On the Discovery of New Elements (IUPAC/IUPAP Provisional Report)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

CHEMISTRY for the Bottom of the Periodic Table
http://cyclotron.tamu.edu CHEMISTRY for the Bottom of the Periodic Table Techniques to investigate chemical properties of superheavy elements lead to improved methods for separating heavy metals THE SCIENCE The chemical properties of superheavy element 113, nihonium, are almost completely unknown, so a team of researchers from the Cyclotron Institute at Texas A&M University and the Institut Pluridisciplinaire Hubert Curien in France are developing techniques that could be used to study this fleeting element. As part of that effort, they are comparing the properties of nihonium to the chemically similar elements indium and thallium; to do so, the team studied the separation of these two elements using a new class of designer molecules called ionic liquids. THE IMPACT Measuring the chemical properties of nihonium and other superheavy elements will increase our understanding of the principles that control the Periodic Table. Comparing the data from nihonium to results for similar elements, obtained using the team’s fast, efficient, single-step process, reveals trends that arise from the structure of the Periodic Table. This research could also lead to better methods of re- using indium, a metal that is part of flat-panel displays but not currently mined in the United States. The proposed mechanism of transfer. Thallium (Tl) bonds with chlorine (Cl) and moves into the ionic liquid (in blue). SUMMARY The distribution of indium and thallium between the aque- ous and organic phases is the key to understanding the PUBLICATIONS separation of these elements. An aqueous solution, con- E.E. Tereshatov, M. Yu. Boltoeva, V. Mazan, M.F. -

CME Search Before Isobar Collisions and Blind Analysis from STAR
CME Search Before Isobar Collisions and Blind Analysis From STAR Prithwish Tribedy for the STAR collaboration The 36th Winter Workshop on Nuclear Dynamics 1-7th March, 2020, Puerto Vallarta, Mexico In part supported by Introduction Solenoidal Tracker at RHIC (STAR) RHIC has collided multiple ion species; year 2018 was dedicated to search for effects driven by strong electromagnetic fields by STAR Isobars: Ru+Ru, Zr+Zr @ 200GeV (2018) Low energy: Au+Au 27 GeV (2018) Large systems : U+U, Au+Au @ 200 GeV P.Tribedy, WWND 2 The Chiral Magnetic Effect (Cartoon Picture) Quarks Quarks randomly aligned oriented along B antiquarks I II III IV More More right left handed handed quarks quarks J || B J || -B CME converts chiral imbalance to observable electric current P.Tribedy, WWND 2020 3 New Theory Guidance : Complexity Of An Event Magnetic field map Pb+Pb @ 2.76 TeV Axial charge profile b=11.4 fm, Npart=56 Pb+Pb 2.76 TeV, b=11.4 fm, dN5/dxT [a.u.] 0.3 6 uR>uL 0.2 4 uR<uL 0.1 2 0 0 y [fm] -2 -0.1 -4 -0.2 -6 -0.3 -6 -4 -2 0 2 4 6 x [fm] Based on: Chatterjee, Tribedy, Phys. Rev. C 92, Based on: Lappi, Schlichting, Phys. Rev. D 97, 011902 (2015) 034034 (2018) Going beyond cartoon picture: 1) Fluctuations dominate e-by-e physics, 2) B-field & domain size of axial-charge change with √s P.Tribedy, WWND 2020 4 New Theory Guidance : Complexity Of An Event Pb+Pb @ 2.76 TeV Magnetic field b=11.4 fm, N Axial charge profile Pb+Pb 2.76 TeV, b=11.4 fm, dN5/dxT [a.u.] 0.3 6 u 0.2 4 u 0.1 2 0 0 y [fm] -2 -0.1 -4 -0.2 -6 -0.3 -6 -4 -2 0 2 4 6 x [fm] Based on: Chatterjee, -

The Development of the Periodic Table and Its Consequences Citation: J
Firenze University Press www.fupress.com/substantia The Development of the Periodic Table and its Consequences Citation: J. Emsley (2019) The Devel- opment of the Periodic Table and its Consequences. Substantia 3(2) Suppl. 5: 15-27. doi: 10.13128/Substantia-297 John Emsley Copyright: © 2019 J. Emsley. This is Alameda Lodge, 23a Alameda Road, Ampthill, MK45 2LA, UK an open access, peer-reviewed article E-mail: [email protected] published by Firenze University Press (http://www.fupress.com/substantia) and distributed under the terms of the Abstract. Chemistry is fortunate among the sciences in having an icon that is instant- Creative Commons Attribution License, ly recognisable around the world: the periodic table. The United Nations has deemed which permits unrestricted use, distri- 2019 to be the International Year of the Periodic Table, in commemoration of the 150th bution, and reproduction in any medi- anniversary of the first paper in which it appeared. That had been written by a Russian um, provided the original author and chemist, Dmitri Mendeleev, and was published in May 1869. Since then, there have source are credited. been many versions of the table, but one format has come to be the most widely used Data Availability Statement: All rel- and is to be seen everywhere. The route to this preferred form of the table makes an evant data are within the paper and its interesting story. Supporting Information files. Keywords. Periodic table, Mendeleev, Newlands, Deming, Seaborg. Competing Interests: The Author(s) declare(s) no conflict of interest. INTRODUCTION There are hundreds of periodic tables but the one that is widely repro- duced has the approval of the International Union of Pure and Applied Chemistry (IUPAC) and is shown in Fig.1. -
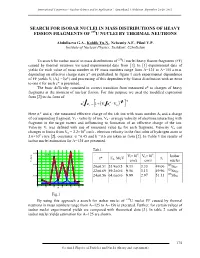
Search for Isobar Nuclei in Mass Distributions of Heavy Fission Fragments of 235U Nuclei by Thermal Neutrons
International Conference “Nuclear Science and its Application”, Samarkand, Uzbekistan, September 25-28, 2012 SEARCH FOR ISOBAR NUCLEI IN MASS DISTRIBUTIONS OF HEAVY FISSION FRAGMENTS OF 235U NUCLEI BY THERMAL NEUTRONS Abdullaeva G.A., Koblik Yu.N., Nebesniy A.F., Pikul V.P. Institute of Nuclear Physics, Tashkent, Uzbekistan To search for isobar nuclei in mass distributions of 235U nuclei heavy fission fragments (FF) caused by thermal neutrons we used experimental data from [1]. In [1] experimental data of yields for each value of mass number in FF mass numbers range from A=125 to A=155 a.m.u. depending on effective charge state z* are published. In figure 1 such experimental dependence of FF yields Yi (Ai) =f(z*) and processing of this dependence by Gauss distribution with an error to one σ for each z* is presented. The basic difficulty consisted in correct transition from measured z* to charges of heavy fragments at the moment of nuclear fission. For this purpose we used the modified expression from [2] in the form of: k * α k1 i i ii VzV1zz 0 . Here zi* and zi - the measured effective charge of the i-th ion with mass number Ai and a charge of corresponding fragment, Vi - velocity of ion, Vo - average velocity of electrons interacting with fragment in the target matter and influencing to formation of an effective charge of the ion. Velocity Vi was defined with use of measured value Ek for each fragment. Velocity Vo can 8 changes in limits from Vo = 2.2×10 cm/s - electron velocity in the first orbit of hydrogen atom to 3.6×108 cm/s [2], constants: α =0.45 and k =0.6 are taken as from [2]. -

The Periodic Table of Elements
The Periodic Table of Elements 1 2 6 Atomic Number = Number of Protons = Number of Electrons HYDROGENH HELIUMHe 1 Chemical Symbol NON-METALS 4 3 4 C 5 6 7 8 9 10 Li Be CARBON Chemical Name B C N O F Ne LITHIUM BERYLLIUM = Number of Protons + Number of Neutrons* BORON CARBON NITROGEN OXYGEN FLUORINE NEON 7 9 12 Atomic Weight 11 12 14 16 19 20 11 12 13 14 15 16 17 18 SODIUMNa MAGNESIUMMg ALUMINUMAl SILICONSi PHOSPHORUSP SULFURS CHLORINECl ARGONAr 23 24 METALS 27 28 31 32 35 40 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 POTASSIUMK CALCIUMCa SCANDIUMSc TITANIUMTi VANADIUMV CHROMIUMCr MANGANESEMn FeIRON COBALTCo NICKELNi CuCOPPER ZnZINC GALLIUMGa GERMANIUMGe ARSENICAs SELENIUMSe BROMINEBr KRYPTONKr 39 40 45 48 51 52 55 56 59 59 64 65 70 73 75 79 80 84 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 RUBIDIUMRb STRONTIUMSr YTTRIUMY ZIRCONIUMZr NIOBIUMNb MOLYBDENUMMo TECHNETIUMTc RUTHENIUMRu RHODIUMRh PALLADIUMPd AgSILVER CADMIUMCd INDIUMIn SnTIN ANTIMONYSb TELLURIUMTe IODINEI XeXENON 85 88 89 91 93 96 98 101 103 106 108 112 115 119 122 128 127 131 55 56 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86 CESIUMCs BARIUMBa HAFNIUMHf TANTALUMTa TUNGSTENW RHENIUMRe OSMIUMOs IRIDIUMIr PLATINUMPt AuGOLD MERCURYHg THALLIUMTl PbLEAD BISMUTHBi POLONIUMPo ASTATINEAt RnRADON 133 137 178 181 184 186 190 192 195 197 201 204 207 209 209 210 222 87 88 104 105 106 107 108 109 110 111 112 113 114 115 116 117 118 FRANCIUMFr RADIUMRa RUTHERFORDIUMRf DUBNIUMDb SEABORGIUMSg BOHRIUMBh HASSIUMHs MEITNERIUMMt DARMSTADTIUMDs ROENTGENIUMRg COPERNICIUMCn NIHONIUMNh -

Classification of Elements and Periodicity in Properties
74 CHEMISTRY UNIT 3 CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES The Periodic Table is arguably the most important concept in chemistry, both in principle and in practice. It is the everyday support for students, it suggests new avenues of research to After studying this Unit, you will be professionals, and it provides a succinct organization of the able to whole of chemistry. It is a remarkable demonstration of the fact that the chemical elements are not a random cluster of • appreciate how the concept of entities but instead display trends and lie together in families. grouping elements in accordance to An awareness of the Periodic Table is essential to anyone who their properties led to the wishes to disentangle the world and see how it is built up development of Periodic Table. from the fundamental building blocks of the chemistry, the understand the Periodic Law; • chemical elements. • understand the significance of atomic number and electronic Glenn T. Seaborg configuration as the basis for periodic classification; • name the elements with In this Unit, we will study the historical development of the Z >100 according to IUPAC Periodic Table as it stands today and the Modern Periodic nomenclature; Law. We will also learn how the periodic classification • classify elements into s, p, d, f follows as a logical consequence of the electronic blocks and learn their main configuration of atoms. Finally, we shall examine some of characteristics; the periodic trends in the physical and chemical properties • recognise the periodic trends in of the elements. physical and chemical properties of elements; 3.1 WHY DO WE NEED TO CLASSIFY ELEMENTS ? compare the reactivity of elements • We know by now that the elements are the basic units of all and correlate it with their occurrence in nature; types of matter. -

The Oganesson Odyssey Kit Chapman Explores the Voyage to the Discovery of Element 118, the Pioneer Chemist It Is Named After, and False Claims Made Along the Way
in your element The oganesson odyssey Kit Chapman explores the voyage to the discovery of element 118, the pioneer chemist it is named after, and false claims made along the way. aving an element named after you Ninov had been dismissed from Berkeley for is incredibly rare. In fact, to be scientific misconduct in May5, and had filed Hhonoured in this manner during a grievance procedure6. your lifetime has only happened to Today, the discovery of the last element two scientists — Glenn Seaborg and of the periodic table as we know it is Yuri Oganessian. Yet, on meeting Oganessian undisputed, but its structure and properties it seems fitting. A colleague of his once remain a mystery. No chemistry has been told me that when he first arrived in the performed on this radioactive giant: 294Og halls of Oganessian’s programme at the has a half-life of less than a millisecond Joint Institute for Nuclear Research (JINR) before it succumbs to α -decay. in Dubna, Russia, it was unlike anything Theoretical models however suggest it he’d ever experienced. Forget the 2,000 ton may not conform to the periodic trends. As magnets, the beam lines and the brand new a noble gas, you would expect oganesson cyclotron being installed designed to hunt to have closed valence shells, ending with for elements 119 and 120, the difference a filled 7s27p6 configuration. But in 2017, a was Oganessian: “When you come to work US–New Zealand collaboration predicted for Yuri, it’s not like a lab,” he explained. that isn’t the case7. -

IUPAC Wire See Also
News and information on IUPAC, its fellows, and member organizations. IUPAC Wire See also www.iupac.org Flerovium and Livermorium Join the Future Earth: Research for Global Periodic Table Sustainability n 30 May 2012, IUPAC officially approved he International Council for Science (ICSU), of the name flerovium, with symbol Fl, for the which IUPAC is a member, announced a new Oelement of atomic number 114 and the name T10-year initiative named Future Earth to unify livermorium, with symbol Lv, for the element of atomic and scale up ICSU-sponsored global environmental- number 116. The names and symbols were proposed change research. by the collaborating team of the Joint Institute for Nuclear Research (Dubna, Russia) and the Lawrence Operational in 2013, this new ICSU initiative will Livermore National Laboratory (Livermore, California, provide a cutting-edge platform to coordinate scien- USA) to whom the priority for the discovery of these tific research to respond to the most critical social and elements was assigned last year. The IUPAC recom- environmental challenges of the 21st century at global mendations presenting these names is to appear in and regional levels. “This initiative will link global envi- the July 2012 issue of Pure and Applied Chemistry. ronmental change and fundamental human develop- ment questions,” said Diana Liverman, co-director of The name flerovium, with symbol Fl, lies within the Institute of the Environment at the University of tradition and honors the Flerov Laboratory of Nuclear Arizona and co-chair of the team Reactions in Dubna, Russia, where the element of that is designing Future Earth. -

The Histories Hidden in the Periodic Table
The Histories Hidden in the Periodic Table From poisoned monks and nuclear bombs to the “transfermium wars,” mapping the atomic world hasn’t been easy. By Neima Jahromi 6:00 A.M. As element hunters have become element makers, the periodic table’s meaning has changed. It now describes what is possible, in addition to what merely exists. Illustration by Ilya Milstein The story of the fifteenth element began in Hamburg, in 1669. The unsuccessful glassblower and alchemist Hennig Brandt was trying to find the philosopher’s stone, a mythical substance that could turn base metals into gold. Instead, he distilled something new. It was foamy and, depending on the preparation, yellow or black. He called it “cold fire,” because it glowed in the dark. Interested parties took a look; some felt that they were in the presence of a miracle. “If anyone had rubbed himself all over with it,” one observer noted, “his whole figure would have shone, as once did that of Moses when he came down from Mt. Sinai.” Robert Boyle, the father of modern chemistry, put some on his hand and noted how “mild and innocent” it seemed. Another scientist saw particles in it twinkling “like little stars.” At first, no one could figure out what the Prometheus of Hamburg had stolen. After one of Brandt’s confidants provided a hint—the main ingredient was “somewhat that belong’d to the Body of Man”—Boyle deduced that he and his peers had been smearing themselves with processed urine. As the Cambridge chemist Peter Wothers explains in his new history of the elements, “Antimony, Gold, and Jupiter’s Wolf” (Oxford), Brandt’s recipe called for a ton of urine. -
![Arxiv:1901.01410V3 [Astro-Ph.HE] 1 Feb 2021 Mental Information Is Available, and One Has to Rely Strongly on Theoretical Predictions for Nuclear Properties](https://docslib.b-cdn.net/cover/8159/arxiv-1901-01410v3-astro-ph-he-1-feb-2021-mental-information-is-available-and-one-has-to-rely-strongly-on-theoretical-predictions-for-nuclear-properties-508159.webp)
Arxiv:1901.01410V3 [Astro-Ph.HE] 1 Feb 2021 Mental Information Is Available, and One Has to Rely Strongly on Theoretical Predictions for Nuclear Properties
Origin of the heaviest elements: The rapid neutron-capture process John J. Cowan∗ HLD Department of Physics and Astronomy, University of Oklahoma, 440 W. Brooks St., Norman, OK 73019, USA Christopher Snedeny Department of Astronomy, University of Texas, 2515 Speedway, Austin, TX 78712-1205, USA James E. Lawlerz Physics Department, University of Wisconsin-Madison, 1150 University Avenue, Madison, WI 53706-1390, USA Ani Aprahamianx and Michael Wiescher{ Department of Physics and Joint Institute for Nuclear Astrophysics, University of Notre Dame, 225 Nieuwland Science Hall, Notre Dame, IN 46556, USA Karlheinz Langanke∗∗ GSI Helmholtzzentrum f¨urSchwerionenforschung, Planckstraße 1, 64291 Darmstadt, Germany and Institut f¨urKernphysik (Theoriezentrum), Fachbereich Physik, Technische Universit¨atDarmstadt, Schlossgartenstraße 2, 64298 Darmstadt, Germany Gabriel Mart´ınez-Pinedoyy GSI Helmholtzzentrum f¨urSchwerionenforschung, Planckstraße 1, 64291 Darmstadt, Germany; Institut f¨urKernphysik (Theoriezentrum), Fachbereich Physik, Technische Universit¨atDarmstadt, Schlossgartenstraße 2, 64298 Darmstadt, Germany; and Helmholtz Forschungsakademie Hessen f¨urFAIR, GSI Helmholtzzentrum f¨urSchwerionenforschung, Planckstraße 1, 64291 Darmstadt, Germany Friedrich-Karl Thielemannzz Department of Physics, University of Basel, Klingelbergstrasse 82, 4056 Basel, Switzerland and GSI Helmholtzzentrum f¨urSchwerionenforschung, Planckstraße 1, 64291 Darmstadt, Germany (Dated: February 2, 2021) The production of about half of the heavy elements found in nature is assigned to a spe- cific astrophysical nucleosynthesis process: the rapid neutron capture process (r-process). Although this idea has been postulated more than six decades ago, the full understand- ing faces two types of uncertainties/open questions: (a) The nucleosynthesis path in the nuclear chart runs close to the neutron-drip line, where presently only limited experi- arXiv:1901.01410v3 [astro-ph.HE] 1 Feb 2021 mental information is available, and one has to rely strongly on theoretical predictions for nuclear properties. -

Nuclear Structure Features of Very Heavy and Superheavy Nuclei—Tracing Quantum Mechanics Towards the ‘Island of Stability’
Physica Scripta INVITED COMMENT Related content - Super-heavy nuclei Nuclear structure features of very heavy and Sigurd Hofmann superheavy nuclei—tracing quantum mechanics - New elements - approaching towards the ‘island of stability’ S Hofmann - Review of metastable states in heavy To cite this article: D Ackermann and Ch Theisen 2017 Phys. Scr. 92 083002 nuclei G D Dracoulis, P M Walker and F G Kondev View the article online for updates and enhancements. Recent citations - Production cross section and decay study of Es243 and Md249 R. Briselet et al - Colloquium : Superheavy elements: Oganesson and beyond S. A. Giuliani et al - Neutron stardust and the elements of Earth Brett F. Thornton and Shawn C. Burdette This content was downloaded from IP address 213.131.7.107 on 26/02/2019 at 12:10 | Royal Swedish Academy of Sciences Physica Scripta Phys. Scr. 92 (2017) 083002 (83pp) https://doi.org/10.1088/1402-4896/aa7921 Invited Comment Nuclear structure features of very heavy and superheavy nuclei—tracing quantum mechanics towards the ‘island of stability’ D Ackermann1,2 and Ch Theisen3 1 Grand Accélérateur National d’Ions Lourds—GANIL, CEA/DSM-CNRS/IN2P3, Bd. Becquerel, BP 55027, F-14076 Caen, France 2 GSI Helmholtzzentrum für Schwerionenforschung, Planckstr. 1, D-62491 Darmstadt, Germany 3 Irfu, CEA, Université Paris-Saclay, F-91191 Gif-sur-Yvette, France E-mail: [email protected] Received 13 March 2017, revised 19 May 2017 Accepted for publication 13 June 2017 Published 18 July 2017 Abstract The quantum-mechanic nature of nuclear matter is at the origin of the vision of a region of enhanced stability at the upper right end of the chart of nuclei, the so-called ‘island of stability’. -

About That Atomic Weight
BOOKS & ARTS All about that atomic weight creating and proving the existence of these ele- Shaughnessy was involved in the discovery of ments, the stories in the book almost certainly all the recently discovered elements, and was would have been rejected as implausible fiction also the undisputed champion of the unoffi- were they included in a script for a blockbuster cial LLNL contest to see who could watch Star movie on the Transfermium Wars. Wars: The Force Awakens the most times in Chapman’s book is part science, part his- theatres. These stories will undoubtedly make tory, part multi-subject biography told chrono- Superheavy more accessible to audiences with logically with his travelogue interspersed limited knowledge of nuclear chemistry, such throughout. When he reaches Berkeley, he as high school and college chemistry students. realizes, as I once did, that one should pack a Although Chapman demonstrates a deep sweatshirt or two when visiting the Bay Area knowledge of science fiction, one might expect during the summer. Chapman also makes stops the next meeting of the Avengers to be a little in Sweden, Japan, Italy and several other coun- awkward when Thor cites Chapman’s asser- tries to trace the steps of the element hunters tion that thorium is the only comic book char- who pushed the limits of nuclear chemistry to acter to appear on the periodic table. Captain fill in the periodic table. In particular, Chapman America and Iron Man might disagree with Superheavy: Making and Breaking the spends time at the Joint Institute for Nuclear that claim. Chapman should be prepared as Periodic Table Research in Dubna, Russia, interviewing Yuri Titanium Man, Cobalt Man and all the Metal Edited by Kit Chapman Oganessian, who lends his name to element Men also might pay him a visit in the not too Bloomsbury Sigma, 2019, 304pp, £16.99.