Texts Adopted by the European Phamacopoeia Commission
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Comparative Study of Neuromuscular Blocking Effects
IndianJournalofAnesthesiaandAnalgesia OriginalResearchArticle January–February2020;7(1)(Part-I):58-63 DOI:http://dx.doi.org/10.21088/ijaa.2349.8471.7120.9 AComparativeStudyofNeuromuscularBlockingEffectsandReversibility ofCisatracuriumandVecuronium DivyaNKheskani1,HeenaSChhanwal2,BhaktiSJain3 1AssistantProfessor,2ProfessorandHead,3rdYearResident,DepartmentofAnaesthesia,GCSMedicalCollege,Hospital&Research Centre,NearChamundaBridge,Ahmedabad,Gujarat380025,India. Abstract Context: Cisatracurium is a cis isomer of parent compound atracurium, devoid of histamine release, thus possessing hemodynamic & cardiovascular stability. Aims: We compared the intubating conditions, hemodynamic stability & recovery of atracurium & vecuronium. Settings and Design: We carried out prospective, double blind randomized study after approval of ethical committee. 100 adult patients of ASA 1 &2with comparable demographicdatawereselected. Dividedin TwoGroupsC(Cisatracurium) &V(Vecuronium).Standardmonitoringwasdone&followingroutinepremedication&inductionagents, patientswereintubatedat2minsafteradministrationofcisatracurium0.15mg/kg&vecuronium0.1mg/ kgrespectively,maintainedonintermittentdoseofcisatracurium:0.03mg/kg&vecuronium:0.02mg/kg. IntubationconditionswereassessedaccordingtoTimeto25%recoveryoft1/tcfollowinginitialdoses,Time to25%recoveryoft1/tcfollowingrepeatedboluses,Timeto25%recoveryoft1/tcfollowinglastdose,Return oft4/t1ratio0.8spontaneousrecoveryatendofoperation.Statisticalanalysisused:Theresultswereevaluated byapplyingpairedt-testandp-valueusingSPSSStatisticalSoftware.Results:Intubatingconditionsat2mins -

Use of Chemical Chelators As Reversal Agents for Drug
(19) TZZ_ _ZZZ_T (11) EP 1 210 090 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: A61K 31/724 (2006.01) A61K 31/194 (2006.01) 18.06.2014 Bulletin 2014/25 A61P 39/04 (2006.01) (21) Application number: 00964006.1 (86) International application number: PCT/EP2000/007694 (22) Date of filing: 07.08.2000 (87) International publication number: WO 2001/012202 (22.02.2001 Gazette 2001/08) (54) USE OF CHEMICAL CHELATORS AS REVERSAL AGENTS FOR DRUG- INDUCED NEUROMUSCULAR BLOCK VERWENDUNG VON CHEMISCHEN CHELATOREN ZUR UMKEHRUNG VON PHARMAKOLOGISCH-INDUZIERTER NEUROMUSKULÄRER BLOCKIERUNG UTILISATION D’AGENTS CHIMIQUES CHELATANTS COMME AGENTS DE NEUTRALISATION DU BLOCAGE NEUROMUSCULAIRE PROVOQUE PAR DES MEDICAMENTS (84) Designated Contracting States: (56) References cited: AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU AU-A- 3 662 895 MC NL PT SE • B DESIRE: "Inactivaton of sarin and soman by (30) Priority: 13.08.1999 EP 99306411 cyclodextrins in vitro" EXPERIENTIA, vol. 43, no. 4, 1987, pages 395-397, XP000907287 (43) Date of publication of application: • B. DESIRE: "Interaction of soman with beta- 05.06.2002 Bulletin 2002/23 cyclodextrin" FUNDAMENTAL AND APPLIED TOXICOLOGY, vol. 7, no. 4, 1986, pages 647-657, (73) Proprietor: Merck Sharp & Dohme B.V. XP000911170 2031 BN Haarlem (NL) • C. MAY: "Development of a toxin-binding agent as a treatment for tunicamycinuracil toxicity: (72) Inventors: protection against tunicamycin poisoning of • BOM, Antonius, Helena, Adolf sheep" AUSTRALIAN VETERINARY JOURNAL, Ratho, Midlothian EH28 8NY (GB) vol. 76, no. -

Download Document
FAM001159-0001 intervals of 4-7 days to usual dose of 75-100 mg Dolmatil® (Sanofi-Synthelabo) ~ 100 rag/5 mL, thioridazine 100 rag/ per course and max. 4 injections; max. duration of daily according to response; CHILD not recom- Tablets, both scored, sulpiride 200 rag, net price ’ net price 300 mL = £7.14. Label: 2 treatment 2 weeks---if maintenance treatment mended 100-tabpack=£13.85;400mg(f/c), 100-tab ! fNote. These suspensions should not be diluted but the necessary change to an oral antipsychotic 2-3 Short-term adjunctive management of severe pack = £36¯29. Label: 2 .: :~t~a preparations may be mixed with each other to days after last injection, or to a longer acting anti- anxiety, 15-20rag daily in divided doses; max. Sulpltil® (Pharmacia) I~ ~0iovid¢ intermediate strengths psychotic depot injection given concomitantly 40 mg daily; CHILD not recommended Tablets, scored, sulpiride 200 rag. Net price 28-tab "ff~-~p, brown, thioridazine (as hydrochloride) with last injection of zuclopenthixol acetate; By deep intramuscular injection, psychoses, mania, pack = £4.29; 112-tab pack = £12.85. Label: 2 ~ff.~5"~mg/5 mL, net price 300 mL = £1.98. Label: 2 CHILD not recommended prochlorperazine mesilate 12.5-25 mg 2-3 times Sulpor® (Rosemont) IPoMI Clopi~ol Acuphase® (Lundbeck) daily; CHILD not recommended Oral solution, sugar-free, lemon- and aniseed-fla. LUOPERAZINE Injection (oily), zuclopenthixol acetate 50 mg/mL. By rectum in suppositories, psychoses, mania, the voured, sulpiride 200 mg/5 mL, net price 150 mL (’~’n~ications: see under Dose; anti-emetic (section Net price I-mL amp = £5.20; 2-mL amp = £10.03 equivalent of prochlorperazine maleate 25 mg 2- = £27.00. -

The Study Programme for the Quality Management of Essential Medicines - Good Manufacturing Practical (GMP) and Inspection
The Study Programme for the Quality Management of Essential Medicines - Good Manufacturing Practical (GMP) and Inspection - Country Reports Japan International Corporation of Welfare Services (JICWELS) Contents 1. Cambodia 1 2. Indonesia 70 3. Malaysia 91 4. Philippines 116 5. Sri Lanka 141 6. Thailand 161 The Study Programme for the Quality Management of Essential Medicines - Good Manufacturing Practical (GMP) and Inspection - Cambodia -1- KINGDOM OF CAMBODIA Nation Religion King Ministry of Health Department of Drugs and Food Country Report The Study Program on Quality Management of Essential Medicines Good Manufacturing Practice (GMP) and Inspection November 4, 2012 – November 30, 2012 Sponsored by : The Government of Japan Japan International Cooperation Agency (JICA) Department of Drugs and Food Ministry of Health, Cambodia. -2- I- COUNTRY PROFILE -3- A-Geography Cambodia is an agricultural country located in South East Asia which bordering the Gulf of Thailand, between Thailand, Vietnam, and Laos. Its approximate geographical coordinates are 13°N 105°E. Its 2,572 km border is split among Vietnam (1,228 km), Thailand (803 km) and Laos (541 km), as well as 443 km of coastline. Cambodia covers 181,035 square kilometers in the southwestern part of the Indochina, Cambodia lies completely within the tropics; its southernmost points are only slightly more than 10° above the equator. The country is bounded on the north by Thailand and by Laos, on the east and southeast by Vietnam, and on the west by the Gulf of Thailand and by Thailand. It consists of the Tonle Sap Basin and the Mekong Lowlands. To the southeast of this great basin is the Mekong Delta, which extends through Vietnam to the South China Sea. -
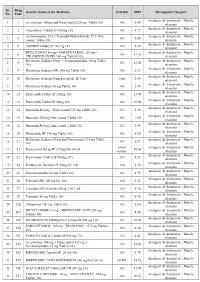
PMBJP Product.Pdf
Sr. Drug Generic Name of the Medicine Unit Size MRP Therapeutic Category No. Code Analgesic & Antipyretic / Muscle 1 1 Aceclofenac 100mg and Paracetamol 325 mg Tablet 10's 10's 8.00 relaxants Analgesic & Antipyretic / Muscle 2 2 Aceclofenac Tablets IP 100mg 10's 10's 4.37 relaxants Acetaminophen 325 + Tramadol Hydrochloride 37.5 film Analgesic & Antipyretic / Muscle 3 4 10's 8.00 coated Tablet 10's relaxants Analgesic & Antipyretic / Muscle 4 5 ASPIRIN Tablets IP 150 mg 14's 14's 2.70 relaxants DICLOFENAC 50 mg+ PARACETAMOL 325 mg+ Analgesic & Antipyretic / Muscle 5 6 10's 11.30 CHLORZOXAZONE 500 mg Tablets 10's relaxants Diclofenac Sodium 50mg + Serratiopeptidase 10mg Tablet Analgesic & Antipyretic / Muscle 6 8 10's 12.00 10's relaxants Analgesic & Antipyretic / Muscle 7 9 Diclofenac Sodium (SR) 100 mg Tablet 10's 10's 6.12 relaxants Analgesic & Antipyretic / Muscle 8 10 Diclofenac Sodium 25mg per ml Inj. IP 3 ml 3 ml 2.00 relaxants Analgesic & Antipyretic / Muscle 9 11 Diclofenac Sodium 50 mg Tablet 10's 10's 2.90 relaxants Analgesic & Antipyretic / Muscle 10 12 Etoricoxilb Tablets IP 120mg 10's 10's 33.00 relaxants Analgesic & Antipyretic / Muscle 11 13 Etoricoxilb Tablets IP 90mg 10's 10's 25.00 relaxants Analgesic & Antipyretic / Muscle 12 14 Ibuprofen 400 mg + Paracetamol 325 mg Tablet 10's 15's 5.50 relaxants Analgesic & Antipyretic / Muscle 13 15 Ibuprofen 200 mg film coated Tablet 10's 10's 1.80 relaxants Analgesic & Antipyretic / Muscle 14 16 Ibuprofen 400 mg film coated Tablet 10's 15's 3.50 relaxants Analgesic & Antipyretic -

Cost Analysis and Safety Comparison of Cisatracurium and Atracurium in Patients Undergoing General Anesthesia
Eur opean Rev iew for Med ical and Pharmacol ogical Sci ences 2013; 17: 447-450 Cost analysis and safety comparison of Cisatracurium and Atracurium in patients undergoing general anesthesia A. MOVAFEGH, S. AMINI*, H. SHARIFNIA, H. TORKAMANDI*, A. HAYATSHAHI*, M. JAVADI* Intensive Care Unit, and *Pharmaceutical Care Department; Dr. Shariati Hospital, Tehran University of Medical Sciences, Tehran, Iran Abstract. – BACKGROUND : Non-depolarizing Introduction neuromuscular blocking agents (NMB) differ in pharmacokinetic and pharmacodynamic parame - The introduction of neuromuscular blocking ters. An anesthesiologist according to these simi - larities and differences is able to choose the least agents in 1942 into anesthetic practice was an im - costly one if the same safety profile and same clin - portant development. Non-depolarizing NMB ical benefit achieved with the different alternatives. agents differ in the onset of action, duration of ac - AIM: The main objective of this study is to eval - tion, metabolic route, potency, adverse effects and uate the economic and adverse drug reactions cost. An anesthesiologist is able to choose NMB prevalence and differences between cisatracurium drugs according to these similarities and differ - and atracurium the two non-depolarizing NMB 1,2 drugs, which are widely used in adult patients un - ences . Atracurium and Cisatracurium are two dergoing surgery with general anesthesia in a non-depolarizing NMB agents with intermediate teaching Hospital in Iran. duration of action 1. Cisatracurium besilate is the MATERIALS AND METHODS : A cost analysis R-cis isomer of atracurium besilate and is 3-4 fold and adverse drug reactions (ADR) monitoring more potent than atracurium 3. Compared with were performed. -

Etats Rapides
List of European Pharmacopoeia Reference Standards Effective from 2015/12/24 Order Reference Standard Batch n° Quantity Sale Information Monograph Leaflet Storage Price Code per vial Unit Y0001756 Exemestane for system suitability 1 10 mg 1 2766 Yes +5°C ± 3°C 79 ! Y0001561 Abacavir sulfate 1 20 mg 1 2589 Yes +5°C ± 3°C 79 ! Y0001552 Abacavir for peak identification 1 10 mg 1 2589 Yes +5°C ± 3°C 79 ! Y0001551 Abacavir for system suitability 1 10 mg 1 2589 Yes +5°C ± 3°C 79 ! Y0000055 Acamprosate calcium - reference spectrum 1 n/a 1 1585 79 ! Y0000116 Acamprosate impurity A 1 50 mg 1 3-aminopropane-1-sulphonic acid 1585 Yes +5°C ± 3°C 79 ! Y0000500 Acarbose 3 100 mg 1 See leaflet ; Batch 2 is valid until 31 August 2015 2089 Yes +5°C ± 3°C 79 ! Y0000354 Acarbose for identification 1 10 mg 1 2089 Yes +5°C ± 3°C 79 ! Y0000427 Acarbose for peak identification 3 20 mg 1 Batch 2 is valid until 31 January 2015 2089 Yes +5°C ± 3°C 79 ! A0040000 Acebutolol hydrochloride 1 50 mg 1 0871 Yes +5°C ± 3°C 79 ! Y0000359 Acebutolol impurity B 2 10 mg 1 -[3-acetyl-4-[(2RS)-2-hydroxy-3-[(1-methylethyl)amino] propoxy]phenyl] 0871 Yes +5°C ± 3°C 79 ! acetamide (diacetolol) Y0000127 Acebutolol impurity C 1 20 mg 1 N-(3-acetyl-4-hydroxyphenyl)butanamide 0871 Yes +5°C ± 3°C 79 ! Y0000128 Acebutolol impurity I 2 0.004 mg 1 N-[3-acetyl-4-[(2RS)-3-(ethylamino)-2-hydroxypropoxy]phenyl] 0871 Yes +5°C ± 3°C 79 ! butanamide Y0000056 Aceclofenac - reference spectrum 1 n/a 1 1281 79 ! Y0000085 Aceclofenac impurity F 2 15 mg 1 benzyl[[[2-[(2,6-dichlorophenyl)amino]phenyl]acetyl]oxy]acetate -

(12) Patent Application Publication (10) Pub. No.: US 2006/0024365A1 Vaya Et Al
US 2006.0024.365A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2006/0024365A1 Vaya et al. (43) Pub. Date: Feb. 2, 2006 (54) NOVEL DOSAGE FORM (30) Foreign Application Priority Data (76) Inventors: Navin Vaya, Gujarat (IN); Rajesh Aug. 5, 2002 (IN)................................. 699/MUM/2002 Singh Karan, Gujarat (IN); Sunil Aug. 5, 2002 (IN). ... 697/MUM/2002 Sadanand, Gujarat (IN); Vinod Kumar Jan. 22, 2003 (IN)................................... 80/MUM/2003 Gupta, Gujarat (IN) Jan. 22, 2003 (IN)................................... 82/MUM/2003 Correspondence Address: Publication Classification HEDMAN & COSTIGAN P.C. (51) Int. Cl. 1185 AVENUE OF THE AMERICAS A6IK 9/22 (2006.01) NEW YORK, NY 10036 (US) (52) U.S. Cl. .............................................................. 424/468 (22) Filed: May 19, 2005 A dosage form comprising of a high dose, high Solubility active ingredient as modified release and a low dose active ingredient as immediate release where the weight ratio of Related U.S. Application Data immediate release active ingredient and modified release active ingredient is from 1:10 to 1:15000 and the weight of (63) Continuation-in-part of application No. 10/630,446, modified release active ingredient per unit is from 500 mg to filed on Jul. 29, 2003. 1500 mg, a process for preparing the dosage form. Patent Application Publication Feb. 2, 2006 Sheet 1 of 10 US 2006/0024.365A1 FIGURE 1 FIGURE 2 FIGURE 3 Patent Application Publication Feb. 2, 2006 Sheet 2 of 10 US 2006/0024.365A1 FIGURE 4 (a) 7 FIGURE 4 (b) Patent Application Publication Feb. 2, 2006 Sheet 3 of 10 US 2006/0024.365 A1 FIGURE 5 100 ov -- 60 40 20 C 2 4. -

10 Mg/Ml Solution for Injection/Infusion Atracurium
Package leaflet: Information for the patient <Product name> 10 mg/ml solution for injection/infusion Atracurium besilate Read all of this leaflet carefully before you start using this medicine because it contains important information for you. - Keep this leaflet. You may need to read it again. - If you have any further questions, ask your doctor or nurse. - If you get any side effects, talk to your doctor or nurse. This includes any possible side effects not listed in this leaflet. See section 4. What is in this leaflet 1. What <Product name> is and what it is used for 2. What you need to know before you receive <Product name> 3. How to use <Product name> 4. Possible side effects 5. How to store <Product name> 6. Contents of the pack and other information 1. What <Product name> is and what it is used for <Product name> belongs to a group of medicines called muscle relaxants. <Product name> is used during surgery to relax muscles and to assist with inserting a breathing tube and with artificial breathing. It is also used to help with artificial breathing at patients in intensive care units. 2. What you need to know before you receive <Product name> <Product name> cannot be administered: - if you are allergic to atracurium besilate, cisatracurium or any of the other ingredients of this medicine (listed in section 6). If you think that this applies to you talk to your doctor before <Product name> is administered to you. Warnings and precautions Talk to your doctor or nurse before using <Product name>: - if you have an allergy or bronchial -

(12) United States Patent (10) Patent N0.: US 7,265,099 B1 Born Et A1
US007265099B1 (12) United States Patent (10) Patent N0.: US 7,265,099 B1 Born et a1. (45) Date of Patent: *Sep. 4, 2007 (54) USE OF CHEMICAL CHELATORS AS Tarver, G. et al “2-O-Substituted cyclodextrins as reversal REVERSAL AGENTS FOR DRUG-INDUCED agents . ” Bioorg. Med. Chem. (2002) vol. 10, pp 1819-1827.* NEUROMUSCULAR BLOCK Zhang, M. “Drug-speci?c cyclodextrins . ” Drugs of the Future (2003) vol. 28, no 4, pp 347-354.* (75) Inventors: Antonius Helena Adolf Bom, Lee, C. “Structure, conformation, and action of neuromuscular Midlothian (GB); Alan William Muir, blocking drugs” Brit. J. Anesth. (2001) vol. 87, no 5, pp 755-769.* Lanark (GB); David Rees, Gothenburg B Desire: “Inactivation of sarin and soman by cyclodextrins in (SE) vitro” EXPERIENTIA, vol. 43, No. 4, 1987, pp. 395-397. B. Desire: “Interaction of soman with beta-cyclodextrin” Funda (73) Assignee: Organon N.V., Oss (NL) mental and Applied Toxicology, vol. 7, No. 4, 1986, pp. 647-657. ( * ) Notice: Subject to any disclaimer, the term of this C. May: “Development of a toxin-bindng agent as a treatment for patent is extended or adjusted under 35 tunicamycinuracil toxicity: protection against tunicamycin poison U.S.C. 154(b) by 0 days. ing of sheep” Australian Veterinary Journal, vol. 76, No. 11, 1998 pp. 752-756. This patent is subject to a terminal dis K. Uekama: “Effects of cyclodextrins on chlorpromaZine-induced claimer. haemolysis and nervous systems responses” J. Pharm. Pharmacol., vol. 33, No. 11, 1981, pp. 707-710. (21) Appl. No.: 10/049,393 T. Irie: “Protective mechanism of beta-cyclodextrin for the hemolysis induced With phenothiazine neuroleptics in vitro” J. -

Observations on the Use of Different Modes of Cisatracurium Administration in Hyperthermic Intraperitoneal Chemotherapy
Observations on the Use of Different Modes of Cisatracurium Administration in Hyperthermic Intraperitoneal Chemotherapy Jing Zhang Capital Medical University Aliated Beijing Shijitan Hospital Danyang Gao Capital Medical University Aliated Beijing Shijitan Hospital https://orcid.org/0000-0002-8491-5376 Pengfei Liu Capital Medical University Aliated Beijing Shijitan Hospital Tianzuo Li ( [email protected] ) Capital Medical University Aliated Beijing Shijitan Hospital Research Article Keywords: Cis-atracurium, Intraperitoneal hyperperfusion chemotherapy (HIPEC), Anesthesia, Residual muscle relaxation Posted Date: April 12th, 2021 DOI: https://doi.org/10.21203/rs.3.rs-392323/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 1/10 Abstract Objective: To investigate the residual effects of cis-atracurium with different administrations in patients with hyperthermic intraperitoneal chemotherapy. Methods: 60 patients undergoing hyperthermic intraperitoneal chemotherapy were randomly divided into two groups with 30 cases in each group. All patients were induced with sufentanil 0.4ug/kg, propofol 2.0mg/kg and cis atracurium 0.2mg/kg. During the operation, sevourane and remifentanil were used to maintain anesthesia. BIS value was 45-55. The control group intermittently received cis-atracurium injection with 0.1mg/kg/h. The experimental group received continuous injection of cis-atracurium at a rate of 1.5ug/kg/min. Cis-atracurium was discontinued 30min before the end of the operation, sevourane inhalation was stopped 20min before the end of the surgery and remifentanil was discontinued 10min before the end. At the end of the operation, no muscle relaxant antagonism were applied. General information of patients was recorded. -

The New Neuromuscular Blocking Agents: Do They Offer Any Advantages?
British Journal of Anaesthesia 87 (6): 912±25 (2001) REVIEW ARTICLE The new neuromuscular blocking agents: do they offer any advantages? E. W. Moore* and J. M. Hunter² University Department of Anaesthesia, University Clinical Department, The Duncan Building, Daulby Street, Liverpool L69 3GA, UK *Corresponding author Br J Anaesth 2001; 87: 912±25 Keywords: neuromuscular block, aminosteroids; neuromuscular block, benzylisoquinoliniums; neuromuscular block, tropinyl diesters; neuromuscular block, tetrahydroisoquinolinium chlorofumarates The introduction of neuromuscular blocking drugs in 1942 animal and human studies. The bis-quaternary ammonium marked a new era in anaesthetic and surgical practice. The salt of bistropinyl diester G-1-64 is the most studied of the anaesthetist was enabled to provide respiratory support tropinyl diester group51 although investigations in humans during long and complex surgery whilst the surgeon was have yet to be reported (Fig. 3). allowed access to body cavities without voluntary or re¯ex muscle movement. In recent years, a generation of anaesthetists trained since atracurium and vecuronium GW280430A became available in 1982, have taken for granted the The structure of this mixed-tetrahydroisoquinolinium speed of onset, shorter and more predictable duration of chlorofumarate is given in Figure 2. The similarity in action, and lack of cardiovascular side-effects that these structure to mivacurium is demonstrated. The presence of drugs have in comparison with their predecessors.56 58 59 three methyl groups between the quaternary nitrogen and The latest generation of neuromuscular blocking drugs aims oxygen atom at each end of the carbon chain suggests that, to provide even greater advantages: as rapid an onset and similar to mivacurium, this compound will not undergo offset as succinylcholine; disposition independent of organ Hofmann degradation.