Test Items for Licensing Examination Krok 2 PHARMACY
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Wo 2010/075090 A2
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date 1 July 2010 (01.07.2010) WO 2010/075090 A2 (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every C07D 409/14 (2006.01) A61K 31/7028 (2006.01) kind of national protection available): AE, AG, AL, AM, C07D 409/12 (2006.01) A61P 11/06 (2006.01) AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, (21) International Application Number: DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, PCT/US2009/068073 HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, (22) International Filing Date: KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, 15 December 2009 (15.12.2009) ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, PE, PG, PH, PL, PT, RO, RS, RU, SC, SD, (25) Filing Language: English SE, SG, SK, SL, SM, ST, SV, SY, TJ, TM, TN, TR, TT, (26) Publication Language: English TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: (84) Designated States (unless otherwise indicated, for every 61/122,478 15 December 2008 (15.12.2008) US kind of regional protection available): ARIPO (BW, GH, GM, KE, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, ZM, (71) Applicant (for all designated States except US): AUS- ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, PEX PHARMACEUTICALS, INC. -

The Organic Chemistry of Drug Synthesis
The Organic Chemistry of Drug Synthesis VOLUME 2 DANIEL LEDNICER Mead Johnson and Company Evansville, Indiana LESTER A. MITSCHER The University of Kansas School of Pharmacy Department of Medicinal Chemistry Lawrence, Kansas A WILEY-INTERSCIENCE PUBLICATION JOHN WILEY AND SONS, New York • Chichester • Brisbane • Toronto Copyright © 1980 by John Wiley & Sons, Inc. All rights reserved. Published simultaneously in Canada. Reproduction or translation of any part of this work beyond that permitted by Sections 107 or 108 of the 1976 United States Copyright Act without the permission of the copyright owner is unlawful. Requests for permission or further information should be addressed to the Permissions Department, John Wiley & Sons, Inc. Library of Congress Cataloging in Publication Data: Lednicer, Daniel, 1929- The organic chemistry of drug synthesis. "A Wiley-lnterscience publication." 1. Chemistry, Medical and pharmaceutical. 2. Drugs. 3. Chemistry, Organic. I. Mitscher, Lester A., joint author. II. Title. RS421 .L423 615M 91 76-28387 ISBN 0-471-04392-3 Printed in the United States of America 10 987654321 It is our pleasure again to dedicate a book to our helpmeets: Beryle and Betty. "Has it ever occurred to you that medicinal chemists are just like compulsive gamblers: the next compound will be the real winner." R. L. Clark at the 16th National Medicinal Chemistry Symposium, June, 1978. vii Preface The reception accorded "Organic Chemistry of Drug Synthesis11 seems to us to indicate widespread interest in the organic chemistry involved in the search for new pharmaceutical agents. We are only too aware of the fact that the book deals with a limited segment of the field; the earlier volume cannot be considered either comprehensive or completely up to date. -

(19) 11 Patent Number: 6165500
USOO6165500A United States Patent (19) 11 Patent Number: 6,165,500 Cevc (45) Date of Patent: *Dec. 26, 2000 54 PREPARATION FOR THE APPLICATION OF WO 88/07362 10/1988 WIPO. AGENTS IN MINI-DROPLETS OTHER PUBLICATIONS 75 Inventor: Gregor Cevc, Heimstetten, Germany V.M. Knepp et al., “Controlled Drug Release from a Novel Liposomal Delivery System. II. Transdermal Delivery Char 73 Assignee: Idea AG, Munich, Germany acteristics” on Journal of Controlled Release 12(1990) Mar., No. 1, Amsterdam, NL, pp. 25–30. (Exhibit A). * Notice: This patent issued on a continued pros- C.E. Price, “A Review of the Factors Influencing the Pen ecution application filed under 37 CFR etration of Pesticides Through Plant Leaves” on I.C.I. Ltd., 1.53(d), and is subject to the twenty year Plant Protection Division, Jealott's Hill Research Station, patent term provisions of 35 U.S.C. Bracknell, Berkshire RG12 6EY, U.K., pp. 237-252. 154(a)(2). (Exhibit B). K. Karzel and R.K. Liedtke, “Mechanismen Transkutaner This patent is Subject to a terminal dis- Resorption” on Grandlagen/Basics, pp. 1487–1491. (Exhibit claimer. C). Michael Mezei, “Liposomes as a Skin Drug Delivery Sys 21 Appl. No.: 07/844,664 tem” 1985 Elsevier Science Publishers B.V. (Biomedical Division), pp 345-358. (Exhibit E). 22 Filed: Apr. 8, 1992 Adrienn Gesztes and Michael Mazei, “Topical Anesthesia of 30 Foreign Application Priority Data the Skin by Liposome-Encapsulated Tetracaine” on Anesth Analg 1988; 67: pp 1079–81. (Exhibit F). Aug. 24, 1990 DE) Germany ............................... 40 26834 Harish M. Patel, "Liposomes as a Controlled-Release Sys Aug. -
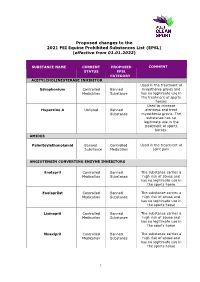
Proposed Changes to the 2021 FEI Equine Prohibited Substances List (EPSL) (Effective from 01.01.2022)
Proposed changes to the 2021 FEI Equine Prohibited Substances List (EPSL) (effective from 01.01.2022) SUBSTANCE NAME CURRENT PROPOSED COMMENT STATUS EPSL CATEGORY ACETYLCHOLINESTERASE INHIBITOR Used in the treatment of Edrophonium Controlled Banned myasthenia gravis and Medication Substance has no legitimate use in the treatment of sports horses Used to increase Huperzine A Unlisted Banned alertness and treat Substance myasthenia gravis. The substance has no legitimate use in the treatment of sports horses. AMIDES Palmitoylethanolamid Banned Controlled Used in the treatment of Substance Medication joint pain ANGIOTENSIN CONVERTING ENZYME INHIBITORS Enalapril Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse Enalaprilat Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse Lisinopril Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse Moexipril Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse 1 Perindoprilat Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse ANTIHISTAMINES Antazoline Controlled Banned The substance has no Medication Substance legitimate use in the sports horse Azatadine Controlled Banned The substance has Medication Substance sedative effects -

BMJ Open Is Committed to Open Peer Review. As Part of This Commitment We Make the Peer Review History of Every Article We Publish Publicly Available
BMJ Open is committed to open peer review. As part of this commitment we make the peer review history of every article we publish publicly available. When an article is published we post the peer reviewers’ comments and the authors’ responses online. We also post the versions of the paper that were used during peer review. These are the versions that the peer review comments apply to. The versions of the paper that follow are the versions that were submitted during the peer review process. They are not the versions of record or the final published versions. They should not be cited or distributed as the published version of this manuscript. BMJ Open is an open access journal and the full, final, typeset and author-corrected version of record of the manuscript is available on our site with no access controls, subscription charges or pay-per-view fees (http://bmjopen.bmj.com). If you have any questions on BMJ Open’s open peer review process please email [email protected] BMJ Open Pediatric drug utilization in the Western Pacific region: Australia, Japan, South Korea, Hong Kong and Taiwan Journal: BMJ Open ManuscriptFor ID peerbmjopen-2019-032426 review only Article Type: Research Date Submitted by the 27-Jun-2019 Author: Complete List of Authors: Brauer, Ruth; University College London, Research Department of Practice and Policy, School of Pharmacy Wong, Ian; University College London, Research Department of Practice and Policy, School of Pharmacy; University of Hong Kong, Centre for Safe Medication Practice and Research, Department -

Clinical Pharmacology of Antiallergic Drugs and Drugs with Action on Immune Processes A
CLINICAL PHARMACOLOGY OF ANTIALLERGIC DRUGS AND DRUGS WITH ACTION ON IMMUNE PROCESSES A. Actuality Allergy is a universal response of the body to the action of various endogenous and exogenous noxious factors, it is a pathological process characteristic of most diseases. Technical and scientific progress modified significantly the environment, this favoring the vertiginous growth of allergic affections incidence, systemic diseases and immunodeficiency states. Currently there is a rich arsenal of these drugs, but for efficient and rational using of their is important to know their pharmacological features. B. Training aim To deepen knowledge of the pharmacology of drug groups used in the treatment allergic diseases and in pathologies involving immune system. Acquire and apply the pharmacokinetic and pharmacodynamic principles to individualize and optimize the administration of these drugs. C. Teaching objectives The students should be able to: a) Elucidate pharmacodynamic and pharmacokinetic peculiarities of drug groups used in immune system diseases. b)Establish dosage principles of drugs depending on group affiliation and etiology and severity of the disease. c)Forecast possible side effects and complications, to establish their dependence on dosing regimen, their prophylaxis. d)Apply contemporary methods of pharmacological and non-pharmacological correction of drug-induced adverse drug reactions. e)Write down the personal form (P-medicines) of drugs used to treat immune system diseases. D. Knowledge from previously studied disciplines and related subjects Medico-biological subjects. Allergic reactions. The basic components of the immune response. Interdependence of alteration, protection and adaptation reactions in pathologies involving immune system. Clinical subjects. Allergic diseases. Their classification, clinical manifestation, pathogenesis and etiology, treatment principles of systemic diseases and immunodeficiency states. -

Pharmacology
STATE ESTABLISHMENT «DNIPROPETROVSK MEDICAL ACADEMY OF HEALTH MINISTRY OF UKRAINE» V.I. MAMCHUR, V.I. OPRYSHKO, А.А. NEFEDOV, A.E. LIEVYKH, E.V.KHOMIAK PHARMACOLOGY WORKBOOK FOR PRACTICAL CLASSES FOR FOREIGN STUDENTS STOMATOLOGY DEPARTMENT DNEPROPETROVSK - 2016 2 UDC: 378.180.6:61:615(075.5) Pharmacology. Workbook for practical classes for foreign stomatology students / V.Y. Mamchur, V.I. Opryshko, A.A. Nefedov. - Dnepropetrovsk, 2016. – 186 p. Reviewed by: N.I. Voloshchuk - MD, Professor of Pharmacology "Vinnitsa N.I. Pirogov National Medical University.‖ L.V. Savchenkova – Doctor of Medicine, Professor, Head of the Department of Clinical Pharmacology, State Establishment ―Lugansk state medical university‖ E.A. Podpletnyaya – Doctor of Pharmacy, Professor, Head of the Department of General and Clinical Pharmacy, State Establishment ―Dnipropetrovsk medical academy of Health Ministry of Ukraine‖ Approved and recommended for publication by the CMC of State Establishment ―Dnipropetrovsk medical academy of Health Ministry of Ukraine‖ (protocol №3 from 25.12.2012). The educational tutorial contains materials for practical classes and final module control on Pharmacology. The tutorial was prepared to improve self-learning of Pharmacology and optimization of practical classes. It contains questions for self-study for practical classes and final module control, prescription tasks, pharmacological terms that students must know in a particular topic, medical forms of main drugs, multiple choice questions (tests) for self- control, basic and additional references. This tutorial is also a student workbook that provides the entire scope of student’s work during Pharmacology course according to the credit-modular system. The tutorial was drawn up in accordance with the working program on Pharmacology approved by CMC of SE ―Dnipropetrovsk medical academy of Health Ministry of Ukraine‖ on the basis of the standard program on Pharmacology for stomatology students of III - IV levels of accreditation in the specialties Stomatology – 7.110105, Kiev 2011. -

Histamine Receptor
Histamine Receptor Histamine Receptors are a class of G protein-coupled receptors with histamine as their endogenous ligand. There are four known histamine receptors: H1 receptor, H2 receptor, H3 receptor, H4 receptor. The H1 receptor is a histamine receptor belonging to the family of Rhodopsin-like G-protein-coupled receptors. This receptor, which is activated by the biogenic amine histamine, is expressed throughout the body, to be specific, in smooth muscles, on vascular endothelial cells, in the heart, and in the central nervous system. H2 receptors are positively coupled to adenylate cyclase via Gs. It is a potent stimulant of cAMP production, which leads to activation of Protein Kinase A. Histamine H3 receptors are expressed in the central nervous system and to a lesser extent the peripheral nervous system, where they act asautoreceptors in presynaptic histaminergic neurons, and also control histamine turnover by feedback inhibition of histamine synthesis and release. The Histamine H4 receptor has been shown to be involved in mediating eosinophil shape change and mast cell chemotaxis. www.MedChemExpress.com 1 Histamine Receptor Inhibitors & Modulators (±)-Methotrimeprazine (D6) (±)-Tazifylline (dl-Methotrimeprazine D6) Cat. No.: HY-19489S Cat. No.: HY-U00018 Bioactivity: (±)-Methotrimeprazine (D6) is the deuterium labeled Bioactivity: (±)-Tazifylline is a potent, selective and long-acting Methotrimeprazine, which is a D3 dopamine and Histamine H1 histamine H1 receptor antagonist. receptor antagonist. Purity: >98% Purity: >98% Clinical Data: No Development Reported Clinical Data: No Development Reported Size: 1 mg Size: 1 mg, 5 mg, 10 mg, 20 mg ABT-239 Acrivastine Cat. No.: HY-12195 (BW825C) Cat. No.: HY-B1510 Bioactivity: ABT-239 is a novel, highly efficacious, Bioactivity: Acrivastine (BW825C) is a short acting histamine 1 non-imidazole class of H3R antagonist and a transient receptor antagonist for the treatment of allergic rhinitis. -

(12) Patent Application Publication (10) Pub. No.: US 2007/0042030 A1 Cevc (43) Pub
US 20070042030A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2007/0042030 A1 Cevc (43) Pub. Date: Feb. 22, 2007 (54) PREPARATION FOR THE APPLICATION OF Mar. 6, 1991 (DE).............................................. 4107 153 AGENTS IN MINI-DROPLETS Mar. 6, 1991 (DE).............................................. 4107 152 Aug. 22, 1991 (WO)........................... PCT/EP91/O1596 (75) Inventor: Gregor Cevc, Heinstetten (DE) Publication Classification Correspondence Address: EDWARDS & ANGELL, LLP (51) Int. Cl. P.O. BOX SS874 A 6LX 9/27 (2006.01) BOSTON, MA 02205 (US) (52) U.S. Cl. ............................................ 424/450; 977/907 (73) Assignee: IDEA AG, Munchen (DE) (57) ABSTRACT (21) Appl. No.: 11/481,804 The invention relates to a preparation for the application of 1-1. agents in the form of minuscule droplets of fluid, in par (22) Filed: Jul. 5, 2006 ticular provided with membrane-like structures consisting of Related U.S. Application Data one or several layers of amphiphilic molecules, or an AV amphiphilic carrier Substance, in particular for transporting (63) Continuation of application No. 09/621,574, filed on the agent into and through natural barriers such as skin and Jul. 21, 2000, which is a continuation of application similar materials. The preparation contains a concentration No. 07/844 644 filed on Apr. 23, 1992, now aban- of edge active substances which amounts to up to 99 mol-% doned. sy is s s of the agent concentration which is required for the induc tion of droplet solubilization. Such preparations are suitable, (30) Foreign Application Priority Data for example, for the non-invasive applications of antidia betics, in particular of insulin. -

The Discovery of New and More Potent Chloropyramine (C4) Analogues for the Potential Treatment of Invasive Breast Cancer
The discovery of new and more potent chloropyramine (C4) analogues for the potential treatment of invasive breast cancer Sahar Kandil*, Filippo Prencipe, Samuel Jones, Stephen Hiscox and Andrew D. Westwell School of Pharmacy & Pharmaceutical Sciences, Cardiff University, King Edward VII Avenue, Cardiff, CF10 3NB, Wales, United Kingdom. Keywords: Focal adhesion kinase (FAK), metastatic breast cancer (BC), chloropyramine (C4), lead optimisation, reductive amination. Graphical abstract Highlights • 17 New chloropyramine analogues were designed and synthesised. • All new compounds were evaluated for their activity in three different breast cancer cell lines. • For the most active compound 5c, in vitro pharmacokinetic studies were performed. ABSTRACT Breast cancer (BC) is the second most common cancer worldwide, accounting for 25% of all female cancers. Although the survival rate has increased significantly in the past few decades, patients who develop secondary site metastasis as well as those diagnosed with triple negative breast cancer (TNBC) still represent a real unmet medical challenge. Previous studies have shown that chloropyramine (C4) inhibits FAK-VEGFR3 signaling. More recently, C4 is reported to have SASH1 inducing properties. However, C4 exerts its antitumour and antiangiogenic effects at high micromolar concentrations (>100 µM) that would not be compatible with further drug development against invasive breast cancer driven by FAK signaling. In this study, molecular modeling guided structural modifications have been introduced to the chloropyramine C4 scaffold to improve its activity in breast cancer cell lines. Seventeen compounds were designed and synthesised and their anti-proliferative activity was evaluated against three human breast cancer lines (MDA-MB-231, BT474 and T47D). Compound 5c was identified to display an average activity of IC50 = 23.5- 31.3 µM, which represents a significant improvement of C4 activity in the same assay model. -

SASH1 Mediates Sensitivity of Breast Cancer Cells to Chloropyramine and Is Associated with Prognosis in Breast Cancer
www.impactjournals.com/oncotarget/ Oncotarget, Vol. 7, No. 45 Research Paper SASH1 mediates sensitivity of breast cancer cells to chloropyramine and is associated with prognosis in breast cancer Joshua T. Burgess1, Emma Bolderson1,2, Jodi M. Saunus3,4, Shu-Dong Zhang5,6, Lynne E. Reid3,4, Anne Marie McNicol3, Sunil R. Lakhani3,7,8, Katharine Cuff2, Kerry Richard8,10, Derek J. Richard1,9, Kenneth J. O’Byrne1,2,9 1Cancer and Ageing Research Program, Institute of Health and Biomedical Innovation at the Translational Research Institute (TRI), Queensland University of Technology, Brisbane, Australia 2Princess Alexandra Hospital, Woolloongabba, Brisbane, Queensland, Australia 3The University of Queensland (UQ), UQ Centre for Clinical Research, Herston, Queensland, Australia 4QIMR Berghofer Medical Research Institute, Herston, Queensland, Australia 5 Northern Ireland Centre for Stratified Medicine, University of Ulster, Altnagelvin Hospital Campus, Londonderry, UK 6Center for Cancer Research and Cell Biology, Queen’s University Belfast, United Kingdom 7Pathology Queensland, Royal Brisbane Women’s Hospital, Herston, Queensland, Australia 8UQ School of Medicine, Herston, Queensland, Australia 9Translational Cell Imaging Queensland, Translational Research Institute, Queensland, Australia 10Conjoint Endocrine Laboratory, Pathology Queensland, Queensland Health, Herston, Australia Correspondence to: Derek J. Richard, email: [email protected] Kenneth J. O’Byrne, email: [email protected] Keywords: SASH1, biomarker, breast cancer, chloropyramine Received: March 29, 2016 Accepted: September 02, 2016 Published: September 14, 2016 ABSTRACT Expression of the SASH1 protein is reduced in a range of human cancers and has been implicated in apoptotic cancer cell death. This study investigated whether increasing SASH1 expression could be a useful therapeutic strategy in breast cancer. -
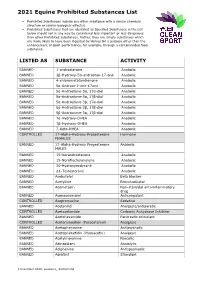
2021 Equine Prohibited Substances List
2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). Prohibited Substances that are identified as Specified Substances in the List below should not in any way be considered less important or less dangerous than other Prohibited Substances. Rather, they are simply substances which are more likely to have been ingested by Horses for a purpose other than the enhancement of sport performance, for example, through a contaminated food substance. LISTED AS SUBSTANCE ACTIVITY BANNED 1-androsterone Anabolic BANNED 3β-Hydroxy-5α-androstan-17-one Anabolic BANNED 4-chlorometatandienone Anabolic BANNED 5α-Androst-2-ene-17one Anabolic BANNED 5α-Androstane-3α, 17α-diol Anabolic BANNED 5α-Androstane-3α, 17β-diol Anabolic BANNED 5α-Androstane-3β, 17α-diol Anabolic BANNED 5α-Androstane-3β, 17β-diol Anabolic BANNED 5β-Androstane-3α, 17β-diol Anabolic BANNED 7α-Hydroxy-DHEA Anabolic BANNED 7β-Hydroxy-DHEA Anabolic BANNED 7-Keto-DHEA Anabolic CONTROLLED 17-Alpha-Hydroxy Progesterone Hormone FEMALES BANNED 17-Alpha-Hydroxy Progesterone Anabolic MALES BANNED 19-Norandrosterone Anabolic BANNED 19-Noretiocholanolone Anabolic BANNED 20-Hydroxyecdysone Anabolic BANNED Δ1-Testosterone Anabolic BANNED Acebutolol Beta blocker BANNED Acefylline Bronchodilator BANNED Acemetacin Non-steroidal anti-inflammatory drug BANNED Acenocoumarol Anticoagulant CONTROLLED Acepromazine Sedative BANNED Acetanilid Analgesic/antipyretic CONTROLLED Acetazolamide Carbonic Anhydrase Inhibitor BANNED Acetohexamide Pancreatic stimulant CONTROLLED Acetominophen (Paracetamol) Analgesic BANNED Acetophenazine Antipsychotic BANNED Acetophenetidin (Phenacetin) Analgesic BANNED Acetylmorphine Narcotic BANNED Adinazolam Anxiolytic BANNED Adiphenine Antispasmodic BANNED Adrafinil Stimulant 1 December 2020, Lausanne, Switzerland 2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s).