Additional Restrictions for Select Ppis and Auto-Substitutions for Most
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
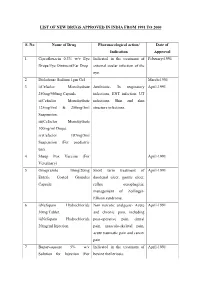
List of New Drugs Approved in India from 1991 to 2000
LIST OF NEW DRUGS APPROVED IN INDIA FROM 1991 TO 2000 S. No Name of Drug Pharmacological action/ Date of Indication Approval 1 Ciprofloxacin 0.3% w/v Eye Indicated in the treatment of February-1991 Drops/Eye Ointment/Ear Drop external ocular infection of the eye. 2 Diclofenac Sodium 1gm Gel March-1991 3 i)Cefaclor Monohydrate Antibiotic- In respiratory April-1991 250mg/500mg Capsule. infections, ENT infection, UT ii)Cefaclor Monohydrate infections, Skin and skin 125mg/5ml & 250mg/5ml structure infections. Suspension. iii)Cefaclor Monohydrate 100mg/ml Drops. iv)Cefaclor 187mg/5ml Suspension (For paediatric use). 4 Sheep Pox Vaccine (For April-1991 Veterinary) 5 Omeprazole 10mg/20mg Short term treatment of April-1991 Enteric Coated Granules duodenal ulcer, gastric ulcer, Capsule reflux oesophagitis, management of Zollinger- Ellison syndrome. 6 i)Nefopam Hydrochloride Non narcotic analgesic- Acute April-1991 30mg Tablet. and chronic pain, including ii)Nefopam Hydrochloride post-operative pain, dental 20mg/ml Injection. pain, musculo-skeletal pain, acute traumatic pain and cancer pain. 7 Buparvaquone 5% w/v Indicated in the treatment of April-1991 Solution for Injection (For bovine theileriosis. Veterinary) 8 i)Kitotifen Fumerate 1mg Anti asthmatic drug- Indicated May-1991 Tablet in prophylactic treatment of ii)Kitotifen Fumerate Syrup bronchial asthma, symptomatic iii)Ketotifen Fumerate Nasal improvement of allergic Drops conditions including rhinitis and conjunctivitis. 9 i)Pefloxacin Mesylate Antibacterial- In the treatment May-1991 Dihydrate 400mg Film Coated of severe infection in adults Tablet caused by sensitive ii)Pefloxacin Mesylate microorganism (gram -ve Dihydrate 400mg/5ml Injection pathogens and staphylococci). iii)Pefloxacin Mesylate Dihydrate 400mg I.V Bottles of 100ml/200ml 10 Ofloxacin 100mg/50ml & Indicated in RTI, UTI, May-1991 200mg/100ml vial Infusion gynaecological infection, skin/soft lesion infection. -

Appendix a Common Abbreviations Used in Medication
UNIVERSITY OF AMSTERDAM MASTERS THESIS Impact of Medication Grouping on Fall Risk Prediction in Elders: A Retrospective Analysis of MIMIC-III Critical Care Database Student: SRP Mentor: Noman Dormosh Dr. Martijn C. Schut Student No. 11412682 – SRP Tutor: Prof. dr. Ameen Abu-Hanna SRP Address: Amsterdam University Medical Center - Location AMC Department Medical Informatics Meibergdreef 9, 1105 AZ Amsterdam Practice teaching period: November 2018 - June 2019 A thesis submitted in fulfillment of the requirements for the degree of Master of Medical Informatics iii Abstract Background: Falls are the leading cause of injury in elderly patients. Risk factors for falls in- cluding among others history of falls, old age, and female gender. Research studies have also linked certain medications with an increased risk of fall in what is called fall-risk-increasing drugs (FRIDs), such as psychotropics and cardiovascular drugs. However, there is a lack of consistency in the definitions of FRIDs between the studies and many studies did not use any systematic classification for medications. Objective: The aim of this study was to investigate the effect of grouping medications at different levels of granularity of a medication classification system on the performance of fall risk prediction models. Methods: This is a retrospective analysis of the MIMIC-III cohort database. We created seven prediction models including demographic, comorbidity and medication variables. Medica- tions were grouped using the anatomical therapeutic chemical classification system (ATC) starting from the most specific scope of medications and moving up to the more generic groups: one model used individual medications (ATC level 5), four models used medication grouping at levels one, two, three and four of the ATC and one model did not include med- ications. -

Protonix Protonix
NDA 22-020 Page 4 PROTONIX® (pantoprazole sodium) Delayed-Release Tablets PROTONIX® (pantoprazole sodium) For Delayed-Release Oral Suspension Rx only DESCRIPTION ® The active ingredient in PROTONIX (pantoprazole sodium) Delayed-Release Tablets and ® PROTONIX (pantoprazole sodium) For Delayed-Release Oral Suspension is a substituted benzimidazole, sodium 5-(difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridinyl)methyl] sulfinyl]- 1H-benzimidazole sesquihydrate, a compound that inhibits gastric acid secretion. Its empirical formula is C16H14F2N3NaO4S x 1.5 H2O, with a molecular weight of 432.4. The structural formula is: Pantoprazole sodium sesquihydrate is a white to off-white crystalline powder and is racemic. Pantoprazole has weakly basic and acidic properties. Pantoprazole sodium sesquihydrate is freely soluble in water, very slightly soluble in phosphate buffer at pH 7.4, and practically insoluble in n-hexane. The stability of the compound in aqueous solution is pH-dependent. The rate of degradation increases with decreasing pH. At ambient temperature, the degradation half-life is approximately 2.8 hours at pH 5.0 and approximately 220 hours at pH 7.8. PROTONIX (pantoprazole sodium) is supplied as a delayed-release tablet for oral administration, available in 2 strengths (40 mg and 20 mg); and as delayed-release granules for oral administration, available in the 40 mg strength. NDA 22-020 Page 5 Each PROTONIX (pantoprazole sodium) Delayed-Release tablet contains 45.1 mg or 22.6 mg of pantoprazole sodium sesquihydrate (equivalent to 40 mg or 20 mg pantoprazole, respectively) with the following inactive ingredients: calcium stearate, crospovidone, hypromellose, iron oxide, mannitol, methacrylic acid copolymer, polysorbate 80, povidone, propylene glycol, sodium carbonate, sodium lauryl sulfate, titanium dioxide, and triethyl citrate. -

Clinical Indications for Proton Pump Inhibitors and Tapering Information When Indicated
Clinical Indications for Proton Pump Inhibitors and Tapering Information When Indicated MacKenzie Crist, Pharm.D. Candidate 2017 Nabila Ahmed -Sarwar, Pharm.D, BCPS, CDE October 2016 Appropriate indications for PPIs Treatment Recommendations for GERD ● Initial eight-week course of therapy Try to lower dose, as needed therapy, or Short-Term Treatment Long-Term Treatment intermittent therapy ● ○ ● GERD ● Refractory GERD Refractory GERD—No response to PPI therapy after two to ● Gastric and duodenal ● Erosive esophagitis three months (needs GI consult and endoscopy) ulcers ● Barrett’s esophagus Add a bedtime H2 blocker if nocturnal symptoms ● H. pylori co-therapy ● History of NSAID induced Double the dose ○ bleeding ulcers Concerns with Chronic PPI Therapy ○ ● Chronic anticoagulation ● Clostridium difficile infections after a GI bleed ● Hypomagnesaemia ● NSAID or dual anti-platelet ● Increased fracture risk agent use* ● Acute interstitial nephritis (rare, idiopathic) ● Chronic kidney disease (weak observational data may be *presence of multiple risk factors: Age>65 years, high dose NSAID associated with dose and duration) therapy, PUD, concurrent corticosteroids or anticoagulants When PPIs Should Be Tapered and Not Abruptly Discontinued ● Patients who do not have any appropriate indications for continuing a PPI, but have been on a PPI for > six months, and asymptomatic at least 3 months ● Counsel patient on initial worsening of symptoms during tapering phase PPI Tapering Algorithm Step 1: Tapering Schedule Step 2: Management of Recurrence H2 Antagonist and antacids may be added while tapering for breakthrough symptoms Katz PO, Gerson LB, Vela MF. Guidelines for the diagnosis and management of gastroesophageal reflux disease. Am J Gastroenterology. 2013; 108:308-328. Bundeff AW, Zaiken K. -

PHARMACEUTICAL APPENDIX to the TARIFF SCHEDULE 2 Table 1
Harmonized Tariff Schedule of the United States (2020) Revision 19 Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE Harmonized Tariff Schedule of the United States (2020) Revision 19 Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 2 Table 1. This table enumerates products described by International Non-proprietary Names INN which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service CAS registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. -

Proton Pump Inhibitors
MEDICATION POLICY: Proton Pump Inhibitors Generic Name: Proton Pump Inhibitors Preferred: Esomeprazole (generic), Lansoprazole (generic), Omeprazole (generic), Therapeutic Class or Brand Name: Proton Pantoprazole (generic), and Rabeprazole Pump Inhibitors (generic) Applicable Drugs (if Therapeutic Class): Non-preferred: Aciphex® (rabeprazole), Esomeprazole (generic), Lansoprazole Dexilant® (dexlansoprazole), Nexium® (generic), Omeprazole (generic), Pantoprazole (esomeprazole), Not Medically Necessary: (generic), and Rabeprazole (generic). Omeprazole/Sodium Bicarbonate (generic), Aciphex® (rabeprazole), Dexilant® Zegerid® (omeprazole/sodium bicarbonate). (dexlansoprazole), Nexium® (esomeprazole), Prevacid® (lansoprazole), Prilosec® Date of Origin: 2/1/2013 (omeprazole), and Protonix® (pantoprazole). Date Last Reviewed / Revised: 12/23/2020 Omeprazole/Sodium Bicarbonate (generic), Zegerid® (omeprazole/sodium bicarbonate). Policy also applies to any other Proton Pump Inhibitors not listed. GPI Code: 4927002000, 4927002510, 4927004000, 4927006000, 4927007010, 4927007610, 4999600260 PRIOR AUTHORIZATION CRITERIA (May be considered medically necessary when criteria I and II are met) I. Documented diagnosis of one of the following A through G: A. Gastroesophageal reflux disease (GERD). B. Erosive esophagitis. C. Gastric ulcers. D. Risk reduction of NSAID-associated gastric ulcer. E. Duodenal ulcers. F. Eradication of H. pylori. G. Hypersecretory conditions II. Non-preferred PPIs require documented trials and failures of all generic PPIs. EXCLUSION -

2018 Annual Results Presentation
2018 Annual Results Presentation Sihuan Pharmaceutical Holdings Group Ltd. 四环医药控股集团有限公司 0 Disclaimer The sole purpose of this Presentation (the “Presentation”) is to assist the recipient in deciding whether it wishes to proceed with a further investigation of Sihuan Pharmaceutical Holdings Group Ltd. (the “Company”) and it is not intended to form the basis of any decision to purchase securities, interests or assets in or of the Company. This Presentation does not constitute or contain an offer or invitation or recommendation or solicitation for the sale or purchase of securities, interests or assets in or of the Company and neither this document nor anything contained herein shall form the basis of, or be relied upon in connection with, any contract or commitment whatsoever. Any decision to purchase or subscribe for securities in any offering must be made solely on the basis of the information contained in the prospectus or offering circular issued by the company in connection with such offerings. All the information in this Presentation has been provided by the Company and has not been independently verified. No representation or warranty, express or implied, is or will be made in or in relation to, and no responsibility or liability is or will be accepted by the Company or any of its subsidiaries as to the appropriateness, accuracy, completeness or reliability of, this Presentation or any other written or oral information made available to any interested party or its advisers and any liability therefore is hereby expressly disclaimed. And no reliance should be placed on the accuracy, fairness, completeness or correctness of the information contained in this Presentation. -

PHRP March 2015
March 2015; Vol. 25(2):e2521518 doi: http://dx.doi.org/10.17061/phrp2521518 www.phrp.com.au Research Manual versus automated coding of free-text self-reported medication data in the 45 and Up Study: a validation study Danijela Gnjidica,b,i, Sallie-Anne Pearsona,c, Sarah N Hilmerb,d, Jim Basilakise, Andrea L Schaffera, Fiona M Blythb,f,g and Emily Banksg,h, on behalf of the High Risk Prescribing Investigators a Faculty of Pharmacy, University of Sydney, NSW, Australia b Sydney Medical School, University of Sydney, NSW, Australia c Sydney School of Public Health, University of Sydney, NSW, Australia d Royal North Shore Hospital and Kolling Institute of Medical Research, Sydney, NSW, Australia e School of Computing, Engineering and Mathematics, University of Western Sydney, NSW, Australia f Centre for Education and Research on Ageing (CERA), Concord Hospital, Sydney, NSW, Australia g The Sax Institute, Sydney, NSW, Australia h National Centre for Epidemiology and Population Health, Australian National University, Canberra, ACT i Corresponding author: [email protected] Article history Abstract Publication date: March 2015 Background: Increasingly, automated methods are being used to code free- Citation: Gnjidic D, Pearson S, Hilmer S, text medication data, but evidence on the validity of these methods is limited. Basilakis J, Schaffer AL, Blyth FM, Banks E. To examine the accuracy of automated coding of previously keyed Manual versus automated coding of free-text Aim: in free-text medication data compared with manual coding of original self-reported medication data in the 45 and Up Study: a validation study. Public Health handwritten free-text responses (the ‘gold standard’). -

Acid-Reducing Medications: Interactions with Harvoni and Epclusa
November 2016 | www.hepatitis.va.gov Acid-Reducing Medications: Interactions with Harvoni® and Epclusa® Sofosbuvir/Velpatasvir = Epclusa® Ledipasvir/Sofosbuvir = Harvoni® • Acid-reducing medications can decrease the effectiveness of Harvoni® and Epclusa® and could prevent successful cure. • Let your provider know if you are taking any acid-reducing medications—either prescribed to you or purchased over the counter. Types of Acid-Reducing What to Do if What to Do if Medications Taking Epclusa® Taking Harvoni® Antacids Separate taking the antacid and Separate taking the antacid and Epclusa® by 4 hours Harvoni® by 4 hours Combinations of aluminum, magnesium, and calcium. Brand names include: Alka-Seltzer® Maalox® Mylanta® Rolaids® TUMS® H2 Blockers Do not take more than the Do not take more than the recommended doses recommended doses Cimetidine (Tagamet®) Famotidine (Pepcid®) May take together with Epclusa® May take together with Harvoni® Nizatidine (Axid®) or 12 hours apart or 12 hours apart Ranitidine (Zantac®) Proton Pump Inhibitors (PPI) It is best to avoid PPIs during treat- It is best to avoid PPIs during ment with Epclusa®. If there are no treatment with Harvoni®. If there Esomeprazole (Nexium®) alternatives, follow these instructions: are no alternatives, follow these Lansoprazole (Prevacid®) instructions: Omeprazole (Prilosec®) Take Epclusa® with food Pantoprazole (Protonix®) Take the proton pump inhibitor 4 Take Harvoni® and the proton Rabeprazole (Aciphex®) hours after taking Epclusa® pump inhibitor at the same time in the morning after fasting overnight Do not take the proton pump (on an empty stomach) inhibitor at the same time as Epclusa® Do not take proton pump Do not take proton pump inhibitors inhibitors at more than the at more than the recommended dose recommended dose U.S. -

Risks of Proton Pump Inhibitors for Gastroesophageal Reflux Disease and a Diet Alternative
Journal of Nutritional Health & Food Engineering Literature Review Open Access Risks of proton pump inhibitors for gastroesophageal reflux disease and a diet alternative Abstract Volume 10 Issue 1 - 2020 Gastroesophageal reflux disease (GERD) is one of the most common digestive conditions Mark Stengler NMD treated by gastroenterologists. The most commonly prescribed drugs for GERD are proton Stengler Center for Integrative Medicine, USA pump inhibitors (PPIs) that reduce stomach acid. While these medications are effective for relieving GERD symptoms, their long-term use is associated with several side effects Correspondence: Mark Stengler NMD, Stengler Center for and chronic diseases. Several publications have questioned the long-term safety of PPIs. Integrative Medicine, 324 Encinitas Blvd. Moreover, recent studies have demonstrated that the Mediterranean Diet may be more Encinitas, CA 92024 1-760-274-2377, effective than PPIs for people suffering from GERD. Email Keywords: GERD, mediterranean diet, proton pump inhibitors, PPI, acid reflux, LES, Received: April 14, 2020 | Published: April 28, 2020 lower esophageal sphincter, omeprazole, prilosec Introduction Proton pump inhibitors to treat GERD Gastroesophageal reflux disease (GERD) is one of the most common The most commonly prescribed drugs for GERD are proton digestive conditions treated by gastroenterologists and primary care pump inhibitors (PPIs).2 PPIs currently available on prescription in doctors. Typical symptoms include heartburn, regurgitation, and the USA are: dexlansoprazole -

Proton-Pump Inhibitor (PPI) Use
Proton-Pump Inhibitor (PPI) Use The FDA has issued multiple warnings on the long-term use of PPIs. These include: increased risk of C. difficile infection1, hypomagnesemia2, and fractures of the hip, wrist, and spine3. Therefore, prudent prescribing of PPIs is warranted. The FDA recommends use of the lowest dose and shortest duration of PPI therapy appropriate for the condition being treated1-3. Patient compliance, time of administration (prior to meals), and dietary indiscretions (i.e. alcohol or irritating foods) should be assessed prior to titration of PPI doses. Indication Treatment Duration Gastroesophageal reflux disease Initial 8 week course for symptom (GERD)6 relief or esophagitis Symptomatic relief Maintenance therapy determined Omeprazole 20 mg PO by response and severity of Acute healing of erosive or ulcerative once daily disease esophagitis OR Pantoprazole 40mg PO For patients that require more Maintenance healing of erosive or once daily long-term therapy, consider a ulcerative esophagitis trial of a lower dose, on-demand therapy, or intermittent therapy to minimize exposure Stress ulcer prophylaxis Transition to PO when possible Reserve PPIs for critically ill patients Continue until resolution of with increased risk of bleeding:4,5 underlying risk factors and/or At least one of the following: critical illness - Coagulopathy (platelet count <50,000 mm3, INR >1.5, or aPTT Recommend discontinuation at >2x control) discharge, unless there is - Mechanical ventilation >48 hours Omeprazole another indication for use - History -

Proton Pump Inhibitors Therapeutic Class Review (TCR) September 1, 2017 the Literature Review Is Current Through February 17, 2020
Proton Pump Inhibitors Therapeutic Class Review (TCR) September 1, 2017 The literature review is current through February 17, 2020. No part of this publication may be reproduced or transmitted in any form or by any means, electronic or mechanical, including photocopying, recording, digital scanning, or via any information storage or retrieval system without the express written consent of Magellan Rx Management. All requests for permission should be mailed to: Magellan Rx Management Attention: Legal Department 6950 Columbia Gateway Drive Columbia, Maryland 21046 The materials contained herein represent the opinions of the collective authors and editors and should not be construed to be the official representation of any professional organization or group, any state Pharmacy and Therapeutics committee, any state Medicaid Agency, or any other clinical committee. This material is not intended to be relied upon as medical advice for specific medical cases and nothing contained herein should be relied upon by any patient, medical professional or layperson seeking information about a specific course of treatment for a specific medical condition. All readers of this material are responsible for independently obtaining medical advice and guidance from their own physician and/or other medical professional in regard to the best course of treatment for their specific medical condition. This publication, inclusive of all forms contained herein, is intended to be educational in nature and is intended to be used for informational purposes only. Send comments and suggestions to [email protected]. September 2017 Proprietary Information. Restricted Access – Do not disseminate or copy without approval. © 2004-2020 Magellan Rx Management. All rights reserved.