2018 Annual Results Presentation
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
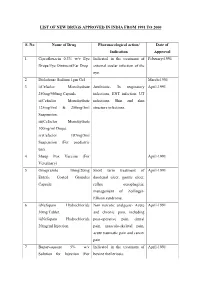
List of New Drugs Approved in India from 1991 to 2000
LIST OF NEW DRUGS APPROVED IN INDIA FROM 1991 TO 2000 S. No Name of Drug Pharmacological action/ Date of Indication Approval 1 Ciprofloxacin 0.3% w/v Eye Indicated in the treatment of February-1991 Drops/Eye Ointment/Ear Drop external ocular infection of the eye. 2 Diclofenac Sodium 1gm Gel March-1991 3 i)Cefaclor Monohydrate Antibiotic- In respiratory April-1991 250mg/500mg Capsule. infections, ENT infection, UT ii)Cefaclor Monohydrate infections, Skin and skin 125mg/5ml & 250mg/5ml structure infections. Suspension. iii)Cefaclor Monohydrate 100mg/ml Drops. iv)Cefaclor 187mg/5ml Suspension (For paediatric use). 4 Sheep Pox Vaccine (For April-1991 Veterinary) 5 Omeprazole 10mg/20mg Short term treatment of April-1991 Enteric Coated Granules duodenal ulcer, gastric ulcer, Capsule reflux oesophagitis, management of Zollinger- Ellison syndrome. 6 i)Nefopam Hydrochloride Non narcotic analgesic- Acute April-1991 30mg Tablet. and chronic pain, including ii)Nefopam Hydrochloride post-operative pain, dental 20mg/ml Injection. pain, musculo-skeletal pain, acute traumatic pain and cancer pain. 7 Buparvaquone 5% w/v Indicated in the treatment of April-1991 Solution for Injection (For bovine theileriosis. Veterinary) 8 i)Kitotifen Fumerate 1mg Anti asthmatic drug- Indicated May-1991 Tablet in prophylactic treatment of ii)Kitotifen Fumerate Syrup bronchial asthma, symptomatic iii)Ketotifen Fumerate Nasal improvement of allergic Drops conditions including rhinitis and conjunctivitis. 9 i)Pefloxacin Mesylate Antibacterial- In the treatment May-1991 Dihydrate 400mg Film Coated of severe infection in adults Tablet caused by sensitive ii)Pefloxacin Mesylate microorganism (gram -ve Dihydrate 400mg/5ml Injection pathogens and staphylococci). iii)Pefloxacin Mesylate Dihydrate 400mg I.V Bottles of 100ml/200ml 10 Ofloxacin 100mg/50ml & Indicated in RTI, UTI, May-1991 200mg/100ml vial Infusion gynaecological infection, skin/soft lesion infection. -

Protonix Protonix
NDA 22-020 Page 4 PROTONIX® (pantoprazole sodium) Delayed-Release Tablets PROTONIX® (pantoprazole sodium) For Delayed-Release Oral Suspension Rx only DESCRIPTION ® The active ingredient in PROTONIX (pantoprazole sodium) Delayed-Release Tablets and ® PROTONIX (pantoprazole sodium) For Delayed-Release Oral Suspension is a substituted benzimidazole, sodium 5-(difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridinyl)methyl] sulfinyl]- 1H-benzimidazole sesquihydrate, a compound that inhibits gastric acid secretion. Its empirical formula is C16H14F2N3NaO4S x 1.5 H2O, with a molecular weight of 432.4. The structural formula is: Pantoprazole sodium sesquihydrate is a white to off-white crystalline powder and is racemic. Pantoprazole has weakly basic and acidic properties. Pantoprazole sodium sesquihydrate is freely soluble in water, very slightly soluble in phosphate buffer at pH 7.4, and practically insoluble in n-hexane. The stability of the compound in aqueous solution is pH-dependent. The rate of degradation increases with decreasing pH. At ambient temperature, the degradation half-life is approximately 2.8 hours at pH 5.0 and approximately 220 hours at pH 7.8. PROTONIX (pantoprazole sodium) is supplied as a delayed-release tablet for oral administration, available in 2 strengths (40 mg and 20 mg); and as delayed-release granules for oral administration, available in the 40 mg strength. NDA 22-020 Page 5 Each PROTONIX (pantoprazole sodium) Delayed-Release tablet contains 45.1 mg or 22.6 mg of pantoprazole sodium sesquihydrate (equivalent to 40 mg or 20 mg pantoprazole, respectively) with the following inactive ingredients: calcium stearate, crospovidone, hypromellose, iron oxide, mannitol, methacrylic acid copolymer, polysorbate 80, povidone, propylene glycol, sodium carbonate, sodium lauryl sulfate, titanium dioxide, and triethyl citrate. -

Proton Pump Inhibitors
MEDICATION POLICY: Proton Pump Inhibitors Generic Name: Proton Pump Inhibitors Preferred: Esomeprazole (generic), Lansoprazole (generic), Omeprazole (generic), Therapeutic Class or Brand Name: Proton Pantoprazole (generic), and Rabeprazole Pump Inhibitors (generic) Applicable Drugs (if Therapeutic Class): Non-preferred: Aciphex® (rabeprazole), Esomeprazole (generic), Lansoprazole Dexilant® (dexlansoprazole), Nexium® (generic), Omeprazole (generic), Pantoprazole (esomeprazole), Not Medically Necessary: (generic), and Rabeprazole (generic). Omeprazole/Sodium Bicarbonate (generic), Aciphex® (rabeprazole), Dexilant® Zegerid® (omeprazole/sodium bicarbonate). (dexlansoprazole), Nexium® (esomeprazole), Prevacid® (lansoprazole), Prilosec® Date of Origin: 2/1/2013 (omeprazole), and Protonix® (pantoprazole). Date Last Reviewed / Revised: 12/23/2020 Omeprazole/Sodium Bicarbonate (generic), Zegerid® (omeprazole/sodium bicarbonate). Policy also applies to any other Proton Pump Inhibitors not listed. GPI Code: 4927002000, 4927002510, 4927004000, 4927006000, 4927007010, 4927007610, 4999600260 PRIOR AUTHORIZATION CRITERIA (May be considered medically necessary when criteria I and II are met) I. Documented diagnosis of one of the following A through G: A. Gastroesophageal reflux disease (GERD). B. Erosive esophagitis. C. Gastric ulcers. D. Risk reduction of NSAID-associated gastric ulcer. E. Duodenal ulcers. F. Eradication of H. pylori. G. Hypersecretory conditions II. Non-preferred PPIs require documented trials and failures of all generic PPIs. EXCLUSION -

2019 Chinese Guideline for the Management of Hypertension in the Elderly
Journal of Geriatric Cardiology (2019) 16: 6799 ©2019 JGC All rights reserved; www.jgc301.com Guidelines Open Access 2019 Chinese guideline for the management of hypertension in the elderly Hypertension Branch of Chinese Geriatrics Society National Clinical Research Center of the Geriatric Diseases-Chinese Alliance of Geriatric Cardiovascular Disease Guideline Writing And Review Committee Co-Chairs: Qi HUA*, Li FAN* Vice Chairs: Jun CAI, Lu-Yuan CHEN, Wei-Wei CHEN, Ping-Jin GAO, Yi-Fang GUO, Qing HE, Jing LI, Nan-Fang LI, Wei-Min LI, Yue LI, Mei-Lin LIU, Ning-Ling SUN, Wen WANG, Liang-Di XIE, Jin-Gang YANG, Hong YUAN Guideline Writing Committee Members Jing LI, Qi HUA, Li FAN, Jun CAI, Lu-Yuan CHEN, Wei-Wei CHEN, Xiao-Ping CHEN, Yi-Fang GUO, Qing HE, Yi-Xin HU, Yi-Nong JIANG, Nan-Fang LI, Wei-Min LI, Yan LI, Yue LI, Yong LI, Qing-Feng MA, Lin PI, Hai-Qing SONG, Xi-Peng SUN, Qing WANG, Zeng-Wu WANG, Hai-Ying WU, Hai-Yun WU, Liang-Di XIE, Jin-Gang YANG, Wei YANG Guideline Review Committee Members (Listed in alphabetic order by last name in Chinese Pinyin) Jun CAI, Jian CAO, Bu-Xing CHEN, Hong CHEN, Lu-Yuan CHEN, Wei-Wei CHEN, Xiao-Ping CHEN, Yuan-Yuan CHEN, Hong-Liang CONG, Ai-Min DANG, Li FAN, Zhen-Xing FAN, Ning-Yuan FANG, Ying-Qing FENG, Yan FU, Hai-Qing GAO, Ping-Jin GAO, Cai-Xia GUO, Jin-Cheng GUO, Jun GUO, Yi-Fang GUO, Qing-Hua HAN, Qing HE, Da-Yi HU, Shao-Dong HU, Yi-Xin HU, Qi HUA, Yi-Nong JIANG, Bo-Yu LI, Dong-Bao LI, Hong-Wei LI, Jing LI, Nan-Fang LI, Wei-Min LI, Yan-Fang LI, Yan LI, Yue LI, Yong LI, Li LIN, Zhan-Yi LIN, De-Ping -

PHRP March 2015
March 2015; Vol. 25(2):e2521518 doi: http://dx.doi.org/10.17061/phrp2521518 www.phrp.com.au Research Manual versus automated coding of free-text self-reported medication data in the 45 and Up Study: a validation study Danijela Gnjidica,b,i, Sallie-Anne Pearsona,c, Sarah N Hilmerb,d, Jim Basilakise, Andrea L Schaffera, Fiona M Blythb,f,g and Emily Banksg,h, on behalf of the High Risk Prescribing Investigators a Faculty of Pharmacy, University of Sydney, NSW, Australia b Sydney Medical School, University of Sydney, NSW, Australia c Sydney School of Public Health, University of Sydney, NSW, Australia d Royal North Shore Hospital and Kolling Institute of Medical Research, Sydney, NSW, Australia e School of Computing, Engineering and Mathematics, University of Western Sydney, NSW, Australia f Centre for Education and Research on Ageing (CERA), Concord Hospital, Sydney, NSW, Australia g The Sax Institute, Sydney, NSW, Australia h National Centre for Epidemiology and Population Health, Australian National University, Canberra, ACT i Corresponding author: [email protected] Article history Abstract Publication date: March 2015 Background: Increasingly, automated methods are being used to code free- Citation: Gnjidic D, Pearson S, Hilmer S, text medication data, but evidence on the validity of these methods is limited. Basilakis J, Schaffer AL, Blyth FM, Banks E. To examine the accuracy of automated coding of previously keyed Manual versus automated coding of free-text Aim: in free-text medication data compared with manual coding of original self-reported medication data in the 45 and Up Study: a validation study. Public Health handwritten free-text responses (the ‘gold standard’). -

Acid-Reducing Medications: Interactions with Harvoni and Epclusa
November 2016 | www.hepatitis.va.gov Acid-Reducing Medications: Interactions with Harvoni® and Epclusa® Sofosbuvir/Velpatasvir = Epclusa® Ledipasvir/Sofosbuvir = Harvoni® • Acid-reducing medications can decrease the effectiveness of Harvoni® and Epclusa® and could prevent successful cure. • Let your provider know if you are taking any acid-reducing medications—either prescribed to you or purchased over the counter. Types of Acid-Reducing What to Do if What to Do if Medications Taking Epclusa® Taking Harvoni® Antacids Separate taking the antacid and Separate taking the antacid and Epclusa® by 4 hours Harvoni® by 4 hours Combinations of aluminum, magnesium, and calcium. Brand names include: Alka-Seltzer® Maalox® Mylanta® Rolaids® TUMS® H2 Blockers Do not take more than the Do not take more than the recommended doses recommended doses Cimetidine (Tagamet®) Famotidine (Pepcid®) May take together with Epclusa® May take together with Harvoni® Nizatidine (Axid®) or 12 hours apart or 12 hours apart Ranitidine (Zantac®) Proton Pump Inhibitors (PPI) It is best to avoid PPIs during treat- It is best to avoid PPIs during ment with Epclusa®. If there are no treatment with Harvoni®. If there Esomeprazole (Nexium®) alternatives, follow these instructions: are no alternatives, follow these Lansoprazole (Prevacid®) instructions: Omeprazole (Prilosec®) Take Epclusa® with food Pantoprazole (Protonix®) Take the proton pump inhibitor 4 Take Harvoni® and the proton Rabeprazole (Aciphex®) hours after taking Epclusa® pump inhibitor at the same time in the morning after fasting overnight Do not take the proton pump (on an empty stomach) inhibitor at the same time as Epclusa® Do not take proton pump Do not take proton pump inhibitors inhibitors at more than the at more than the recommended dose recommended dose U.S. -

Risks of Proton Pump Inhibitors for Gastroesophageal Reflux Disease and a Diet Alternative
Journal of Nutritional Health & Food Engineering Literature Review Open Access Risks of proton pump inhibitors for gastroesophageal reflux disease and a diet alternative Abstract Volume 10 Issue 1 - 2020 Gastroesophageal reflux disease (GERD) is one of the most common digestive conditions Mark Stengler NMD treated by gastroenterologists. The most commonly prescribed drugs for GERD are proton Stengler Center for Integrative Medicine, USA pump inhibitors (PPIs) that reduce stomach acid. While these medications are effective for relieving GERD symptoms, their long-term use is associated with several side effects Correspondence: Mark Stengler NMD, Stengler Center for and chronic diseases. Several publications have questioned the long-term safety of PPIs. Integrative Medicine, 324 Encinitas Blvd. Moreover, recent studies have demonstrated that the Mediterranean Diet may be more Encinitas, CA 92024 1-760-274-2377, effective than PPIs for people suffering from GERD. Email Keywords: GERD, mediterranean diet, proton pump inhibitors, PPI, acid reflux, LES, Received: April 14, 2020 | Published: April 28, 2020 lower esophageal sphincter, omeprazole, prilosec Introduction Proton pump inhibitors to treat GERD Gastroesophageal reflux disease (GERD) is one of the most common The most commonly prescribed drugs for GERD are proton digestive conditions treated by gastroenterologists and primary care pump inhibitors (PPIs).2 PPIs currently available on prescription in doctors. Typical symptoms include heartburn, regurgitation, and the USA are: dexlansoprazole -

Proton-Pump Inhibitor (PPI) Use
Proton-Pump Inhibitor (PPI) Use The FDA has issued multiple warnings on the long-term use of PPIs. These include: increased risk of C. difficile infection1, hypomagnesemia2, and fractures of the hip, wrist, and spine3. Therefore, prudent prescribing of PPIs is warranted. The FDA recommends use of the lowest dose and shortest duration of PPI therapy appropriate for the condition being treated1-3. Patient compliance, time of administration (prior to meals), and dietary indiscretions (i.e. alcohol or irritating foods) should be assessed prior to titration of PPI doses. Indication Treatment Duration Gastroesophageal reflux disease Initial 8 week course for symptom (GERD)6 relief or esophagitis Symptomatic relief Maintenance therapy determined Omeprazole 20 mg PO by response and severity of Acute healing of erosive or ulcerative once daily disease esophagitis OR Pantoprazole 40mg PO For patients that require more Maintenance healing of erosive or once daily long-term therapy, consider a ulcerative esophagitis trial of a lower dose, on-demand therapy, or intermittent therapy to minimize exposure Stress ulcer prophylaxis Transition to PO when possible Reserve PPIs for critically ill patients Continue until resolution of with increased risk of bleeding:4,5 underlying risk factors and/or At least one of the following: critical illness - Coagulopathy (platelet count <50,000 mm3, INR >1.5, or aPTT Recommend discontinuation at >2x control) discharge, unless there is - Mechanical ventilation >48 hours Omeprazole another indication for use - History -

Short‑ and Long‑Term Treatment with Angiotensin‑Converting Enzyme
EXPERIMENTAL AND THERAPEUTIC MEDICINE 21: 14, 2021 Short‑ and long‑term treatment with angiotensin‑converting enzyme inhibitors or calcium channel blockers for the prevention of diabetic nephropathy progression: A meta‑analysis JIALANG LIANG1*, JIARONG LAN2*, QIZHI TANG1, WENJING LING3 and MIN LI4 1Endocrinology Department, Integrated Traditional Chinese and Western Medicine Hospital of Guangdong Province, Foshan, Guangdong 528200; 2Nephrology Department, Huzhou Hospital of Traditional Chinese Medicine Affiliated Zhejiang University of Traditional Chinese Medicine, Huzhou, Zhejiang 313000;3 Emergency Department, Integrated Traditional Chinese and Western Medicine Hospital of Guangdong Province, Foshan, Guangdong 528200; 4Endocrinology Department, Huzhou Hospital of Traditional Chinese Medicine Affiliated Zhejiang University of Traditional Chinese Medicine, Huzhou, Zhejiang 313000, P.R. China Received May 21, 2020; Accepted October 14, 2020 DOI: 10.3892/etm.2020.9446 Abstract. Treatments with angiotensin‑converting enzyme better outcomes with ACE inhibitors [odds ratio (OR), 0.70; (ACE) inhibitors or calcium channel blockers (CCBs) may 95% CI, 0.49‑1.00; P=0.05]. There was no statistically signifi‑ delay the development of albuminuria in patients with early cant difference between ACE inhibitors and CCBs regarding diabetic nephropathy. However, evidence in the literature the progression from microalbuminuria to macroalbuminuria has not been consistent. The present meta‑analysis aimed to (OR, 1.78; 95% CI, 0.82‑3.87; P=0.15). In conclusion, the compare the short‑ and long‑term therapeutic effects of ACE present study indicated that the antiproteinuric efficacy of inhibitors and CCBs (when used separately) for preventing the CCBs may be less than that of ACE inhibitors after short‑term progression of nephropathy in patients with diabetes mellitus. -

Levamlodipine)Tablets, for Oral Use
HIGHLIGHTS OF PRESCRIBING INFORMATION ------------------------WARNINGS AND PRECAUTIONS---------------------- These highlights do not include all the information needed to use • Symptomatic hypotension is possible, particularly in patients with CONJUPRI safely and effectively. See full prescribing information severe aortic stenosis. However, acute hypotension is unlikely. for CONJUPRI. (5.1) • Worsening angina and acute myocardial infarction can develop CONJUPRI®(levamlodipine)tablets, for oral use. after starting or increasing the dose of amlodipine, particularly in Initial U.S. Approval: 1992 patients with severe obstructive coronary artery disease. (5.2) • Titrate slowly in patients with severe hepatic impairment. (5.3) -----------------------------INDICATIONS AND USAGE------------------------- CONJUPRI is calcium channel blocker and may be used alone or in -------------------------------ADVERSE REACTIONS----------------------------- combination with other antihypertensive agents for the treatment of Most common adverse reactions to amlodipine is edema which hypertension, to lower blood pressure. Lowering blood pressure occurred in a dose related manner. Other adverse experiences not reduces the risk of fatal and nonfatal cardiovascular events, primarily dose related but reported with an incidence >1.0% are fatigue, nausea, strokes and myocardial infarctions. abdominal pain and somnolence. (6) ----------------------DOSAGE AND ADMINISTRATION---------------------- To report SUSPECTED ADVERSE REACTIONS, call CSPC Ouyi • Adult recommended starting dose: 2.5 mg orally once daily with Pharmaceutical Co., Ltd at 1-877-436-7220 or FDA at 1-800-FDA maximum dose 5 mg once daily. (2.1) 1088 or www.fda.gov/medwatch. o Small, fragile, or elderly patients, or patients with hepatic insufficiency may be started on 1.25 mg once daily. (2.1) ------------------------------DRUG INTERACTIONS------------------------------ • Pediatric starting dose: 1.25 mg to 2.5 mg once daily. -

Pharmacokinetics, Pharmacodynamics
Supplementary Pharmacokinetics, Pharmacodynamics and Drug-Drug Interactions of New Anti-Migraine Drugs–Lasmiditan, Gepants, and Calcitonin-Gene-Related Peptide (CGRP) Receptor Monoclonal Antibodies Danuta Szkutnik-Fiedler Table S1. Possible drug-drug interactions of lasmiditan [14,28,31,35–38,40,42,45–47]. The risk or severity of Serum concentration of the Serum concentration of Lasmiditan may serotonin syndrome can following drugs (P-gp lasmiditan (P-gp substrate) increase the be potentially increased and/or BCRP substrates) may potentially increase bradycardic when lasmiditan is may potentially increase when it is combined with effects of the combined with the when combined with the following drugs3,. following drugs. following drugs1,. lasmiditan2,. 5-hydroxytryptophan* afatinib acebutolol alfentanil* alpelisib amlodipine almotriptan* ambrisentan atenolol amitriptiline* apixaban betaxolol amoxapine* belinostat carteolol buspirone* bisoprolol carvedilol citalopram* brentuximab vedotin diltiazem clomipramine* cabazitaxel esmolol cyclobenzaprine* ceritinib felodipine desipramine* cladribine isradipine desvenlavaxine* cobimetinib clobazam ivabradine dexfenfluramine* colchicine* daclatasvir labetalol dextromethorphan* cyclosporine erythromycin levobetaxolol dihydroergotamine* daunorubicin fexofenadine levobunolol dolasetron* delafloxacin lapatinib methyldopa doxepin* digitoxin ritonavir metipranolol doxepin topical* digoxin metoprolol duloxetine* donepezil nadolol eletriptan* doxorubicin nebivolol ergotamine* edoxaban* nicardipine escitalopram* -

New and Future Drug Development for Gastroesophageal Reflux Disease
J Neurogastroenterol Motil, Vol. 20 No. 1 January, 2014 pISSN: 2093-0879 eISSN: 2093-0887 http://dx.doi.org/10.5056/jnm.2014.20.1.6 JNM Journal of Neurogastroenterology and Motility Review New and Future Drug Development for Gastroesophageal Reflux Disease Carla Maradey-Romero and Ronnie Fass* The Esophageal and Swallowing Center, Division of Gastroenterology and Hepatology, MetroHealth Medical Center, Case Western Reserve University, Cleveland, Ohio, USA Medical therapy remains the most popular treatment for gastroesophageal reflux disease (GERD). Whilst interest in drug devel- opment for GERD has declined over the last few years primarily due to the conversion of most proton pump inhibitor (PPI)’s to generic and over the counter compounds, there are still numerous areas of unmet needs in GERD. Drug development has been focused on potent histamine type 2 receptor antagonist’s, extended release PPI’s, PPI combination, potassium-competitive acid blockers, transient lower esophageal sphincter relaxation reducers, prokinetics, mucosal protectants and esophageal pain modulators. It is likely that the aforementioned compounds will be niched for specific areas of unmet need in GERD, rather than compete with the presently available anti-reflux therapies. (J Neurogastroenterol Motil 2014;20:6-16) Key Words Erosive esophagitis; Gastroesophageal reflux; Heartburn; Proton pump inhibitors Most patients with GERD fall into 1 of 3 categories: non- erosive reflux disease (NERD), erosive esophagitis (EE), and Introduction Barrett’s esophagus (BE). The 2 main phenotypes of GERD, Gastroesophageal reflux disease (GERD) is a common con- NERD and EE, appear to have different pathophysiological and dition that develops when reflux of stomach contents cause trou- clinical characteristics.