Type of Pipette Smallest Division A
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Laboratory Glassware N Edition No
Laboratory Glassware n Edition No. 2 n Index Introduction 3 Ground joint glassware 13 Volumetric glassware 53 General laboratory glassware 65 Alphabetical index 76 Índice alfabético 77 Index Reference index 78 [email protected] Scharlau has been in the scientific glassware business for over 15 years Until now Scharlab S.L. had limited its sales to the Spanish market. However, now, coinciding with the inauguration of the new workshop next to our warehouse in Sentmenat, we are ready to export our scientific glassware to other countries. Standard and made to order Products for which there is regular demand are produced in larger Scharlau glassware quantities and then stocked for almost immediate supply. Other products are either manufactured directly from glass tubing or are constructed from a number of semi-finished products. Quality Even today, scientific glassblowing remains a highly skilled hand craft and the quality of glassware depends on the skill of each blower. Careful selection of the raw glass ensures that our final products are free from imperfections such as air lines, scratches and stones. You will be able to judge for yourself the workmanship of our glassware products. Safety All our glassware is annealed and made stress free to avoid breakage. Fax: +34 93 715 67 25 Scharlab The Lab Sourcing Group 3 www.scharlab.com Glassware Scharlau glassware is made from borosilicate glass that meets the specifications of the following standards: BS ISO 3585, DIN 12217 Type 3.3 Borosilicate glass ASTM E-438 Type 1 Class A Borosilicate glass US Pharmacopoeia Type 1 Borosilicate glass European Pharmacopoeia Type 1 Glass The typical chemical composition of our borosilicate glass is as follows: O Si 2 81% B2O3 13% Na2O 4% Al2O3 2% Glass is an inorganic substance that on cooling becomes rigid without crystallising and therefore it has no melting point as such. -
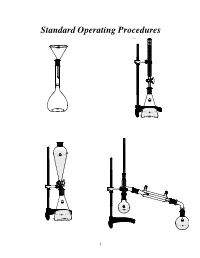
Standard Operating Procedures
Standard Operating Procedures 1 Standard Operating Procedures OVERVIEW In the following laboratory exercises you will be introduced to some of the glassware and tech- niques used by chemists to isolate components from natural or synthetic mixtures and to purify the individual compounds and characterize them by determining some of their physical proper- ties. While working collaboratively with your group members you will become acquainted with: a) Volumetric glassware b) Liquid-liquid extraction apparatus c) Distillation apparatus OBJECTIVES After finishing these sessions and reporting your results to your mentor, you should be able to: • Prepare solutions of exact concentrations • Separate liquid-liquid mixtures • Purify compounds by recrystallization • Separate mixtures by simple and fractional distillation 2 EXPERIMENT 1 Glassware Calibration, Primary and Secondary Standards, and Manual Titrations PART 1. Volumetric Glassware Calibration Volumetric glassware is used to either contain or deliver liquids at a specified temperature. Glassware manufacturers indicate this by inscribing on the volumetric ware the initials TC (to contain) or TD (to deliver) along with the calibration temperature, which is usually 20°C1. Volumetric glassware must be scrupulously clean before use. The presence of streaks or droplets is an indication of the presence of a grease film. To eliminate grease from glassware, scrub with detergent solution, rinse with tap water, and finally rinse with a small portion of distilled water. Volumetric flasks (TC) A volumetric flask has a large round bottom with only one graduation mark positioned on the long narrow neck. Graduation Mark Stopper The position of the mark facilitates the accurate and precise reading of the meniscus. If the flask is used to prepare a solution starting with a solid compound, add small amounts of sol- vent until the entire solid dissolves. -

Environmental Protection Agency Pt. 63, App. A
Environmental Protection Agency Pt. 63, App. A APPENDIX A TO PART 63—TEST METHODS posed as an alternative test method to meet an applicable requirement or in the absence METHOD 301—FIELD VALIDATION OF POLLUT- of a validated method. Additionally, the val- ANT MEASUREMENT METHODS FROM VARIOUS idation procedures of Method 301 are appro- WASTE MEDIA priate for demonstration of the suitability of alternative test methods under 40 CFR parts USING METHOD 301 59, 60, and 61. If, under 40 CFR part 63 or 60, 1.0 What is the purpose of Method 301? you choose to propose a validation method other than Method 301, you must submit and 2.0 What approval must I have to use Method obtain the Administrator’s approval for the 301? candidate validation method. 3.0 What does Method 301 include? 2.0 What approval must I have to use Method 301? 4.0 How do I perform Method 301? If you want to use a candidate test method REFERENCE MATERIALS to meet requirements in a subpart of 40 CFR part 59, 60, 61, 63, or 65, you must also request 5.0 What reference materials must I use? approval to use the candidate test method according to the procedures in Section 16 of SAMPLING PROCEDURES this method and the appropriate section of 6.0 What sampling procedures must I use? the part (§ 59.104, § 59.406, § 60.8(b), § 61.13(h)(1)(ii), § 63.7(f), or § 65.158(a)(2)(iii)). 7.0 How do I ensure sample stability? You must receive the Administrator’s writ- ten approval to use the candidate test meth- DETERMINATION OF BIAS AND PRECISION od before you use the candidate test method to meet the applicable federal requirements. -

2017 Automated Liquid Handling Resource Guide
The Lab Manager AUTOMATED LIQUID HANDLING RESOURCE GUIDE What You Need to Know When Buying an Automated Liquid Handling System BY RYAN ACKERMAN Upgrading Stand-Alone Automated Liquid Handling Systems to Workstations BY ANGELO DEPALMA, PhD Small-Volume Liquid Handling Requires Advanced Technology BY MIKE MAY, PhD 20 10 Common Liquid Handling Errors & How to Avoid Them 17 BY LAB MANAGER The Latest in Pipette Tip Design BY ANGELO DEPALMA, PhD Automated Liquid Handling Product Finder BY LAB MANAGER Automatic Liquid Handling Resource Guide 2017 What You Need to Know When Buying an Automated Liquid Handling System Automated liquid handlers come in a seemingly endless variety of configurations, with many different specifications. By Ryan Ackerman MAINTENANCE TIP: AUTOMATED LIQUID HANDLING The signs that you should get your automated liquid handler serviced are fairly obvious. If the system is constantly experiencing glitches or producing inconsistent, unreliable results, it’s probably time to do some maintenance. Mechanical problems are another hint that you may want to call your service technician—these issues can include pipette tips being out of calibration, the deck not being “framed” correctly, belts being worn out, or the pipette stages being out of alignment. Why is it important to know the typical sample How does the sensitivity to contamination affect the type volume being used? of automated liquid handler required? Automated liquid handlers come in a seemingly endless variety of As the instrumentation used to analyze samples becomes more configurations, with many different specifications. An important one sensitive, the methods used to transport and prepare the samples to consider is what minimum, or maximum dispensing volume is must become resistant to contamination to ensure no carryover correct for your processes. -

Q & a – Accuracy and Precision
Food Analysis – FScN 4312W Laboratory: Assessment of Accuracy and Precision Key to Questions 1. Theoretically, how are standard deviation, coefficient of variation, mean, percent relative error, and 95% confidence interval affected by: (1) more replicates, and (2) a larger size of measurement? Was this evident in looking at the actual results obtained using the volumetric pipettes and the buret, with n = 3 vs n = 6, and with 1mL vs 10mL? Ans: (1) As the sample size increases (more replicates) - Calculated mean approaches the true mean - Standard deviation is inversely proportional to the square root of sample size, hence it decreases - CV decreases, as standard deviation approaches 0 for larger sample size - % Relative error decreases as calculated mean approaches true mean - 95% confidence interval narrows down (2) Larger size of measurement - Mean is close to true mean - Standard deviation might be larger due to larger size - CV decreases - % Relative error decreases, as calculated mean is close to true mean - 95% confidence interval narrows down Not all of the above assumptions were confirmed in this experiment according to the data observed. One reason could be that the sample size was very small (n = 3 and n = 6), to notice significant differences, another reason could be human error and variation from one person’s measuring technique to another. 2. Why are percent relative error and coefficient of variation used to compare the accuracy and precision, respectively, of the volumes from pipetting/dispensing 1 and 10mL with the volumetric pipettes and buret in parts 2 and 3, rather than simply the mean and standard deviation, respectively? Ans: Mean calculated from the readings gives the calculated mean, which may differ from the true mean. -

Introducing Automation to Your Lab
Introducing Automation to Your Lab A step-by-step reference guide for the 21st Century biologist Written by Opentrons © OPENTRONS 2019 | INTRODUCING AUTOMATION TO YOUR LAB 1 INTRODUCTION Table of Contents CONTENT FIGURES Introduction Dispelling Myths About Lab Automation 03 Figure 1 Busting The Top 5 Automation Myths 03 Chapter 1 The Overall Benefits Of Lab Automation 05 Figure 2 Top 5 Benefits Of Lab Automation 05 Chapter 2 Choosing The Best Workflow To Automate 06 Figure 3 Your Workflow Is A Good Candidate For 06 Automation If... Chapter 3 Precision And Lab Automation 08 Figure 4 Translating Manual Processes To Automation 07 Chapter 4 How Much Liquid To Move? 09 – Volume Ranges Figure 5 Levels of Throughput 09 – Low, Medium, or High Throughput? Figure 6 Robot Pricing Table 12 Chapter 5 Space Considerations 10 Figure 7 The Opentrons Approach: 14 – Sterility Figure 8 Next Steps When Purchasing An Automated 16 – Size Lab Robot Chapter 6 Cash Considerations 11 Chapter 7 Developing Your Protocol 14 Chapter 8 Choosing The Right Vendor 15 Conclusion Next Steps On The Path To Automation 16 Appendix Resources 17 © OPENTRONS 2019 | INTRODUCING AUTOMATION TO YOUR LAB 2 INTRODUCTION Dispelling the Myths of Lab Automation “Repetitio est mater studiorum,” says the Latin FIGURE 1 proverb: “Repetition is the mother of all learning.” Busting the top 5 automation myths Scientists live this ancient saying every day, winning hard-earned discoveries by repeating the same process over and over. In some cases, this simply MYTH REALITY builds a large enough sample size to create statistical significance in the results. -

2021 Product Guide
2021 PRODUCT GUIDE | LIQUID HANDLING | PURIFICATION | EXTRACTION | SERVICES TABLE OF CONTENTS 2 | ABOUT GILSON 56 | FRACTION COLLECTORS 4 | COVID-19 Solutions 56 | Fraction Collector FC 203B 6 | Service Experts Ready to Help 57 | Fraction Collector FC 204 7 | Services & Support 8 | OEM Capabilities 58 | AUTOMATED LIQUID HANDLERS 58 | Liquid Handler Overview/selection Guide 10 | LIQUID HANDLING 59 | GX-271 Liquid Handler 11 | Pipette Selection Guide 12 | Pipette Families 60 | PUMPS 14 | TRACKMAN® Connected 60 | Pumps Overview/Selection Guide 16 | PIPETMAN® M Connected 61 | VERITY® 3011 18 | PIPETMAN® M 62 | Sample Loading System/Selection Guide 20 | PIPETMAN® L 63 | VERITY® 4120 22 | PIPETMAN® G 64 | DETECTORS 24 | PIPETMAN® Classic 26 | PIPETMAN® Fixed Models 66 | PURIFICATION 28 | Pipette Accessories 67 | VERITY® CPC Lab 30 | PIPETMAN® DIAMOND Tips 68 | VERITY® CPC Process 34 | PIPETMAN® EXPERT Tips 70 | LC Purification Systems 36 | MICROMAN® E 71 | Gilson Glider Software 38 | DISTRIMAN® 72 | VERITY® Oligonucleotide Purification System 39 | REPET-TIPS 74 | Accessories Overview/Selection Guide 40 | MACROMAN® 75 | Racks 41 | Serological Pipettes 43 | PLATEMASTER® 76 | GEL PERMEATION 44 | PIPETMAX® CHROMATOGRAPHY (GPC) 76 | GPC Overview/Selection Guide 46 | BENCHTOP INSTRUMENTS 77 | VERITY® GPC Cleanup System 46 | Safe Aspiration Station & Kit 47 | DISPENSMAN® 78 | EXTRACTION 48 | TRACKMAN® 78 | Automated Extraction Overview/ 49 | Digital Dry Bath Series Selection Guide 49 | Roto-Mini Plus 80 | ASPEC® 274 System 50 | Mini Vortex Mixer 81 | ASPEC® PPM 50 | Vortex Mixer 82 | ASPEC® SPE Cartridges 51 | Digital Mini Incubator 84 | Gilson SupaTop™ Syringe Filters 86 | EXTRACTMAN® 52 | CENTRIFUGES 52 | CENTRY™ 103 Minicentrifuge 88 | SOFTWARE 53 | CENTRY™ 117 Microcentrifuge 88 | Software Selection Guide 53 | CENTRY™ 101 Plate Centrifuge 54 | PERISTALTIC PUMP 54 | MINIPULS® 3 Pump & MINIPULS Tubing SHOP ONLINE WWW.GILSON.COM 1 ABOUT US Gilson is a family-owned global manufacturer of sample management and purification solutions for the life sciences industry. -

Laboratory Equipment Used in Filtration
KNOW YOUR LAB EQUIPMENTS Test tube A test tube, also known as a sample tube, is a common piece of laboratory glassware consisting of a finger-like length of glass or clear plastic tubing, open at the top and closed at the bottom. Beakers Beakers are used as containers. They are available in a variety of sizes. Although they often possess volume markings, these are only rough estimates of the liquid volume. The markings are not necessarily accurate. Erlenmeyer flask Erlenmeyer flasks are often used as reaction vessels, particularly in titrations. As with beakers, the volume markings should not be considered accurate. Volumetric flask Volumetric flasks are used to measure and store solutions with a high degree of accuracy. These flasks generally possess a marking near the top that indicates the level at which the volume of the liquid is equal to the volume written on the outside of the flask. These devices are often used when solutions containing dissolved solids of known concentration are needed. Graduated cylinder Graduated cylinders are used to transfer liquids with a moderate degree of accuracy. Pipette Pipettes are used for transferring liquids with a fixed volume and quantity of liquid must be known to a high degree of accuracy. Graduated pipette These Pipettes are calibrated in the factory to release the desired quantity of liquid. Disposable pipette Disposable transfer. These Pipettes are made of plastic and are useful for transferring liquids dropwise. Burette Burettes are devices used typically in analytical, quantitative chemistry applications for measuring liquid solution. Differing from a pipette since the sample quantity delivered is changeable, graduated Burettes are used heavily in titration experiments. -

Thermo Scientific Pipetting Guide
Thermo Scientific Pipetting Guide Tips for Good Laboratory Pipetting Part of Thermo Fisher Scientific Thermo Scientific Thermo Scientific Contents Introduction 3 Pipetting Pipetting Guide Pipetting terms 3 Types of pipettes 4 General pipetting guidelines 6 Pipetting techniques 6 Tip information 7 Recommendations for pipetting different compounds 8 Ensuring optimum performance 8 Usage of Finpipette Novus 9 Factors affecting the accuracy of air displacement pipettes 10 Preventing cross-contamination 11 Maintenance of your Finnpipettes 1 Autoclaving 1 UV resistance 1 Calibrating your pipettes (incl. conversion table) 13 On-line pipette calibration 15 General guidelines for decontaminating pipettes 16 Chemical compatibility of plastics 18 Customer support 19 Trouble shooting 19 Thermo Scientific Over 35 Years of Innovation A leader in pipetting For over 35 years we have led the way in liquid handling products and microplate instrumentation. We have always ensured that innovation, ergonomics, accuracy, precision and safety are key aspects of our products’ designs. In 1971 we introduced Thermo Scientific Finnpipettes, the world’s first continuously variable micropi- pettes. In 1976 we introduced the world’s first multichannel pipette. Since then we have continuously improved our pipettes, always leading the way with ergonomic design. Over the last 35 years, innovations such as the improved finger rest, soft-touch tip ejection and super blow-out have made Finnpipettes increasingly user-friendly. Our intensive research program and commitment to our customers forms the foundation for future innovations. The new demands in pipette applications are the key drivers of our product development. To date, over 3 mil- lion Finnpipettes have been sold in 150 countries. -

TOP-014, Technical Operating Procedure for Spectrophotometric
CENTER FOR NUCLEAR WASTE Proc. T0P-014 REGULATORY ANALYSES Revision ° TECHNICAL OPERATING PROCEDURE Page I of 11 Title TECHNICAL OPERATING PROCEDURE FOR SPECTROPHOTOMETRIC DETERMINATION OF SILICA EFFECTIVITY AND APPROVAL Revision n of this procedure became effective on 03/04/91 This procedure consists of the pages and changes listed below. Page No. Change Date Effective ALL 03/04/91 Superseaes Procedure No. None Approvals Written By Technical Revie / CENTER FOR NUCLEAR WASTE Proc.TOP-014 REGULATORY ANALYSES Revision _ TECHNICAL OPERATING PROCEDURE Page 2 of 1il TOP-014 TECHNICAL OPERATING PROCEDURE FOR SPECTROPHOTOMETRIC DETERMINATION OF SILICA 1. PURPOSE The purpose of this procedure is to describe a general method for the spectrophotometric determination of silica (Si02) in aqueous solutions. This procedure will be used for aqueous solutions generated by geochemical experiments involving silicate minerals (e.g., zeolites and feldspars) but may also be used for analyses of water samples collected from laboratory experiments or in the field. This procedure implements the requirements of CQAM Section 3. 2. APPLICABLE DOCUMENT The following document forms a part of this procedure, as applicable: Operator's Manual for Milton Roy Spectronic 1201 Spectrophotometer (1988) 3. RESPONSIBILITY (1) The Geosciences Element Manager shall be responsible for the development and maintainance of this procedure. (2) The cognizant Principal Investigator shall be responsible for the implementation of this procedure. (3) Personnel performing tasks described in this procedure are responsible for complying with its requirements. 4. EQUIPMENT (1) Milton Roy Spectronic 1201 Spectrophotometer with microprocessor-controlled functions and features including a linear curve fit test mode or equivalent. -

10. Resource Requirement for Medical Laboratory Technology
10. RESOURCE REQUIREMENT FOR MEDICAL LABORATORY TECHNOLOGY 10.1 PHYSICAL RESOURCES 10.1.1. SPACE REQUIREMENT The total space for lecture halls, laboratories may be worked out as per latest AICTE norms 10.1.2 LIST OF EQUIPMENT Note: All the glass wares to be of Borosil make The chemicals may be purchased of reputed make. Sr. Item Quantity No. LIST OF CHEMICALS FOR CHEMISTRY LABORATORY 1. Ferrous ammonium sulphate (Analytical reagent) 3 x 500 grams 2. Oxalic Acid (A.R) 2 x 500 grams 3. Potassium Permanganate (BDH) 3 x 500 grams 4. Sodium Hydroxide (BDH) 2 x 500 grams 5. Sulphuric Acid (AR) 5 litre 6. Hydrochloric Acid (AR) 5 litre 7. Sodium carbonate (A.R) 2 x 500 grams 8. Phenolphthalen (BDH/Merck) 10 grams 9. Methyl orange (SDH) 10 grams 10. Ethyl Alcohol 4 x 500 ml 11. Methyl Alcohol 500 ml 12. Nitric acid 5 litre 13. Glacial Acetic Acid (CP) 1 litre 14. Ammonium sulphate (BDH) 250 grams 15. Copper sulphate (BDH) 500 grams 16. Glucose (BDH) 500 grams 17. Sucrose (BDH) 500 grams 18. Acetone (BDH) 500 grams 19 Potassium Hydroxide (BDH) 2 x 500 grams 20. Iodine 1 x 500 grams 21. Calcium Chloride (BDH) 500 grams 93 Sr. Item Quantity No. 22 NaCl 500 gms 23. Ferric chloride (BDH) 500 grams 24. Starch pH 7.0 500 grams 25. Fructose 250 grams. 26. Gelatin 500 grams. 27. Benzaldehyde 500 ml 28. Ether 500 ml 29. Carbon Tetrachloride 500 ml 30. Ferrous Bicarbonate 500 grams 31. Sodium Bicarbonate 500 grams 32. Albumin 500 grams 33. -

Practical Laboratory Skills for Molecular Biologists.Pdf 3.0 MB
Practical laboratory skills for molecular biologists skills best practice training training best practice skills Practical laboratory skills for molecular biologists First Edition 2016 Co-authors Elena Sanchez Timothy Wilkes Contributors Nicholas Redshaw Acknowledgements With special thanks to Alison Woolford and Vicki Barwick for their help in the production of this guide. Production of this Guide was funded by the UK National Measurement System. First edition 2016 ISBN: 978-0-948926-28-0 This publication should be cited as: E Sanchez, T Wilkes, Practical laboratory skills for Copyright © 2016 LGC molecular biologists, LGC 2016. ISBN: 978-0-948926-28-0 Practical laboratory skills for molecular biologists OBJECTIVES OF THIS GUIDE 3 1 WORKING IN THE LABORATORY 4 1.1 Health and Safety issues 4 1.2 Laboratory environment 10 1.3 Equipment 14 1.4 Method selection 15 2 MEASURING VOLUME 16 2.1 Types of equipment available 16 2.2 Markings on equipment used for volumetric measurements 22 2.3 Selecting a suitable piece of equipment 24 2.4 Cleaning and maintenance of volumetric equipment 26 2.5 Checking the accuracy of volumetric equipment 27 2.6 Checklists for making measurements of volume 28 3 MEASURING MASS 33 3.1 Mass versus weight 33 3.2 Types of balance available 33 3.3 Selecting a suitable balance 34 4 QUANTIFICATION OF NUCLEIC ACID MATERIAL 41 4.1 Measurement of nucleic acid concentration using UV spectroscopy 41 4.2 Fluorescent based systems. 43 4.3 Gel electrophoresis 45 5 MEASURING PH 47 5.1 What is pH? 47 5.2 Equipment for measuring