Acid-Reducing Medications: Interactions with Harvoni and Epclusa
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
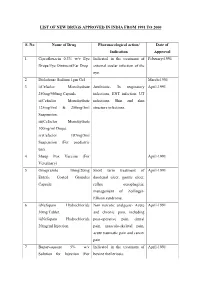
List of New Drugs Approved in India from 1991 to 2000
LIST OF NEW DRUGS APPROVED IN INDIA FROM 1991 TO 2000 S. No Name of Drug Pharmacological action/ Date of Indication Approval 1 Ciprofloxacin 0.3% w/v Eye Indicated in the treatment of February-1991 Drops/Eye Ointment/Ear Drop external ocular infection of the eye. 2 Diclofenac Sodium 1gm Gel March-1991 3 i)Cefaclor Monohydrate Antibiotic- In respiratory April-1991 250mg/500mg Capsule. infections, ENT infection, UT ii)Cefaclor Monohydrate infections, Skin and skin 125mg/5ml & 250mg/5ml structure infections. Suspension. iii)Cefaclor Monohydrate 100mg/ml Drops. iv)Cefaclor 187mg/5ml Suspension (For paediatric use). 4 Sheep Pox Vaccine (For April-1991 Veterinary) 5 Omeprazole 10mg/20mg Short term treatment of April-1991 Enteric Coated Granules duodenal ulcer, gastric ulcer, Capsule reflux oesophagitis, management of Zollinger- Ellison syndrome. 6 i)Nefopam Hydrochloride Non narcotic analgesic- Acute April-1991 30mg Tablet. and chronic pain, including ii)Nefopam Hydrochloride post-operative pain, dental 20mg/ml Injection. pain, musculo-skeletal pain, acute traumatic pain and cancer pain. 7 Buparvaquone 5% w/v Indicated in the treatment of April-1991 Solution for Injection (For bovine theileriosis. Veterinary) 8 i)Kitotifen Fumerate 1mg Anti asthmatic drug- Indicated May-1991 Tablet in prophylactic treatment of ii)Kitotifen Fumerate Syrup bronchial asthma, symptomatic iii)Ketotifen Fumerate Nasal improvement of allergic Drops conditions including rhinitis and conjunctivitis. 9 i)Pefloxacin Mesylate Antibacterial- In the treatment May-1991 Dihydrate 400mg Film Coated of severe infection in adults Tablet caused by sensitive ii)Pefloxacin Mesylate microorganism (gram -ve Dihydrate 400mg/5ml Injection pathogens and staphylococci). iii)Pefloxacin Mesylate Dihydrate 400mg I.V Bottles of 100ml/200ml 10 Ofloxacin 100mg/50ml & Indicated in RTI, UTI, May-1991 200mg/100ml vial Infusion gynaecological infection, skin/soft lesion infection. -

Medicines That Affect Fluid Balance in the Body
the bulk of stools by getting them to retain liquid, which encourages the Medicines that affect fluid bowels to push them out. balance in the body Osmotic laxatives e.g. Lactulose, Macrogol - these soften stools by increasing the amount of water released into the bowels, making them easier to pass. Older people are at higher risk of dehydration due to body changes in the ageing process. The risk of dehydration can be increased further when Stimulant laxatives e.g. Senna, Bisacodyl - these stimulate the bowels elderly patients are prescribed medicines for chronic conditions due to old speeding up bowel movements and so less water is absorbed from the age. stool as it passes through the bowels. Some medicines can affect fluid balance in the body and this may result in more water being lost through the kidneys as urine. Stool softener laxatives e.g. Docusate - These can cause more water to The medicines that can increase risk of dehydration are be reabsorbed from the bowel, making the stools softer. listed below. ANTACIDS Antacids are also known to cause dehydration because of the moisture DIURETICS they require when being absorbed by your body. Drinking plenty of water Diuretics are sometimes called 'water tablets' because they can cause you can reduce the dry mouth, stomach cramps and dry skin that is sometimes to pass more urine than usual. They work on the kidneys by increasing the associated with antacids. amount of salt and water that comes out through the urine. Diuretics are often prescribed for heart failure patients and sometimes for patients with The major side effect of antacids containing magnesium is diarrhoea and high blood pressure. -

Nexium Control, Esomeprazole
27 June 2013 EMA/498929/2013 Committee for Medicinal Products for Human Use (CHMP) Assessment report Nexium Control International non-proprietary name: esomeprazole Procedure No. EMEA/H/C/002618 Note Assessment report as adopted by the CHMP with all information of a commercially confidential nature deleted. 7 Westferry Circus ● Canary Wharf ● London E14 4HB ● United Kingdom Telephone +44 (0)20 7418 8400 Facsimile +44 (0)20 7418 8613 E -mail [email protected] Website www.ema.europa.eu An agency of the European Union © European Medicines Agency, 2013. Reproduction is authorised provided the source is acknowledged. Product information Marketing authorisation application Name of the medicinal product: Nexium Control Applicant: AstraZeneca AB Building 411A, Floor 4 S - 151 85 Södertälje SWEDEN Active substance: esomeprazole (as magnesium trihydrate) International Nonproprietary Name/Common Name: esomeprazole Pharmaco-therapeutic group Proton pump inhibitors (ATC Code): (A02BC05) Therapeutic indication: Nexium Control is indicated for the short-term treatment of reflux symptoms (e.g. heartburn and acid regurgitation) in adults. Pharmaceutical form: Gastro-resistant tablet Strength: 20 mg Route of administration: Oral use Packaging: blister (PVC/PVDC) Package sizes: 7 tablets and 14 tablets Assessment report EMA/498929/2013 Page 2/70 Table of contents 1. Background information on the procedure .............................................. 6 1.1. Submission of the dossier ..................................................................................... -

Protonix Protonix
NDA 22-020 Page 4 PROTONIX® (pantoprazole sodium) Delayed-Release Tablets PROTONIX® (pantoprazole sodium) For Delayed-Release Oral Suspension Rx only DESCRIPTION ® The active ingredient in PROTONIX (pantoprazole sodium) Delayed-Release Tablets and ® PROTONIX (pantoprazole sodium) For Delayed-Release Oral Suspension is a substituted benzimidazole, sodium 5-(difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridinyl)methyl] sulfinyl]- 1H-benzimidazole sesquihydrate, a compound that inhibits gastric acid secretion. Its empirical formula is C16H14F2N3NaO4S x 1.5 H2O, with a molecular weight of 432.4. The structural formula is: Pantoprazole sodium sesquihydrate is a white to off-white crystalline powder and is racemic. Pantoprazole has weakly basic and acidic properties. Pantoprazole sodium sesquihydrate is freely soluble in water, very slightly soluble in phosphate buffer at pH 7.4, and practically insoluble in n-hexane. The stability of the compound in aqueous solution is pH-dependent. The rate of degradation increases with decreasing pH. At ambient temperature, the degradation half-life is approximately 2.8 hours at pH 5.0 and approximately 220 hours at pH 7.8. PROTONIX (pantoprazole sodium) is supplied as a delayed-release tablet for oral administration, available in 2 strengths (40 mg and 20 mg); and as delayed-release granules for oral administration, available in the 40 mg strength. NDA 22-020 Page 5 Each PROTONIX (pantoprazole sodium) Delayed-Release tablet contains 45.1 mg or 22.6 mg of pantoprazole sodium sesquihydrate (equivalent to 40 mg or 20 mg pantoprazole, respectively) with the following inactive ingredients: calcium stearate, crospovidone, hypromellose, iron oxide, mannitol, methacrylic acid copolymer, polysorbate 80, povidone, propylene glycol, sodium carbonate, sodium lauryl sulfate, titanium dioxide, and triethyl citrate. -

Receptor Antagonist (H RA) Shortages | May 25, 2020 2 2 2 GERD4,5 • Take This Opportunity to Determine If Continued Treatment Is Necessary
H2-receptor antagonist (H2RA) Shortages Background . 2 H2RA Alternatives . 2 Therapeutic Alternatives . 2 Adults . 2 GERD . 3 PUD . 3 Pediatrics . 3 GERD . 3 PUD . 4 Tables Table 1: Health Canada–Approved Indications of H2RAs . 2 Table 2: Oral Adult Doses of H2RAs and PPIs for GERD . 4 Table 3: Oral Adult Doses of H2RAs and PPIs for PUD . 5 Table 4: Oral Pediatric Doses of H2RAs and PPIs for GERD . 6 Table 5: Oral Pediatric Doses of H2RAs and PPIs for PUD . 7 References . 8 H2-receptor antagonist (H2RA) Shortages | May 25, 2020 1 H2-receptor antagonist (H2RA) Shortages BACKGROUND Health Canada recalls1 and manufacturer supply disruptions may be causing shortages of commonly used acid-reducing medications called histamine H2-receptor antagonists (H2RAs) . H2RAs include cimetidine, famotidine, nizatidine and ranitidine . 2 There are several Health Canada–approved indications of H2RAs (see Table 1); this document addresses the most common: gastroesophageal reflux disease (GERD) and peptic ulcer disease (PUD) . 2 TABLE 1: HEALTH CANADA–APPROVED INDICATIONS OF H2RAs H -Receptor Antagonists (H RAs) Health Canada–Approved Indications 2 2 Cimetidine Famotidine Nizatidine Ranitidine Duodenal ulcer, treatment ü ü ü ü Duodenal ulcer, prophylaxis — ü ü ü Benign gastric ulcer, treatment ü ü ü ü Gastric ulcer, prophylaxis — — — ü GERD, treatment ü ü ü ü GERD, maintenance of remission — ü — — Gastric hypersecretion,* treatment ü ü — ü Self-medication of acid indigestion, treatment and prophylaxis — ü† — ü† Acid aspiration syndrome, prophylaxis — — — ü Hemorrhage from stress ulceration or recurrent bleeding, — — — ü prophylaxis ü = Health Canada–approved indication; GERD = gastroesophageal reflux disease *For example, Zollinger-Ellison syndrome . -

Proton Pump Inhibitors
MEDICATION POLICY: Proton Pump Inhibitors Generic Name: Proton Pump Inhibitors Preferred: Esomeprazole (generic), Lansoprazole (generic), Omeprazole (generic), Therapeutic Class or Brand Name: Proton Pantoprazole (generic), and Rabeprazole Pump Inhibitors (generic) Applicable Drugs (if Therapeutic Class): Non-preferred: Aciphex® (rabeprazole), Esomeprazole (generic), Lansoprazole Dexilant® (dexlansoprazole), Nexium® (generic), Omeprazole (generic), Pantoprazole (esomeprazole), Not Medically Necessary: (generic), and Rabeprazole (generic). Omeprazole/Sodium Bicarbonate (generic), Aciphex® (rabeprazole), Dexilant® Zegerid® (omeprazole/sodium bicarbonate). (dexlansoprazole), Nexium® (esomeprazole), Prevacid® (lansoprazole), Prilosec® Date of Origin: 2/1/2013 (omeprazole), and Protonix® (pantoprazole). Date Last Reviewed / Revised: 12/23/2020 Omeprazole/Sodium Bicarbonate (generic), Zegerid® (omeprazole/sodium bicarbonate). Policy also applies to any other Proton Pump Inhibitors not listed. GPI Code: 4927002000, 4927002510, 4927004000, 4927006000, 4927007010, 4927007610, 4999600260 PRIOR AUTHORIZATION CRITERIA (May be considered medically necessary when criteria I and II are met) I. Documented diagnosis of one of the following A through G: A. Gastroesophageal reflux disease (GERD). B. Erosive esophagitis. C. Gastric ulcers. D. Risk reduction of NSAID-associated gastric ulcer. E. Duodenal ulcers. F. Eradication of H. pylori. G. Hypersecretory conditions II. Non-preferred PPIs require documented trials and failures of all generic PPIs. EXCLUSION -

2018 Annual Results Presentation
2018 Annual Results Presentation Sihuan Pharmaceutical Holdings Group Ltd. 四环医药控股集团有限公司 0 Disclaimer The sole purpose of this Presentation (the “Presentation”) is to assist the recipient in deciding whether it wishes to proceed with a further investigation of Sihuan Pharmaceutical Holdings Group Ltd. (the “Company”) and it is not intended to form the basis of any decision to purchase securities, interests or assets in or of the Company. This Presentation does not constitute or contain an offer or invitation or recommendation or solicitation for the sale or purchase of securities, interests or assets in or of the Company and neither this document nor anything contained herein shall form the basis of, or be relied upon in connection with, any contract or commitment whatsoever. Any decision to purchase or subscribe for securities in any offering must be made solely on the basis of the information contained in the prospectus or offering circular issued by the company in connection with such offerings. All the information in this Presentation has been provided by the Company and has not been independently verified. No representation or warranty, express or implied, is or will be made in or in relation to, and no responsibility or liability is or will be accepted by the Company or any of its subsidiaries as to the appropriateness, accuracy, completeness or reliability of, this Presentation or any other written or oral information made available to any interested party or its advisers and any liability therefore is hereby expressly disclaimed. And no reliance should be placed on the accuracy, fairness, completeness or correctness of the information contained in this Presentation. -

Esomeprazole: a New Proton Pump Inhibitor for NSAID-Associated Peptic Ulcers and Dyspepsia
DRUG PROFILE Esomeprazole: a new proton pump inhibitor for NSAID-associated peptic ulcers and dyspepsia Grace Lai-Hung Wong Ulcer and ulcer symptoms related to the use of non-steroidal anti-inflammatory drugs & Joseph JY Sung† (NSAIDs) and aspirin constitute a major global health issue. Despite various attempts to †Author for correspondence prevent and heal injuries inflicted by NSAIDs and aspirin, acid suppression remains one of The Prince of Wales Hospital, Department of Medicine and the cornerstones in the management of NSAID-associated ulcers. Esomeprazole, the Therapeutics, 9/F, S-optical isomer (enantiomer) of omeprazole, suppresses gastric acid secretion by 30–32 Ngan Shing Road, inhibiting the parietal cell membrane enzyme H+/K+-ATPase. With improved bioavailability, Shatin, NT, Hong Kong SAR due to reduced first-pass metabolism in the liver, esomeprazole promises to be more Tel.: +852 2632 3132 potent in acid suppression in the stomach. Similar to omeprazole, the safety profile of Fax: +852 2645 1699 esomeprazole has been well established. Clinical studies comparing esomeprazole with [email protected] other proton pump inhibitors (PPIs) in the healing of NSAID-related ulcer are few. Recent multicenter randomized studies demonstrated that esomeprazole significantly improves dyspeptic symptoms in patients taking nonselective NSAIDs and specific cyclooxygenase-2 inhibitors. Esomeprazole also protects the stomach from aspirin-induced ulcer bleeding. Safety profiles of esomeprazole appear promising. Non-steroidal anti-inflammatory drugs both basal and stimulated acid secretion. PPIs are (NSAIDs) are the most commonly used medica- most effective against meal-induced gastric acid tions for chronic pain and arthritis due to their secretion [3]. -

Comparison of the Efficacies of Proton Pump Inhibitors and H2 Receptor Antagonists in On-Demand Treatment of Gastroesophageal Reflux Disease
8 ORIGINAL ARTICLE COMPARISON OF THE EFFICACIES OF PROTON PUMP INHIBITORS AND H2 RECEPTOR ANTAGONISTS IN ON-DEMAND TREATMENT OF GASTROESOPHAGEAL REFLUX DISEASE Nesibe Vardı1, Emine Köroglu2, Sabah Tüzün3, Can Dolapçıoğlu4, Reşat Dabak5 1Kilimli No: 8104005 Dikmeli Family Health Centre, Duzce, Turkey 2Department of Gastroenterology, Kartal Dr.Lütfi Kırdar Training and Research Hospital, Istanbul, Turkey 3Department of Family Medicine, Kartal Dr. Lütfi Kırdar Training and Research Hospital, Istanbul, Turkey Abstract Objective: On-demand treatment protocols in the maintenance treatment of gastroesophageal reflux disease (GERD) are cost-efficient and easy-to-use treatments.This study aimed to compare the efficacies of H2 receptor antagonists (H2RA) and proton pump inhibitors (PPI) in on-demand treatment of GERD. Methods: Patients with persistent GERD symptoms were enrolledbetween January-November 2015 and were ran- domly separated into two equal groups. The patients in the first group were commenced on ranitidine 300 mg as H2RA and the patients in the second group were commenced on pantoprazole 40 mg. A 4-point Likert-type scale generated by the researchers was applied to evaluate the frequencies of reflux symptoms and their impacts on activities of life and business life. Results: Fifty-two patients were included, of whom 26 (50.0%) were in the PPI group and 26 (50.0%) were in the H2RA group. There were no significant differences between the study groups both before and after treatment in terms of the severity of reflux symptoms. There were significant decreases in the H2RA group in terms of the domains of retrosternal burning sensation, regurgitation, nausea and vomiting, and burping (p=0.036, p=0.027, p=0.020, and p=0.038, respectively). -

Dr. Reddy's – Recall of Ranitidine
Dr. Reddy’s – Recall of ranitidine • On October 23, 2019, Dr. Reddy’s announced the voluntary, consumer-level recall of prescription ranitidine due to potential contamination with N-nitrosodimethylamine (NDMA). • Dr. Reddy’s initially announced retail level recalls for prescription and OTC ranitidine in early October; however, the recall of prescription ranitidine products has been escalated to the consumer level. The recall of OTC ranitidine remains at the retail level. Prescription Ranitidine Capsules Recalled by Dr. Reddy’s Product Description NDC# Ranitidine 150 mg capsules, 60 count bottle 55111-129-60 Ranitidine 150 mg capsules, 500 count bottle 55111-129-05 Ranitidine 300 mg capsules, 30 count bottle 55111-130-30 Ranitidine 300 mg capsules, 100 count bottle 55111-130-01 • Refer to the Dr. Reddy’s announcement for a complete list of the OTC recalled ranitidine products. • Other manufacturers, including Apotex, Perrigo and Sanofi have recently announced retail level recalls of their OTC ranitidine products. • In September, Sandoz issued a consumer level recall of prescription ranitidine products. • These recalls follow a recent FDA statement about NDMA impurities detected in ranitidine medicines. • NDMA is classified as a probable human carcinogen based on results from laboratory tests. NDMA is a known environmental contaminant and found in water and foods, including meats, dairy products, and vegetables. • Ranitidine is an OTC and prescription drug. Ranitidine is an H2 (histamine-2) blocker, which decreases the amount of acid created by the stomach. OTC ranitidine is approved to prevent and relieve heartburn associated with acid ingestion and sour stomach. • Prescription ranitidine is approved for multiple indications, including treatment and prevention of ulcers of the stomach and intestines and treatment of gastroesophageal reflux disease. -

Aciphex (Rabeprazole Sodium)
ACIPHEXÔ çç \ a-sc -**feks\ (rabeprazole sodium) Delayed-Release Tablets DESCRIPTION The active ingredient in ACIPHEXÔ Delayed-Release Tablets is rabeprazole sodium, a substituted benzimidazole that inhibits gastric acid secretion. Rabeprazole sodium is known chemically as 2-[[[4-(3-methoxypropoxy)-3-methyl-2- pyridinyl]-methyl] sulfinyl]-1H–benzimidazole sodium salt. It has an empirical formula of C18H20N3NaO3S and a molecular weight of 381.43. Rabeprazole sodium is a white to slightly yellowish-white solid. It is very soluble in water and methanol, freely soluble in ethanol, chloroform and ethyl acetate and insoluble in ether and n-hexane. The stability of rabeprazole sodium is a function of pH; it is rapidly degraded in acid media, and is more stable under alkaline conditions. The structural formula is: Na N O H3C O CH2 O CH3 S CH2 CH2 N CH2 N RABEPRAZOLE SODIUM ACIPHEXÔ is available for oral administration as delayed-release, enteric-coated tablets containing 20 mg of rabeprazole sodium. Inactive ingredients are mannitol, hydroxypropyl cellulose, magnesium oxide, low-substituted hydroxypropyl cellulose, magnesium stearate, ethylcellulose, hydroxypropyl methylcellulose phthalate, diacetylated monoglycerides, talc, titanium dioxide, carnauba wax, and ferric oxide (yellow) as a coloring agent. NDA 20-973 (AciphexÔ ) 1 Annotated Package Insert March 5, 1999 CLINICAL PHARMACOLOGY Pharmacokinetics and Metabolism ACIPHEXTM delayed-release tablets are enteric-coated to allow rabeprazole sodium, which is acid labile, to pass through TM the stomach relatively intact. After oral administration of 20 mg ACIPHEX , peak plasma concentrations (Cmax) of rabeprazole occur over a range of 2.0 to 5.0 hours (Tmax). The rabeprazole Cmax and AUC are linear over an oral dose range of 10 mg to 40 mg. -

Mast Cell Tumor (Canine)
MAST CELL TUMOR (CANINE) What is a mast cell tumor? A mast cell tumor is a cancer of a specific type of inflammatory cell, usually in the skin. Mast cell tumors may originate from other areas such as the gastrointestinal tract, spleen, liver, or mouth, but they are far more com- monly found on the skin. What are the symptoms of a mast cell tumor? Mast cell tumors on the skin can be red, hairless, and itchy. They can also feel soft or firm, and look like many other types of masses. Because they release histamine, they can grow and shrink periodically. How do you diagnose a mast cell tumor? A fine needle aspirate of the mass is performed and evaluated under a microscope for cytology. Cytology de- termines the type of tumor, however further diagnostics such as a biopsy (histopathology) are needed to further categorize or grade a mast cell tumor. What is the behavior of this type of tumor? Mast cell tumors are graded 1, 2, or 3 based on microscopic analysis of a biopsy specimen. Grade 1 tumors typically behave like benign tumors and have a low potential to spread (metastasize). Grade 3 tumors are aggressive and have a high potential to metastasize to the lymph nodes and possibly the liver, spleen, or bone marrow. Grade 2 tumors may behave either like a Grade 1 or 3 tumor. A newer grading system will determine whether a mast cell tumor is low grade or high grade. Many pathologists will use both systems to help provide as much information as possible.