml Vial Injection BUPITROY Bupivacaine HCl in Dextrose 4 Inj. 20 mg + 320 mg 5 X 4 ml Amp Injection BUPITROY-HEAVY 5 ml & 10 ml Midazolam Inj. a) 1 mg / ml 5 vial Injection BENZOSED b) 5 mg / ml 3 ml Amp Dexmedetomidine 50 mcg/0.5 6 DEXIT 50 1X0.5 ML 1X0.5 ML AMPOULE ml 7 DEXIT 100 1X1 ML Dexmedetomidine 100 mcg/1 ml 1X1 ML AMPOULE 8 DEXIT 200 1X2 ML Dexmedetomidine 200 mcg/2 ml 1X2 ML AMPOULE Isoflurane 100 ml / 250 ml Volatile liquid 9 ISOTROY 100 ml & 250 ml Bottle for Inhalation 10 ISOTROY 30ML 1X30ML Isoflurane 30 Ml 1X30ML BOTTLE Ketamine Inj. 11 KETAMAX-50 50 mg / ml 10 ml Vial Injection Rocuronium Nbromide Inj. 12 10 mg / ml 5 ml Vial Injection ROCUTROY 50 ml / 250 ml Sevoflurane Volatile liquid 13 50 ml & 250 ml Bottle SEVOTROY for Inhalation Suxamethonium Chloride Each ml contains 10 ml clear glass 14 Injection B.p Suxamethonium Chloride 50 mg Injection vial SUXATROY Propofol Inj. 20 ml / 50 ml 15 10mg / ml - 20 ml / 50 ml Vial Injection TROYPOFOL Vial Propofol MCT Inj. -

Stembook 2018.Pdf
The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances FORMER DOCUMENT NUMBER: WHO/PHARM S/NOM 15 WHO/EMP/RHT/TSN/2018.1 © World Health Organization 2018 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition”. Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. Suggested citation. The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances. Geneva: World Health Organization; 2018 (WHO/EMP/RHT/TSN/2018.1). Licence: CC BY-NC-SA 3.0 IGO. Cataloguing-in-Publication (CIP) data. -

Date: 27 Aug 2020 Important Information for Healthcare
Date: 27 Aug 2020 Important information for healthcare professionals Supply of Tracrium 10mg/ml Solution for Injection or Infusion, amps (50mg/5ml). - PA 1691/029/001 Batch: RP3G Exp: 01-Nov-2020 Quantity: 2130 packs This letter must remain with the pack at all times until the point of administration. Ampoules must not be taken out of the outer carton until the point of administration. Dear Healthcare Professional, I am writing to you with regards to supply of the above referenced product. We are currently unable to supply Irish licensed packs and have obtained approval from the Health Products Regulatory Authority (HPRA) to supply Ukrainian-labelled packs for a limited period, due to a large increase in demand. The Ukrainian packs are labelled with Batch No. RP3G and have an expiry date of 01-Nov-2020. Product from this batch will be supplied as a temporary measure until we resume supply of Irish stock. The pharmaceutical composition and specifications of Ukrainian product are equivalent to Irish licenced product. Use of the product should be carried out in-line with the Irish SmPC as usual, which can be found on the HPRA or medicines.ie website. We have also provided a label text comparison, copy of the Irish carton, and Patient Information Leaflet at the end of this letter. The Ukrainian Patient Information Leaflet should be discarded. Please ensure all relevant staff members are made aware of the content of this letter and that the information is communicated to patients. This communication has been agreed with the HPRA. 62000000041294 . • you have a form of cancer called Package leaflet: Information for the patient carcinomatosis • you have severe cardiovascular disease • you have a burn which requires medical Tracrium 10 mg/ml treatment solution for injection or • you have a tendency towards allergies or have asthma. -

Drug/Substance Trade Name(S)
A B C D E F G H I J K 1 Drug/Substance Trade Name(s) Drug Class Existing Penalty Class Special Notation T1:Doping/Endangerment Level T2: Mismanagement Level Comments Methylenedioxypyrovalerone is a stimulant of the cathinone class which acts as a 3,4-methylenedioxypyprovaleroneMDPV, “bath salts” norepinephrine-dopamine reuptake inhibitor. It was first developed in the 1960s by a team at 1 A Yes A A 2 Boehringer Ingelheim. No 3 Alfentanil Alfenta Narcotic used to control pain and keep patients asleep during surgery. 1 A Yes A No A Aminoxafen, Aminorex is a weight loss stimulant drug. It was withdrawn from the market after it was found Aminorex Aminoxaphen, Apiquel, to cause pulmonary hypertension. 1 A Yes A A 4 McN-742, Menocil No Amphetamine is a potent central nervous system stimulant that is used in the treatment of Amphetamine Speed, Upper 1 A Yes A A 5 attention deficit hyperactivity disorder, narcolepsy, and obesity. No Anileridine is a synthetic analgesic drug and is a member of the piperidine class of analgesic Anileridine Leritine 1 A Yes A A 6 agents developed by Merck & Co. in the 1950s. No Dopamine promoter used to treat loss of muscle movement control caused by Parkinson's Apomorphine Apokyn, Ixense 1 A Yes A A 7 disease. No Recreational drug with euphoriant and stimulant properties. The effects produced by BZP are comparable to those produced by amphetamine. It is often claimed that BZP was originally Benzylpiperazine BZP 1 A Yes A A synthesized as a potential antihelminthic (anti-parasitic) agent for use in farm animals. -

Summary of Product Characteristics 1. Name Of
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Atracurium Hikma 10mg/ml Solution for Injection/Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 10 mg of atracurium besilate. Each 2.5ml ampoule contains 25mg of Atracurium Besilate Each 5ml ampoule contains 50mg of Atracurium Besilate For the full list of excipients, see section 6.1 3. PHARMACEUTICAL FORM Solution for injection or Infusion Clear and colourless solution pH: 3.25 – 3.65 Osmolality: 10 – 30 mOsmol/Kg 4. CLINICAL PARTICULARS 4.1 Therapeutic indications Atracurium Hikma is a highly selective, competitive or non-depolarising neuromuscular blocking agent. It is used as an adjunct to general anaesthesia or sedation in the intensive care unit (ICU), to relax skeletal muscles, and to facilitate tracheal intubation and mechanical ventilation. 4.2 Posology and method of administration Administration by injection in adults Route of administration: Intravenous injection or continuous infusion. Atracurium Hikma is administered by intravenous injection. The dosage range recommended for adults is 0.3 to 0.6 mg/kg (depending on the duration of full block required) and will provide adequate relaxation for about 15 to 35 minutes. Endotracheal intubation can usually be accomplished within 90 seconds from the intravenous injection of 0.5 to 0.6 mg/kg. Full block can be prolonged with supplementary doses of 0.1 to 0.2 mg/kg as required. Successive supplementary dosing does not give rise to accumulation of neuromuscular blocking effect. Spontaneous recovery from the end of full block occurs in about 35 minutes as measured by the restoration of the tetanic response to 95% of normal neuromuscular function.