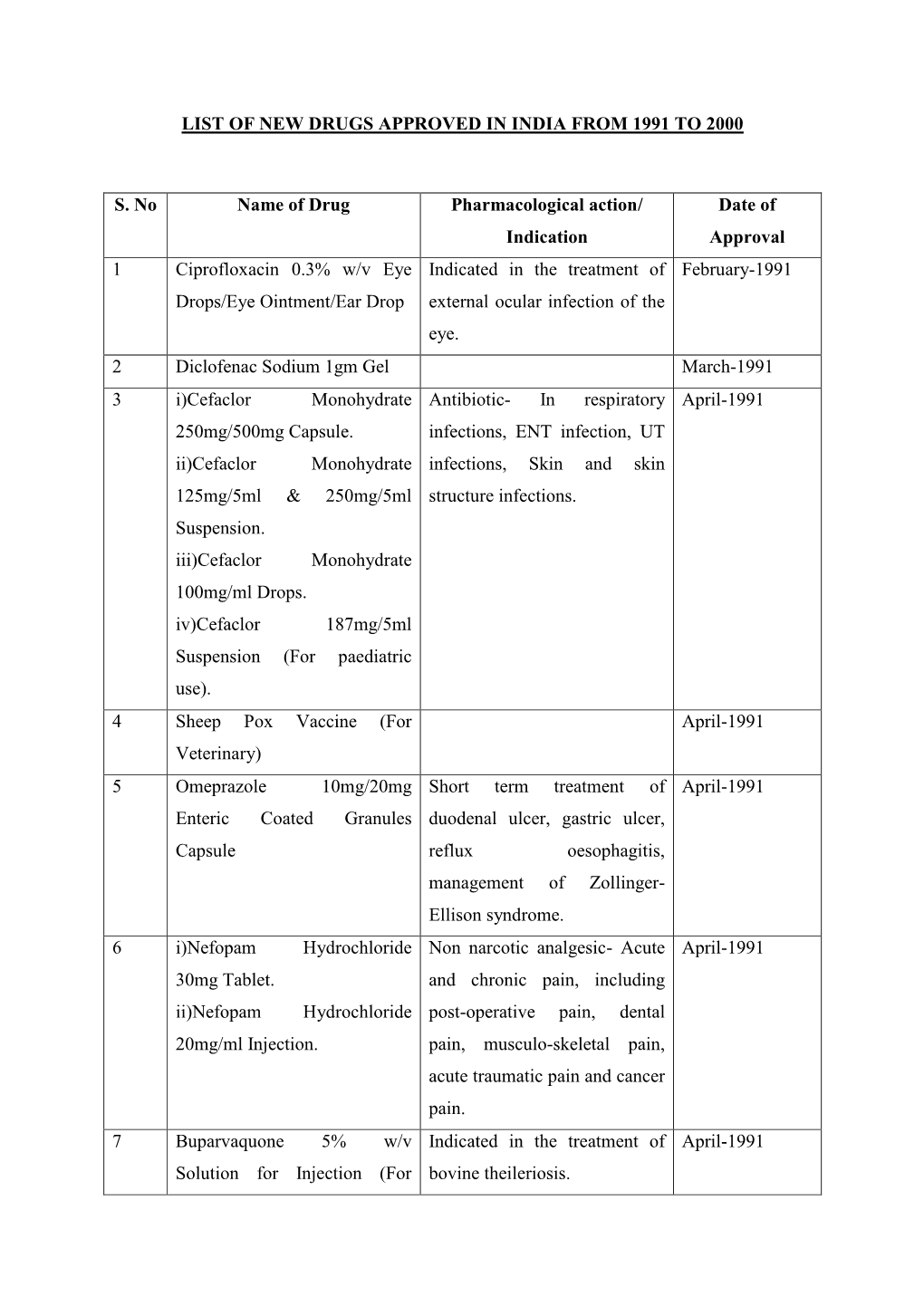
List of New Drugs Approved in India from 1991 to 2000
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Fixed Drug Eruption Due to a Cerumenolytic Ear Drop; Hydrogen Peroxide and Boric Acid: a Case Report
ISSN: 2688-8238 DOI: 10.33552/OJOR.2020.04.000577 Online Journal of Otolaryngology and Rhinology Case Report Copyright © All rights are reserved by Akif İşlek Fixed Drug Eruption Due to A Cerumenolytic Ear Drop; Hydrogen Peroxide and Boric Acid: A Case Report Akif İşlek1* and Engin Karaaslan2 1Nusaybin State Hospital, Otolaryngology-Head & Neck Surgery Clinic, Mardin, Turkey 2Nusaybin State Hospital, Dermatology and Venereology, Mardin, Turkey *Corresponding author: Received Date: October 24, 2020 Published Date: November 03, 2020 Akif İşlek, Nusaybin State Hospital, Otolaryngology-Head & Neck Surgery Clinic, Mardin, Turkey. Abstract Treatment of earwax often involves the use of a wax softening agent (cerumenolytic) with or without antimicrobial agents to easily removing. Cerumenolytic agents used to remove and soften earwax areoil-based treatments, water-based treatments. Fixed drug eruption (FDE) is a well- defined, circular, hyperpigmented plaque that usually occurs on the trunk, hands mucosal surfaces, after a systemical application of drugs. In this report, a case with FDE developed after a cerumenolytic agent (hydrogen peroxide and boric acid in water). A definitive diagnosis was made with skin punch biopsy and FDE was treated with oral levocetirizine and topical mometasone. Introduction is a well-defined, circular, hyper pigmenting plaque that usually Cerumen production is a normal and protective process occurs on the genitals, lips, trunk, and hands and the diagnosis for the external ear canal (EAC), but impact of cerumen causes can be confirmed by histopathologic examination of a small punch symptoms such as hearing loss, itching, pain, tinnitus [1]. Cerumen biopsy specimen [5]. In this report, a case with FDE developed after impaction is defined as an accumulation of cerumen in the a cerumenolytic agent (hydrogen peroxide and boric acid in water) external ear [2]. -

Research Article RP-HPLC Method for Determination of Several Nsaids and Their Combination Drugs
Hindawi Publishing Corporation Chromatography Research International Volume 2013, Article ID 242868, 13 pages http://dx.doi.org/10.1155/2013/242868 Research Article RP-HPLC Method for Determination of Several NSAIDs and Their Combination Drugs Prinesh N. Patel, Gananadhamu Samanthula, Vishalkumar Shrigod, Sudipkumar C. Modh, and Jainishkumar R. Chaudhari Department of Pharmaceutical Analysis, National Institute of Pharmaceutical Education and Research (NIPER), Balanagar, Hyderabad, Andhra Pradesh 500037, India Correspondence should be addressed to Gananadhamu Samanthula; [email protected] Received 29 June 2013; Accepted 13 October 2013 Academic Editor: Andrew Shalliker Copyright © 2013 Prinesh N. Patel et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. An RP-HPLC method for simultaneous determination of 9 NSAIDs (paracetamol, salicylic acid, ibuprofen, naproxen, aceclofenac, diclofenac, ketorolac, etoricoxib, and aspirin) and their commonly prescribed combination drugs (thiocolchicoside, moxifloxacin, clopidogrel, chlorpheniramine maleate, dextromethorphan, and domperidone) was established. The separation was performed on ∘ Kromasil C18 (250 × 4.6 mm, 5 m) at 35 C using 15 mM phosphate buffer pH 3.25 and acetonitrile with gradient elution ata flow rate of 1.1 mL/min. The detection was performed by a diode array detector (DAD) at 230 nm with total run time of 30min. 2 Calibration curves were linear with correlation coefficients of determinationr ( ) > 0.999. Limit of detection (LOD) and Limit of quantification (LOQ) ranged from 0.04 to 0.97 g/mL and from 0.64 to 3.24 g/mL, respectively. -

18512582.Pdf
European Heart Journal (2001) 22, 1938–1947 doi:10.1053/euhj.2001.2627, available online at http://www.idealibrary.com on The TRAPIST Study A multicentre randomized placebo controlled clinical trial of trapidil for prevention of restenosis after coronary stenting, measured by 3-D intravascular ultrasound P. W. Serruys1, D. P. Foley1, M. Pieper2, J. A. Kleijne3 and P. J. de Feyter1 on behalf of the TRAPIST investigators 1Department of Interventional Cardiology, Thoraxcenter, Erasmus Medical Center, Rotterdam, The Netherlands; 2Heartcenter Bodensee, Kreuzlingen, Switzerland; and 3Cardialysis, Rotterdam, The Netherlands Background Studies have reported benefit of oral therapy significant difference between trapidil and placebo-treated with the phosphodiesterase inhibitor, trapidil, in reducing patients regarding in-stent neointimal volume (108·6 restenosis after coronary angioplasty. Coronary stenting is 95·6 mm3 vs 93·379·1 mm3; P=0·16) or % obstruction associated with improved late outcome compared with volume (3818% vs 3621%; P=0·32), in angiographic balloon angioplasty, but significant neointimal hyperplasia minimal luminal diameter at follow-up (1·630·61 mm vs still occurs in a considerable proportion of patients. The 1·740·69 mm; P=0·17), restenosis rate (31% vs 24%; aim of this study was to investigate the safety and efficacy P=0·24), cumulative incidence of major adverse cardiac of trapidil 200 mg in preventing in-stent restenosis. events at 7 months (22% vs 20%; P=0·71) or anginal complaints (30% vs 24%; P=0·29). Methods Patients with a single native coronary lesion requiring revascularization were randomized to placebo or Conclusion Oral trapidil 600 mg daily for 6 months did trapidil at least 1 h before, and continuing for 6 months not reduce in-stent hyperplasia or improve clinical outcome after, successful implantation of a coronary Wallstent. -

Dorset Medicines Advisory Group
DORSET CARDIOLOGY WORKING GROUP GUIDELINE FOR CALCIUM CHANNEL BLOCKERS IN HYPERTENSION SUMMARY The pan-Dorset cardiology working group continues to recommend the use of amlodipine (a third generation dihydropyridine calcium-channel blocker) as first choice calcium channel blocker on the pan-Dorset formulary for hypertension. Lercanidipine is second choice, lacidipine third choice and felodipine is fourth choice. This is due to preferable side effect profiles in terms of ankle oedema and relative costs of the preparations. Note: where angina is the primary indication or is a co-morbidity prescribers must check against the specific product characteristics (SPC) for an individual drug to confirm this is a licensed indication. N.B. Lacidipine and lercandipine are only licensed for use in hypertension. Chapter 02.06.02 CCBs section of the Formulary has undergone an evidence-based review. A comprehensive literature search was carried out on NHS Evidence, Medline, EMBASE, Cochrane Database, and UK Duets. This was for recent reviews or meta-analyses on calcium channel blockers from 2009 onwards (comparative efficacy and side effects) and randomised controlled trials (RCTs). REVIEW BACKGROUND Very little good quality evidence exists. No reviews, meta-analyses or RCTs were found covering all calcium channel blockers currently on the formulary. Another limitation was difficulty obtaining full text original papers for some of the references therefore having to use those from more obscure journals instead. Some discrepancies exist between classification of generations of dihydropyridine CCBs, depending upon the year of publication of the reference/authors’ interpretation. Dihydropyridine (DHP) CCBs tend to be more potent vasodilators than non-dihydropyridine (non-DHP) CCBs (diltiazem, verapamil), but the latter have greater inotropic effects. -

List of Union Reference Dates A
Active substance name (INN) EU DLP BfArM / BAH DLP yearly PSUR 6-month-PSUR yearly PSUR bis DLP (List of Union PSUR Submission Reference Dates and Frequency (List of Union Frequency of Reference Dates and submission of Periodic Frequency of submission of Safety Update Reports, Periodic Safety Update 30 Nov. 2012) Reports, 30 Nov. -

Trapidil Inhibits Human Mesangiai Cell Proliferation: Effect on PDGF Β
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Kidney International, Vol. 46 (1994), pp. 1002—1009 Trapidil inhibits human mesangial cell proliferation: Effect on PDGF a-receptor binding and expression LORETO GESUALDO, SALVATORE Di PAOLO, ELENA RANIERI, and FRANCESCO PAOLO SCHENA, with the technical assistance of ANNALISA BRUNACCINI Chair and Division of Nephrology, University of Bad, Ban, Italy Trapidil inhibits human mesangial cell proliferation: Effect on PDCF mediate, at least in part, the biological effect of other growth a-receptor binding and expression. Mesangial cell (MC) proliferation, a factors, as demonstrated for EGF, and elects PDGF as a potential histopathologic feature common to many human glomerular diseases, is regulated by several growth factors through their binding to specific cell autocrine regulator of MC proliferation [14]. surface receptors. Platelet-derived growth factor (PDGF) is a peptide MC play a pivotal role in the course of glomerular injury: they exerting a potent mitogenic activity on MC. Recently, an increasedfirst act as target cells for several mediators, which are released expression of both PDGF protein and its receptor has been localized in locally or are delivered through the systemic circulation [4, 15, 16]. the mesangial areas of several experimental as well as human proliferative Thereafter, they may drive the evolution of pathologic lesions glomerulonephritides (GN). Thus, it may be postulated that the inhibition of PDGF action could prevent MC proliferation during mesangial prolif- towards resolution or progression of the glomerular disease. MC erative GN. Trapidil, an antiplatelet drug, has been shown to inhibit the proliferation, a histopatologic feature common to different forms growth of several cell types both in vitro and in vivo. -

)&F1y3x PHARMACEUTICAL APPENDIX to THE
)&f1y3X PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE )&f1y3X PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 3 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. Product CAS No. Product CAS No. ABAMECTIN 65195-55-3 ACTODIGIN 36983-69-4 ABANOQUIL 90402-40-7 ADAFENOXATE 82168-26-1 ABCIXIMAB 143653-53-6 ADAMEXINE 54785-02-3 ABECARNIL 111841-85-1 ADAPALENE 106685-40-9 ABITESARTAN 137882-98-5 ADAPROLOL 101479-70-3 ABLUKAST 96566-25-5 ADATANSERIN 127266-56-2 ABUNIDAZOLE 91017-58-2 ADEFOVIR 106941-25-7 ACADESINE 2627-69-2 ADELMIDROL 1675-66-7 ACAMPROSATE 77337-76-9 ADEMETIONINE 17176-17-9 ACAPRAZINE 55485-20-6 ADENOSINE PHOSPHATE 61-19-8 ACARBOSE 56180-94-0 ADIBENDAN 100510-33-6 ACEBROCHOL 514-50-1 ADICILLIN 525-94-0 ACEBURIC ACID 26976-72-7 ADIMOLOL 78459-19-5 ACEBUTOLOL 37517-30-9 ADINAZOLAM 37115-32-5 ACECAINIDE 32795-44-1 ADIPHENINE 64-95-9 ACECARBROMAL 77-66-7 ADIPIODONE 606-17-7 ACECLIDINE 827-61-2 ADITEREN 56066-19-4 ACECLOFENAC 89796-99-6 ADITOPRIM 56066-63-8 ACEDAPSONE 77-46-3 ADOSOPINE 88124-26-9 ACEDIASULFONE SODIUM 127-60-6 ADOZELESIN 110314-48-2 ACEDOBEN 556-08-1 ADRAFINIL 63547-13-7 ACEFLURANOL 80595-73-9 ADRENALONE -

Ijcep0048253.Pdf
Int J Clin Exp Pathol 2017;10(4):4089-4098 www.ijcep.com /ISSN:1936-2625/IJCEP0048253 Original Article Comparative effectiveness of different recommended doses of omeprazole and lansoprazole for gastroesophageal reflux disease: a meta-analysis of published data Feng Liu1, Jing Wang1, Hailong Wu1, Hui Wang2, Jianxiang Wang1, Rui Zhou1, Zhi Zhu3 Departments of 1Surgery, 2Gastroenterology, 3Epidemiology and Biostatistics, Puai Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China Received January 6, 2017; Accepted February 20, 2017; Epub April 1, 2017; Published April 15, 2017 Abstract: This meta-analysis aims to evaluate the effectiveness of different recommended doses of omeprazole and lansoprazole on gastroesophageal reflux diseases (GERD) in adults. The electronic databases of PubMed, EMBASE, Cochrane Library, and ClinicalTrials.gov were searched before September 13 2016. Fifteen eligible studies were identified involving 8752 patients in our meta-analysis. For the healing outcome of esophagitis, compared with 15 mg per day of lansoprazole, there were significant difference of both 30 mg per day of lansoprazole (RR=1.29, 95% CI [1.01, 1.66], I2=79.3%, P=0.028) and 60 mg per day of lansoprazole (RR=1.59, 95% CI [1.28, 1.99], I2=Not ap- plicable (NA), P=NA), and the other result were not significantly different between 60 mg per day and 30 mg per day. For relief of symptoms, our result indicated a significant difference between 20 mg per day and 10 mg per day of omeprazole (RR=1.21, 95% CI [1.06, 1.39], I2=53.9%, P=0.089); the overall result indicated a significant difference between lansoprazole and omeprazole (RR=0.93, 95% CI [0.86, 0.999], I2=60.7%, P=0.038). -

Protonix Protonix
NDA 22-020 Page 4 PROTONIX® (pantoprazole sodium) Delayed-Release Tablets PROTONIX® (pantoprazole sodium) For Delayed-Release Oral Suspension Rx only DESCRIPTION ® The active ingredient in PROTONIX (pantoprazole sodium) Delayed-Release Tablets and ® PROTONIX (pantoprazole sodium) For Delayed-Release Oral Suspension is a substituted benzimidazole, sodium 5-(difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridinyl)methyl] sulfinyl]- 1H-benzimidazole sesquihydrate, a compound that inhibits gastric acid secretion. Its empirical formula is C16H14F2N3NaO4S x 1.5 H2O, with a molecular weight of 432.4. The structural formula is: Pantoprazole sodium sesquihydrate is a white to off-white crystalline powder and is racemic. Pantoprazole has weakly basic and acidic properties. Pantoprazole sodium sesquihydrate is freely soluble in water, very slightly soluble in phosphate buffer at pH 7.4, and practically insoluble in n-hexane. The stability of the compound in aqueous solution is pH-dependent. The rate of degradation increases with decreasing pH. At ambient temperature, the degradation half-life is approximately 2.8 hours at pH 5.0 and approximately 220 hours at pH 7.8. PROTONIX (pantoprazole sodium) is supplied as a delayed-release tablet for oral administration, available in 2 strengths (40 mg and 20 mg); and as delayed-release granules for oral administration, available in the 40 mg strength. NDA 22-020 Page 5 Each PROTONIX (pantoprazole sodium) Delayed-Release tablet contains 45.1 mg or 22.6 mg of pantoprazole sodium sesquihydrate (equivalent to 40 mg or 20 mg pantoprazole, respectively) with the following inactive ingredients: calcium stearate, crospovidone, hypromellose, iron oxide, mannitol, methacrylic acid copolymer, polysorbate 80, povidone, propylene glycol, sodium carbonate, sodium lauryl sulfate, titanium dioxide, and triethyl citrate. -

Proton Pump Inhibitors (PPI) Preferred Step Therapy Program Annual Review Date: 01/18/2021
Policy: Proton Pump Inhibitors (PPI) Preferred Step Therapy Program Annual Review Date: 01/18/2021 Impacted Last Revised Date: Drugs: • Aciphex, Aciphex Sprinkle • Dexilant 06/17/2021 • Esomeprazole DR, esomeprazole strontium DR • Nexium • Omeprazole/sodium bicarbonate • Prevacid, Prevacid 24HR, Prevacid SoluTab • Prilosec • Protonix • Rabeprazole DR OVERVIEW Proton pump inhibitors (PPIs) [i.e., esomeprazole, dexlansoprazole, lansoprazole, omeprazole, pantoprazole, and rabeprazole] are commonly used antisecretory agents that are highly effective at suppressing gastric acid and subsequently treating associated conditions, including gastroesophageal reflux disease (GERD). Omeprazole is available generically and over-the-counter (OTC). Omeprazole OTC is prescription strength (20 mg). Lansoprazole is also available generically and OTC. Lansoprazole OTC is available as 15 mg capsules. Zegerid capsules are available generically and OTC; the OTC product contains omeprazole 20 mg and sodium bicarbonate 1100 mg. Nexium® 24HR (esomeprazole magnesium 22.3 mg delayed-release capsules) is available OTC and is equivalent to 20 mg of esomeprazole base. The OTC products are indicated for the short-term (14 days) treatment of heartburn. Patients should not take the OTC products for more than 14 days or more often than every 4 months unless under the supervision of a physician. Although the PPIs vary in their specific Food and Drug Administration (FDA)-approved indications, all of the PPIs have demonstrated the ability to control GERD symptoms and to heal esophagitis when used at prescription doses. Most comparative studies between PPIs to date have demonstrated comparable efficacies for acid-related diseases, including duodenal and gastric ulcerations, GERD, Zollinger-Ellison syndrome, and H. pylori eradication therapies. Though the available clinical data are not entirely complete for the comparison of these agents, many clinical trials have shown the PPIs to be similar in safety and efficacy. -

Tall Man Lettering List REPORT DECEMBER 2013 1
Tall Man Lettering List REPORT DECEMBER 2013 1 TALL MAN LETTERING LIST REPORT WWW.HQSC.GOVT.NZ Published in December 2013 by the Health Quality & Safety Commission. This document is available on the Health Quality & Safety Commission website, www.hqsc.govt.nz ISBN: 978-0-478-38555-7 (online) Citation: Health Quality & Safety Commission. 2013. Tall Man Lettering List Report. Wellington: Health Quality & Safety Commission. Crown copyright ©. This copyright work is licensed under the Creative Commons Attribution-No Derivative Works 3.0 New Zealand licence. In essence, you are free to copy and distribute the work (including other media and formats), as long as you attribute the work to the Health Quality & Safety Commission. The work must not be adapted and other licence terms must be abided. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nd/3.0/nz/ Copyright enquiries If you are in doubt as to whether a proposed use is covered by this licence, please contact: National Medication Safety Programme Team Health Quality & Safety Commission PO Box 25496 Wellington 6146 ACKNOWLEDGEMENTS The Health Quality & Safety Commission acknowledges the following for their assistance in producing the New Zealand Tall Man lettering list: • The Australian Commission on Safety and Quality in Health Care for advice and support in allowing its original work to be either reproduced in whole or altered in part for New Zealand as per its copyright1 • The Medication Safety and Quality Program of Clinical Excellence Commission, New South -

Proton Pump Inhibitors
MEDICATION POLICY: Proton Pump Inhibitors Generic Name: Proton Pump Inhibitors Preferred: Esomeprazole (generic), Lansoprazole (generic), Omeprazole (generic), Therapeutic Class or Brand Name: Proton Pantoprazole (generic), and Rabeprazole Pump Inhibitors (generic) Applicable Drugs (if Therapeutic Class): Non-preferred: Aciphex® (rabeprazole), Esomeprazole (generic), Lansoprazole Dexilant® (dexlansoprazole), Nexium® (generic), Omeprazole (generic), Pantoprazole (esomeprazole), Not Medically Necessary: (generic), and Rabeprazole (generic). Omeprazole/Sodium Bicarbonate (generic), Aciphex® (rabeprazole), Dexilant® Zegerid® (omeprazole/sodium bicarbonate). (dexlansoprazole), Nexium® (esomeprazole), Prevacid® (lansoprazole), Prilosec® Date of Origin: 2/1/2013 (omeprazole), and Protonix® (pantoprazole). Date Last Reviewed / Revised: 12/23/2020 Omeprazole/Sodium Bicarbonate (generic), Zegerid® (omeprazole/sodium bicarbonate). Policy also applies to any other Proton Pump Inhibitors not listed. GPI Code: 4927002000, 4927002510, 4927004000, 4927006000, 4927007010, 4927007610, 4999600260 PRIOR AUTHORIZATION CRITERIA (May be considered medically necessary when criteria I and II are met) I. Documented diagnosis of one of the following A through G: A. Gastroesophageal reflux disease (GERD). B. Erosive esophagitis. C. Gastric ulcers. D. Risk reduction of NSAID-associated gastric ulcer. E. Duodenal ulcers. F. Eradication of H. pylori. G. Hypersecretory conditions II. Non-preferred PPIs require documented trials and failures of all generic PPIs. EXCLUSION