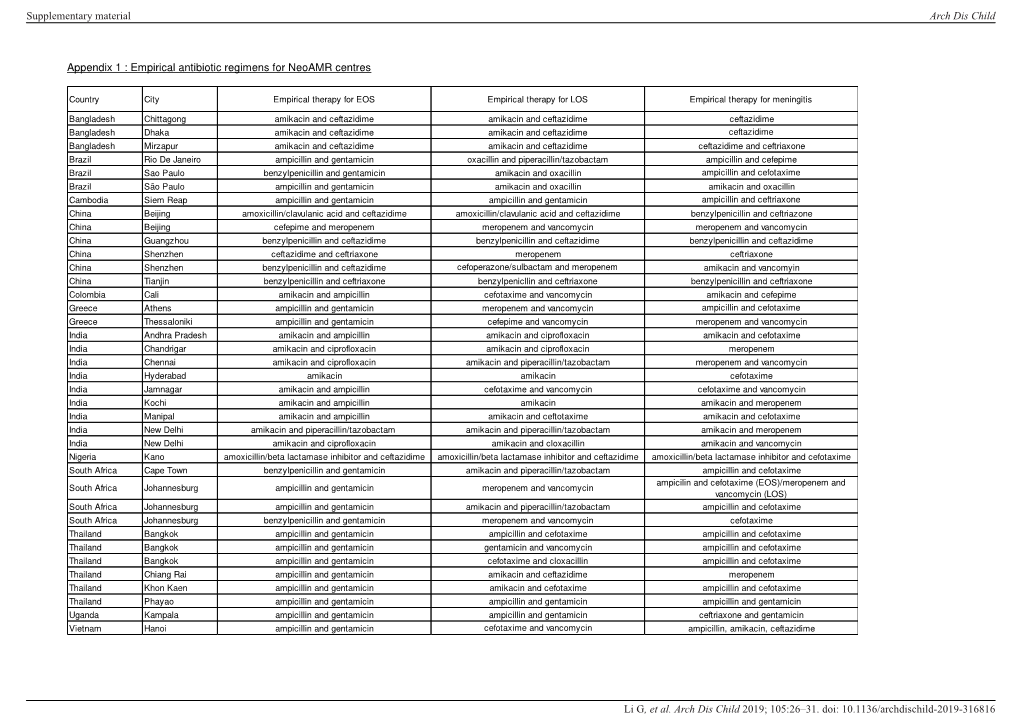
Appendix 1 : Empirical Antibiotic Regimens for Neoamr Centres
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 200327 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 200327 Priority or Standard Standard Submit Date(s) December 29, 2009 Received Date(s) December 30, 2009 PDUFA Goal Date October 30, 2010 Division / Office Division of Anti-Infective and Ophthalmology Products Office of Antimicrobial Products Reviewer Name(s) Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD Review Completion October 29, 2010 Date Established Name Ceftaroline fosamil for injection (Proposed) Trade Name Teflaro Therapeutic Class Cephalosporin; ß-lactams Applicant Cerexa, Inc. Forest Laboratories, Inc. Formulation(s) 400 mg/vial and 600 mg/vial Intravenous Dosing Regimen 600 mg every 12 hours by IV infusion Indication(s) Acute Bacterial Skin and Skin Structure Infection (ABSSSI); Community-acquired Bacterial Pneumonia (CABP) Intended Population(s) Adults ≥ 18 years of age Template Version: March 6, 2009 Reference ID: 2857265 Clinical Review Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD NDA 200327: Teflaro (ceftaroline fosamil) Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 9 1.1 Recommendation on Regulatory Action ........................................................... 10 1.2 Risk Benefit Assessment.................................................................................. 10 1.3 Recommendations for Postmarketing Risk Evaluation and Mitigation Strategies ........................................................................................................................ -

“Ceftriaxone– Sulbactam–EDTA” and Various Antibiotics Against Gram
ORIGINAL ARTICLE A Comparative In Vitro Sensitivity Study of “Ceftriaxone– Sulbactam–EDTA” and Various Antibiotics against Gram- negative Bacterial Isolates from Intensive Care Unit Sweta Singh1, Chinmoy Sahu2, Sangram Singh Patel3, Abhay Singh4, Nidhi Yaduvanshi5 ABSTRACT Introduction: A rapid increase in multidrug-resistant (MDR) strains is being seen across the globe especially in the Southeast Asian region, including India. Carbapenems and colistin form the mainstay of treatment against gram-negative pathogens, especially extended-spectrum beta-lactamase (ESBL)- and metallo-beta-lactamse (MBL)-producing isolates. However, due to increased resistance to carbapenems and toxicity of colistin, especially in intensive care units (ICUs), carbapenem-sparing antibiotics like ceftriaxone–sulbactam–EDTA (CSE) combination needs to be evaluated. Materials and methods: Bacterial isolates cultured from various clinical samples from all ICUs for a period of 9 months were evaluated. Bacterial identification was performed by matrix assisted laser desorption ionization time of flight mass spectrometry (MALDI-TOF MS) and antibiotic susceptibility testing were performed by disk diffusion and E test method. Antibiogram of various antibiotics was noted. Extended-spectrum beta-lactamase- and MBL-producing bacteria were identified by phenotypic methods. Antibiotic sensitivity results of CSE were compared with the comparator drugs like colistin, carbapenems, and tigecycline in Enterobacteriaceae, Acinetobacter spp., and Pseudomonas spp. along with ESBL and MBL producers. Results: A total of 2,760 samples of blood, cerebrospinal fluid (CSF), respiratory samples, tissue, and pus were collected from ICUs with maximum isolates from pus (37%) followed by respiratory samples (31%) and blood (27%). Escherichia coli and Klebsiella pneumoniae were the predominant gram-negative pathogens accounting for 56% of the isolates followed by Acinetobacter spp. -

Cefoperazone Sodium
Disclaimer While these labels are the most current Drug Control Authority (DCA) approved versions of content, these are not necessarily reflective of final printed labeling currently in distribution LPD Title : Cefoperazone Sodium LPD Date : 07 February 2017 Country : Malaysia Reference : CDS version 5.0 dated 07 January 2017 Reason : Addition of adverse drug reactions (ADRs) Haemorrhage and Coagulopathy to Section 4.8 Undesirable effects, and also, addition of serious hemorrhage and coagulopathy to the warning about vitamin K deficiency in Section 4.4 Special warnings and precautions for use Addition of ADRs Anaphylactic reaction and Anaphylactic shock to Section 4.8 Undesirable effects Addition of ADR Dermatitis exfoliative to Section 4.8 Undesirable Effects and a warning statement on severe cutaneous adverse reactions (SCARs) to Section 4.4, Special warnings and precautions for use 1. NAME OF THE MEDICINAL PRODUCT CEFOBID 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Cefoperazone sodium is a semisynthetic broad-spectrum cephalosporin antibiotic for parenteral use only. Cefoperazone contains 34 mg sodium (1.5 mEq) per gram. Cefoperazone is a white crystalline powder which is freely soluble in water. The pH of a 25% aqueous solution is 5.0 to 6.5 and the solution is colorless to straw-yellow, depending on the concentration. The empirical formula is C25H26N9NaO8S2. 3. PHARMACEUTICAL FORM Cefoperazone sodium is available in vials containing 1 and 2 grams. Not For Distribution PFIZER CONFIDENTIAL Not all product strengths or pack sizes -

Lactamase-Producing Antibiotic-Resistant Escherichia Coli and Klebsiella Pneumoniae in Ready-To-Eat Street Foods
antibiotics Article Prevalence and Characterization of Extended-Spectrum β-Lactamase-Producing Antibiotic-Resistant Escherichia coli and Klebsiella pneumoniae in Ready-to-Eat Street Foods Shobha Giri 1 , Vaishnavi Kudva 1, Kalidas Shetty 2 and Veena Shetty 1,* 1 Department of Microbiology, KS Hegde Medical Academy, Nitte (Deemed to be University), Mangalore 575018, India; [email protected] (S.G.); [email protected] (V.K.) 2 Department of Plant Sciences, North Dakota State University, Fargo, ND 58102, USA; [email protected] * Correspondence: [email protected] Abstract: As the global urban populations increase with rapid migration from rural areas, ready-to- eat (RTE) street foods are posing food safety challenges where street foods are prepared with less structured food safety guidelines in small and roadside outlets. The increased presence of extended- spectrum-β-lactamase (ESBL) producing bacteria in street foods is a significant risk for human health because of its epidemiological significance. Escherichia coli and Klebsiella pneumoniae have become important and dangerous foodborne pathogens globally for their relevance to antibiotic resistance. The present study was undertaken to evaluate the potential burden of antibiotic-resistant E. coli and K. pneumoniae contaminating RTE street foods and to assess the microbiological quality of foods in a Citation: Giri, S.; Kudva, V.; Shetty, typical emerging and growing urban suburb of India where RTE street foods are rapidly establishing K.; Shetty, V. Prevalence and with public health implications. A total of 100 RTE food samples were collected of which, 22.88% Characterization of were E. coli and 27.12% K. pneumoniae. The prevalence of ESBL-producing E. -

Consideration of Antibacterial Medicines As Part Of
Consideration of antibacterial medicines as part of the revisions to 2019 WHO Model List of Essential Medicines for adults (EML) and Model List of Essential Medicines for children (EMLc) Section 6.2 Antibacterials including Access, Watch and Reserve Lists of antibiotics This summary has been prepared by the Health Technologies and Pharmaceuticals (HTP) programme at the WHO Regional Office for Europe. It is intended to communicate changes to the 2019 WHO Model List of Essential Medicines for adults (EML) and Model List of Essential Medicines for children (EMLc) to national counterparts involved in the evidence-based selection of medicines for inclusion in national essential medicines lists (NEMLs), lists of medicines for inclusion in reimbursement programs, and medicine formularies for use in primary, secondary and tertiary care. This document does not replace the full report of the WHO Expert Committee on Selection and Use of Essential Medicines (see The selection and use of essential medicines: report of the WHO Expert Committee on Selection and Use of Essential Medicines, 2019 (including the 21st WHO Model List of Essential Medicines and the 7th WHO Model List of Essential Medicines for Children). Geneva: World Health Organization; 2019 (WHO Technical Report Series, No. 1021). Licence: CC BY-NC-SA 3.0 IGO: https://apps.who.int/iris/bitstream/handle/10665/330668/9789241210300-eng.pdf?ua=1) and Corrigenda (March 2020) – TRS1021 (https://www.who.int/medicines/publications/essentialmedicines/TRS1021_corrigenda_March2020. pdf?ua=1). Executive summary of the report: https://apps.who.int/iris/bitstream/handle/10665/325773/WHO- MVP-EMP-IAU-2019.05-eng.pdf?ua=1. -

Cephalosporins Can Be Prescribed Safely for Penicillin-Allergic Patients ▲
JFP_0206_AE_Pichichero.Final 1/23/06 1:26 PM Page 106 APPLIED EVIDENCE New research findings that are changing clinical practice Michael E. Pichichero, MD University of Rochester Cephalosporins can be Medical Center, Rochester, NY prescribed safely for penicillin-allergic patients Practice recommendations an allergic reaction to cephalosporins, ■ The widely quoted cross-allergy risk compared with the incidence of a primary of 10% between penicillin and (and unrelated) cephalosporin allergy. cephalosporins is a myth (A). Most people produce IgG and IgM antibodies in response to exposure to ■ Cephalothin, cephalexin, cefadroxil, penicillin1 that may cross-react with and cefazolin confer an increased risk cephalosporin antigens.2 The presence of of allergic reaction among patients these antibodies does not predict allergic, with penicillin allergy (B). IgE cross-sensitivity to a cephalosporin. ■ Cefprozil, cefuroxime, cefpodoxime, Even penicillin skin testing is generally not ceftazidime, and ceftriaxone do not predictive of cephalosporin allergy.3 increase risk of an allergic reaction (B). Reliably predicting cross-reactivity ndoubtedly you have patients who A comprehensive review of the evidence say they are allergic to penicillin shows that the attributable risk of a cross- U but have difficulty recalling details reactive allergic reaction varies and is of the reactions they experienced. To be strongest when the chemical side chain of safe, we often label these patients as peni- the specific cephalosporin is similar to that cillin-allergic without further questioning of penicillin or amoxicillin. and withhold not only penicillins but Administration of cephalothin, cepha- cephalosporins due to concerns about lexin, cefadroxil, and cefazolin in penicillin- potential cross-reactivity and resultant IgE- allergic patients is associated with a mediated, type I reactions. -

A Thesis Entitled an Oral Dosage Form of Ceftriaxone Sodium Using Enteric
A Thesis entitled An oral dosage form of ceftriaxone sodium using enteric coated sustained release calcium alginate beads by Darshan Lalwani Submitted to the Graduate Faculty as partial fulfillment of the requirements for the Master of Science Degree in Pharmaceutical Sciences with Industrial Pharmacy Option _________________________________________ Jerry Nesamony, Ph.D., Committee Chair _________________________________________ Sai Hanuman Sagar Boddu, Ph.D, Committee Member _________________________________________ Youssef Sari, Ph.D., Committee Member _________________________________________ Patricia R. Komuniecki, PhD, Dean College of Graduate Studies The University of Toledo May 2015 Copyright 2015, Darshan Narendra Lalwani This document is copyrighted material. Under copyright law, no parts of this document may be reproduced without the expressed permission of the author. An Abstract of An oral dosage form of ceftriaxone sodium using enteric coated sustained release calcium alginate beads by Darshan Lalwani Submitted to the Graduate Faculty as partial fulfillment of the requirements for the Master of Science Degree in Pharmaceutical Sciences with Industrial Pharmacy option The University of Toledo May 2015 Purpose: Ceftriaxone (CTZ) is a broad spectrum semisynthetic, third generation cephalosporin antibiotic. It is an acid labile drug belonging to class III of biopharmaceutical classification system (BCS). It can be solvated quickly but suffers from the drawback of poor oral bioavailability owing to its limited permeability through -

National Treatment Guidelines for Antimicrobial Use in Infectious Diseases
National Treatment Guidelines for Antimicrobial Use in Infectious Diseases Version 1.0 (2016) NATIONAL CENTRE FOR DISEASE CONTROL Directorate General of Health Services Ministry of Health & Family Welfare Government of India CONTENTS Chapter 1 .................................................................................................................................................................................................................. 7 Introduction ........................................................................................................................................................................................................ 7 Chapter 2. ................................................................................................................................................................................................................. 9 Syndromic Approach For Empirical Therapy Of Common Infections.......................................................................................................... 9 A. Gastrointestinal & Intra-Abdominal Infections ......................................................................................................................................... 10 B. Central Nervous System Infections ........................................................................................................................................................... 13 C. Cardiovascular Infections ......................................................................................................................................................................... -

The New Fifth-Generation Cephalosporins – a Balance Between Safety and Efficacy
REVIEWS Ref: Ro J Pharm Pract. 2020;13(3) DOI: 10.37897/RJPhP.2020.3.2 The new fifth-generation cephalosporins – a balance between safety and efficacy Aura Rusu1, Ioana-Andreea Lungu2 1 Pharmaceutical and Therapeutical Chemistry Department, Faculty of Pharmacy, George Emil Palade University of Medicine, Pharmacy, Science and Technology, Targu Mures, Romania 2 Doctoral School of Medicine and Pharmacy, George Emil Palade University of Medicine, Pharmacy, Science and Technology, Targu Mures, Romania Abstract Cephalosporins are beta-lactam antibiotics classified into five generations. The newest generation has three representa- tives: ceftaroline fosamile, the combination ceftolozane/tazobactam (cephalosporin/beta-lactamase inhibitor), and ceftobi- prole medocaril. These new cephalosporins are valuable anti-infective agents, with potent activity against multidrug-re- sistant bacteria, and with a positive balance between benefits and side effects. However, the fifth-generation cephalosporins should be judiciously used to prevent the occurrence of bacterial resistance phenomenon. Keywords: cephalosporins, ceftaroline, ceftolozane, ceftobiprole, MRSA, community-acquired pneumonia, complicated skin and soft tissue infections INTRODUCTION emerged as a result of increased only, in the situation where other Cephalosporins (CFs) are beta- bacterial resistance to classical antibacterial drugs were not lactam antibiotics with numerous antibiotics. Based on the efficient. representatives widely used in the antimicrobial activity, the CFs are therapy -

MAGNEX/MAGNEX FORTE Injection
For the use only of a Registered Medical Practitioners or a Hospital or a Laboratory Sulbactam/Cefoperazone 1:1 and 1:2 MAGNEX/MAGNEX FORTE Injection 1. GENERIC NAME Sulbactam/Cefoperazone 1:1 and 1:2 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Sulbactam sodium I.P./cefoperazone sodium I.P. combination is available as a dry powder for reconstitution in a 1:1 and 1:2 ratio in terms of free Sulbactam /Cefoperazone Sulbactam sodium I.P. is a derivative of the basic penicillin nucleus. It is an irreversible beta-lactamase inhibitor for parenteral use only. Chemically it is sodium penicillinate sulfone. It contains 92 mg sodium (4 mEq) per gram. Sulbactam is an off-white crystalline powder which is highly soluble in water. The molecular weight is 255.22. Cefoperazone sodium I.P. is a semisynthetic broad-spectrum cephalosporin antibiotic for parenteral use only. It contains 34 mg sodium (1.5 mEq) per gram. Cefoperazone is a white crystalline powder which is freely soluble in water. The molecular weight is 667.65. List of Excipients None 3. DOSAGE FORM AND STRENGTH Vials of the 1:1 product contain the equivalent of 500 mg + 500 mg and 1000 mg + 1000 mg of sulbactam and cefoperazone, respectively. Vials of the 1:2 product contain the equivalent of 500 mg + 1000 mg and 1000 mg + 2000 mg of sulbactam and cefoperazone, respectively. Trademark Proprietor: Pfizer Products Inc., USA Licensed User: Pfizer Limited, India. MAGNEX/MAGNEX FORTE Injection Page 1 of 18 LPDMGX042020 PfLEET number : 2019-0048457 4. CLINICAL PARTICULARS 4.1 Therapeutic Indications -

ESCMID Online Lecture Library © by Author ESCMID Online Lecture Library
Australasian Society for Infectious Diseases Clinical Research Network Treatment of ESBL Producers David© by Paterson author Brisbane, Australia ESCMID Online Lecture Library © by author ESCMID Online Lecture Library Australasian Society for Infectious Diseases Clinical Research Network © by author ESCMID Online Lecture Library Australasian Society for Infectious Diseases Clinical Research Network © by author ESCMID Online Lecture Library Australasian Society for Infectious Diseases Clinical Research Network © by author ESCMID Online Lecture Library Australasian Society for Infectious Diseases Clinical Research Network © by author ESCMID Online Lecture Library Australasian Society for Infectious Diseases Clinical Research Network © by author ESCMID Online Lecture Library Australasian Society for Infectious Diseases Clinical Research Network © by author ESCMID Online Lecture Library Australasian Society for Infectious Diseases Clinical Research Network © by author ESCMID Online Lecture Library Australasian Society for Infectious Diseases Clinical Research Network Effects of ESBL producers on outcome • Meta-analysis of 16 studies – Increased mortality in patients with bacteremia due to ESBL producing bacteria (RR 1.85; 95% Cis 1.39-2.47) – Increased incidence in delay of effective treatment (RR ©5.56; by 95% author Cis 2.9-10.5) • No increase in mortality for UTI ESCMID Online Lecture Library [Schwaber & Carmeli 2007; Hyle 2005] Australasian Society for Infectious Diseases Clinical Research Network © by author ESCMID Online Lecture Library -

Critically Important Antimicrobials for Human Medicine – 5Th Revision. Geneva
WHO Advisory Group on Integrated Surveillance of Antimicrobial Resistance (AGISAR) Critically Important Antimicrobials for Human Medicine 5th Revision 2016 Ranking of medically important antimicrobials for risk management of antimicrobial resistance due to non-human use Critically important antimicrobials for human medicine – 5th rev. ISBN 978-92-4-151222-0 © World Health Organization 2017, Updated in June 2017 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial- ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/ igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition”. Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. Suggested citation. Critically important antimicrobials for human medicine – 5th rev. Geneva: World Health Organization; 2017. Licence: CC BY-NC-SA 3.0 IGO.