TR-103: Fenthion
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Review of the Mammalian Toxicology
REVIEW OF THE MAMMALIAN TOXICOLOGY AND METABOLISM/TOXICOKINETICS OF FENTHION The APVMA Review of Fenthion © Australian Pesticides and Veterinary Medicines Authority 2012 ISBN 978-0-9873591-5-5 (electronic) This document was originally published in 2005 as part of the preliminary review findings report for Part 1 of the fenthion review. It was subsequently updated in 2007, and revised in 2008. Ownership of intellectual property rights in this publication Unless otherwise noted, copyright (and any other intellectual property rights, if any) in this publication is owned by the Australian Pesticides and Veterinary Medicines Authority (APVMA). Creative Commons licence With the exception of the Coat of Arms, this publication is licensed under a Creative Commons Attribution 3.0 Australia Licence. This is a standard form agreement that allows you to copy, distribute, transmit and adapt this publication provided that you attribute the work. A summary of the licence terms is available from www.creativecommons.org/licenses/by/3.0/au/deed.en. The full licence terms are available from www.creativecommons.org/licenses/by/3.0/au/legalcode. The APVMA’s preference is that you attribute this publication (and any approved material sourced from it) using the following wording: Source: Licensed from the Australian Pesticides and Veterinary Medicines Authority (APVMA) under a Creative Commons Attribution 3.0 Australia Licence. This report was prepared for the APVMA by the Department of Health and Aging Office of Chemical Safety. In referencing this document the Department of Health and Aging Office of Chemical Safety should be cited as the author and the Australian Pesticides and Veterinary Medicines Authority as the publisher and copyright owner. -

Background Information for Chlorpyrifos
64 Appendix A: Background Information for Chlorpyrifos Chlorpyrifos is an organophosphorus insecticide. It belongs to the phosphorothioate (also called phosphorothionate) group, composed of organophosphorus compounds that contain the P=S substructure. Other organophosphorus insecticides in this group include diazinon, methyl parathion, parathion, fenthion, and fenitrothion. These compounds require metabolic activation to their oxon analogs (compounds in which the =S is replaced by =O) for anticholinesterase activity. The structures of chlorpyrifos and its toxic metabolite, chlorpyrifos oxon, are provided in Appendix D. As of 2000, chlorpyrifos was one of the most widely used organophosphorus insecticides in the United States, for both agricultural and residential purposes. Registered uses included food crops, turf and ornamental plants, indoor pest control (including crack and crevice treatment), termite control, mosquito control, and pet collars. It was registered for use in a wide variety of buildings, including residential, commercial, schools, daycare centers, restaurants, hospitals, hotels, and food manufacturing plants (EPA 2000c). Many uses of chlorpyrifos are being phased out (see Section A.4). A study of urinary pesticide metabolites during the third trimester in 386 pregnant women from East Harlem indicated that exposure to chlorpyrifos was prevalent (42% of the women had detectable levels of the chlorpyrifos metabolite), was higher than the median in NHANES III, did not show seasonal variation, and did not change during the -

Qsar Analysis of the Chemical Hydrolysis of Organophosphorus Pesticides in Natural Waters
QSAR ANALYSIS OF THE CHEMICAL HYDROLYSIS OF ORGANOPHOSPHORUS PESTICIDES IN NATURAL WATERS. by Kenneth K. Tanji Principal Investigator and Jonathan 1. Sullivan Graduate Research Assistant Department of Land, Air and Water Resources University of California, Davis Technical Completion Report Project Number W-843 August, 1995 University of California Water Resource Center The research leading to this report was supported by the University of California Water Resource Center as part of Water Resource Center Project W-843. Table of Contents Page Abstract 2 Problem and Research Objectives 3 Introduction 5 Theoretical Background 6 QSAR Methodology 7 Molecular Connectivity Theory 8 Organophosphorus Pesticides 12 Experimental Determination of Rates 15 Results and Discussion 17 Principal Findings and Significance 19 References 34 List of Tables Page Table 1. Statistical relationship between OP pesticides and first-order MC/'s. 30 Table 2. Inherent conditions of waters used in experimental work. 16 Table 3. Estimated half-lives for organophosphorus esters derived from model. 31 Table 4. Half-lives and first-order MCI' sfor model calibration data set. 31 Table 5. Experimental kinetic data for validation set compounds, Sacramento. 33 List of Figures Page Figure 1. Essential Features OfQSAR Modeling Methodology. 21 Figure 2. Regression plot for In hydrolysis rate vs. 1st order MCl' s. 22 Figure 3. a 3-D molecular model, a line-segment model and a graphical model. 23 Figure 4. Molecular connectivity index suborders. 24 Figure 5. Chlorpyrifos and its fourteen fourth order path/cluster fragments. 25 Figure 6. Abridged MClndex output. 26 Figure 7. Parent acids of most common organophosphorus pesticides. 12 Figure 8. -

Environmental Health Criteria 63 ORGANOPHOSPHORUS
Environmental Health Criteria 63 ORGANOPHOSPHORUS INSECTICIDES: A GENERAL INTRODUCTION Please note that the layout and pagination of this web version are not identical with the printed version. Organophophorus insecticides: a general introduction (EHC 63, 1986) INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY ENVIRONMENTAL HEALTH CRITERIA 63 ORGANOPHOSPHORUS INSECTICIDES: A GENERAL INTRODUCTION This report contains the collective views of an international group of experts and does not necessarily represent the decisions or the stated policy of the United Nations Environment Programme, the International Labour Organisation, or the World Health Organization. Published under the joint sponsorship of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization World Health Orgnization Geneva, 1986 The International Programme on Chemical Safety (IPCS) is a joint venture of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization. The main objective of the IPCS is to carry out and disseminate evaluations of the effects of chemicals on human health and the quality of the environment. Supporting activities include the development of epidemiological, experimental laboratory, and risk-assessment methods that could produce internationally comparable results, and the development of manpower in the field of toxicology. Other activities carried out by the IPCS include the development of know-how for coping with chemical accidents, coordination -

Carbaryl Human Health and Ecological Risk Assessment Revised Final Report
SERA TR-052-01-05a Carbaryl Human Health and Ecological Risk Assessment Revised Final Report Submitted to: Paul Mistretta, COR USDA/Forest Service, Southern Region 1720 Peachtree RD, NW Atlanta, Georgia 30309 USDA Forest Service Contract: AG-3187-C-06-0010 USDA Forest Order Number: AG-43ZP-D-06-0009 SERA Internal Task No. 52-01 Submitted by: Patrick R. Durkin and Cynthia King Syracuse Environmental Research Associates, Inc. 5100 Highbridge St., 42C Fayetteville, New York 13066-0950 Fax: (315) 637-0445 E-Mail: [email protected] Home Page: www.sera-inc.com February 9, 2008 Table of Contents Table of Contents............................................................................................................................ ii List of Figures................................................................................................................................. v List of Tables .................................................................................................................................. v List of Attachments........................................................................................................................ vi List of Appendices ......................................................................................................................... vi COMMON UNIT CONVERSIONS AND ABBREVIATIONS................................................... ix CONVERSION OF SCIENTIFIC NOTATION ............................................................................ x EXECUTIVE SUMMARY .......................................................................................................... -
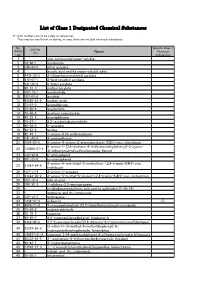
List of Class 1 Designated Chemical Substances
List of Class 1 Designated Chemical Substances *1:CAS numbers are to be solely as references. They may be insufficient or lacking, in case there are multiple chemical substances. No. Specific Class 1 CAS No. (PRTR Chemical (*1) Name Law) Substances 1 - zinc compounds(water-soluble) 2 79-06-1 acrylamide 3 140-88-5 ethyl acrylate 4 - acrylic acid and its water-soluble salts 5 2439-35-2 2-(dimethylamino)ethyl acrylate 6 818-61-1 2-hydroxyethyl acrylate 7 141-32-2 n-butyl acrylate 8 96-33-3 methyl acrylate 9 107-13-1 acrylonitrile 10 107-02-8 acrolein 11 26628-22-8 sodium azide 12 75-07-0 acetaldehyde 13 75-05-8 acetonitrile 14 75-86-5 acetone cyanohydrin 15 83-32-9 acenaphthene 16 78-67-1 2,2'-azobisisobutyronitrile 17 90-04-0 o-anisidine 18 62-53-3 aniline 19 82-45-1 1-amino-9,10-anthraquinone 20 141-43-5 2-aminoethanol 21 1698-60-8 5-amino-4-chloro-2-phenylpyridazin-3(2H)-one; chloridazon 5-amino-1-[2,6-dichloro-4-(trifluoromethyl)phenyl]-3-cyano- 22 120068-37-3 4[(trifluoromethyl)sulfinyl]pyrazole; fipronil 23 123-30-8 p-aminophenol 24 591-27-5 m-aminophenol 4-amino-6-tert-butyl-3-methylthio-1,2,4-triazin-5(4H)-one; 25 21087-64-9 metribuzin 26 107-11-9 3-amino-1-propene 27 41394-05-2 4-amino-3-methyl-6-phenyl-1,2,4-triazin-5(4H)-one; metamitron 28 107-18-6 allyl alcohol 29 106-92-3 1-allyloxy-2,3-epoxypropane 30 - n-alkylbenzenesulfonic acid and its salts(alkyl C=10-14) 31 - antimony and its compounds 32 120-12-7 anthracene 33 1332-21-4 asbestos ○ 34 4098-71-9 3-isocyanatomethyl-3,5,5-trimethylcyclohexyl isocyanate 35 78-84-2 isobutyraldehyde -

Pesticide Resistance of Agricultural Pests in Japan
Pesticide Resistance of Agricultural Pests in Japan By TOSHIKAZU IWATA Chief, 3rd Laboratory of Insect Control Investigation, Department of Phytopathology and Entomology, National Institute of Agricultural Sciences A quarter century has passed since DDT other organophosphorus chemicals such as was firstly applied to control agricultural in methyl paration. As the result the parathion sect pests. The amount of synthesized organic resistant borer was prevented from spreading insecticides applied in fields increased rapidly by changing insecticide to be applied for the since 1952 and the rate of increase was about control. 10 per cent a year. In 1960 an evident report Control of the rice stem borer by BHC dust was made on insecticide resistance of insects, was established in about 1950, and application so 10 years have passed following the first of fine dust or granular formulation of BHC appearance of insecticide-resistant insects. to paddy fields became very popular in 1961 Today, pest species developed or suspected of or 1962. In 1964, declination of susceptibility resistance totals 11 species of insects and 6 of the borers to BHC was found in Kagawa species of mites as shown in Table 1. The Prefecture. After that, similar phenomena author reviews the circumstances of occur were reported in some other prefectures. The rence and some results of studies on major amount of BHC applied to paddy fields has instances of resistant pests. been decreased for its residue in rice plants. Consequently, BHC resistance of rice stem Rice stem borer, Chilo suppressalis borer may not become a serious problem in Walker future. Recently, declination of susceptibility of The rice stem borer is one of the most im the borer to fenitrothion is suspected in a portant pests of rice plants in Japan. -

NMP-Free Formulations of Neonicotinoids
(19) & (11) EP 2 266 400 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: (51) Int Cl.: 29.12.2010 Bulletin 2010/52 A01N 43/40 (2006.01) A01N 43/86 (2006.01) A01N 47/40 (2006.01) A01N 51/00 (2006.01) (2006.01) (2006.01) (21) Application number: 09305544.0 A01P 7/00 A01N 25/02 (22) Date of filing: 15.06.2009 (84) Designated Contracting States: (72) Inventors: AT BE BG CH CY CZ DE DK EE ES FI FR GB GR • Gasse, Jean-Jacques HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL 27600 Saint-Aubin-Sur-Gaillon (FR) PT RO SE SI SK TR • Duchamp, Guillaume Designated Extension States: 92230 Gennevilliers (FR) AL BA RS • Cantero, Maria 92230 Gennevilliers (FR) (71) Applicant: NUFARM 92233 Gennevelliers (FR) (74) Representative: Cabinet Plasseraud 52, rue de la Victoire 75440 Paris Cedex 09 (FR) (54) NMP-free formulations of neonicotinoids (57) The invention relates to NMP-free liquid formulation comprising at least one nicotinoid and at least one aprotic polar component selected from the group comprising the compounds of formula I, II or III below, and mixtures thereof, wherein R1 and R2 independently represent H or an alkyl group having less than 5 carbons, preferably a methyl group, and n represents an integer ranging from 0 to 5, and to their applications. EP 2 266 400 A1 Printed by Jouve, 75001 PARIS (FR) EP 2 266 400 A1 Description Technical Field of the invention 5 [0001] The invention relates to novel liquid formulations of neonicotinoids and to their use for treating plants, for protecting plants from pests and/or for controlling pests infestation. -

ORGANOPHOSPHORUS PESTICIDE RESIDUES in GREEK VIRGIN OLIVE OIL: LEVELS, DIETARY INTAKE and RISK ASSESSMENT Charoula Tsoutsi, Ioannis Konstantinou, Dimitra Hela
ORGANOPHOSPHORUS PESTICIDE RESIDUES IN GREEK VIRGIN OLIVE OIL: LEVELS, DIETARY INTAKE AND RISK ASSESSMENT Charoula Tsoutsi, Ioannis Konstantinou, Dimitra Hela To cite this version: Charoula Tsoutsi, Ioannis Konstantinou, Dimitra Hela. ORGANOPHOSPHORUS PESTI- CIDE RESIDUES IN GREEK VIRGIN OLIVE OIL: LEVELS, DIETARY INTAKE AND RISK ASSESSMENT. Food Additives and Contaminants, 2008, 25 (10), pp.1225-1236. 10.1080/02652030802130025. hal-00577398 HAL Id: hal-00577398 https://hal.archives-ouvertes.fr/hal-00577398 Submitted on 17 Mar 2011 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Food Additives and Contaminants For Peer Review Only ORGANOPHOSPHORUS PESTICIDE RESIDUES IN GREEK VIRGIN OLIVE OIL: LEVELS, DIETARY INTAKE AND RISK ASSESSMENT Journal: Food Additives and Contaminants Manuscript ID: TFAC-2007-433.R1 Manuscript Type: Original Research Paper Date Submitted by the 07-Apr-2008 Author: Complete List of Authors: Tsoutsi, Charoula; University of Ioannina, Department of Chemistry KONSTANTINOU, IOANNIS; UNIVERSITY OF IOANNINA, ENVIRONMENTAL AND NATURAL RESOURCES MANAGEMENT Hela, Dimitra; University of Ioannina, School of Natural Resources and Enterprises Management Chromatographic analysis, Clean-up - SPME, Exposure assessment, Methods/Techniques: Risk assessment Additives/Contaminants: Pesticides - organophosphorous Food Types: Olive oil http://mc.manuscriptcentral.com/tfac Email: [email protected] Page 1 of 39 Food Additives and Contaminants 1 2 3 4 Organophosphorus pesticide residues in Greek virgin olive oil: levels, 5 6 dietary intake and risk assessment 7 8 9 10 11 C. -

Interaction Profile for Mixture of Insecticides: Pyrethroids
44 5. References Abdel-Rahman A, Dechkovskaia AM, Goldstein LB, et al. 2004. Neurological deficits induced by malathion, DEET, and permethrin, alone or in combination in adult rats. J Toxicol Environ Health A 67(4):331-356. ATSDR. 1992. Public health assessment guidance manual. Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service, Agency for Toxic Substances and Disease Registry. ATSDR. 2003a. Toxicological profile for pyrethrins and pyrethroids. Agency for Toxic Substances and Disease Registry. http://www.atsdr.cdc.gov/ToxProfiles/tp155.pdf. March 29, 2013. ATSDR. 2004a. Guidance manual for the assessment of joint toxic action of chemical mixtures. Atlanta, GA: Agency for Toxic Substances and Disease Registry. http://www.atsdr.cdc.gov/interactionprofiles/IP-ga/ipga.pdf. April 2, 2013. ATSDR. 2004b. Interaction profile for benzene, toluene, ethylbenzene, and xylenes (BTEX). Agency for Toxic Substances and Disease Registry. http://www.atsdr.cdc.gov/interactionprofiles/IP- btex/ip05.pdf. March 29, 2013. Belden JB, Lydy MJ. 2006. Joint toxicity of chlorpyrifos and esfenvalerate to fathead minnows and midge larvae. Environ Toxicol Chem 25(2):623-629. Bosgra S, van Eijkeren JC, van der Schans MJ, et al. 2009. Toxicodynamic analysis of the inhibition of isolated human acetylcholinesterase by combinations of methamidophos and methomyl in vitro. Toxicol Appl Pharmacol 236(1):1-8. Burr SA, Ray DE. 2004. Structure-activity and interaction effects of 14 different pyrethroids on voltage- gated chloride ion channels. Toxicol Sci 77(2):341-346. Cao Z, Shafer TJ, Crofton KM, et al. 2011. Additivity of pyrethroid actions on sodium influx in cerebrocortical neurons in primary culture. -

Toxicity of DDT, Dieldrin, Malathion and Fenthion to Rhodnius Prolixus in the Laboratory * by IRVING Fox and ILEANA G
974 NOTES Toxicity of DDT, Dieldrin, Malathion and Fenthion to Rhodnius prolixus in the Laboratory * by IRVING Fox and ILEANA G. BAYONA, School of Tropical Medicine, School of Medicine, University of Puerto Rico, San Juan, Puerto Rico, and HELDA ISABEL OROZCO, Facultad de Medicina, Universidad de Antioquia, Medellin, Colombia Extensive published data on Rhodnius prolixus 11 first-stage and second-stage nymphs brought to cover its distribution and epidemiological role in the School of Tropical Medicine, San Juan, Puerto Chagas' disease in Venezuela," extradomiciliary Rico, by one of us (H.I.O.) on 18 May 1962. These habits,b resistance to practical chemical control specimens originated from a colony in the Facultad efforts,c insect parasites,d life-cycle in the labor- de Medicina, Universidad de Antioquia, Medellin, atory,e, f, ' and effects of radiation on reproduc- Colombia. Concerning this colony, Dr Marcos tion.h, In marked contrast there is a lack of Restrepo I. wrote as follows, in a letter dated toxicological knowledge as derived from laboratory 18 January 1966: tests developed by Busvine & LienJ and by the The strain of Rhodnius prolixus we have in our depart- WHO Expert Committee on Insecticides." It is ment was provided out of one established in BogotA important to obtain this kind of information about from specimens found in various parts of the country, different strains of this serious pest because some especially in the Western Llanos of Colombia and the experts, in their master plans for the control of Department of North Santander. The colony has been Chagas' disease, have contemplated the widespread established for some years (more than 10). -

Federal Register/Vol. 67, No. 147/Wednesday, July 31, 2002/Rules
49606 Federal Register / Vol. 67, No. 147 / Wednesday, July 31, 2002 / Rules and Regulations burden under the provisions of the pesticides acephate, amitraz, carbaryl, I. General Information Paperwork Reduction Act of 1995 (44 chlorpyrifos, cryolite, disulfoton, A. Does this Action Apply to Me? U.S.C. 3501 et seq.). ethalfluralin, ethion, ethoprop, fenthion, The Congressional Review Act, 5 fluvalinate, methamidophos, You may be affected by this action if U.S.C. 801 et seq., as added by the Small metribuzin, oxamyl, phorate, phosalone, you are an agricultural producer, food Business Regulatory Enforcement phosmet, pirimiphos-methyl, manufacturer, or pesticide Fairness Act of 1996, generally provides profenofos, propiconazole, manufacturer. Potentially affected that before a rule may take effect, the tetrachlorvinphos, thiram, and tribufos categories and entities may include, but agency promulgating the rule must because these specific tolerances are are not limited to: submit a rule report, which includes a either no longer needed or are copy of the rule, to each House of the Examples of poten- associated with food uses that are no Categories NAICS tially affected enti- Congress and to the Comptroller General longer registered in the United States. codes ties of the United States. EPA will submit a The regulatory actions in this document report containing this rule and other are part of the Agency’s reregistration Industry 111 Crop production required information to the U.S. Senate, program under the Federal Insecticide, 112 Animal production the U.S. House of Representatives, and Fungicide, and Rodenticide Act 311 Food manufacturing the Comptroller General of the United (FIFRA), and the tolerance reassessment 32532 Pesticide manufac- turing States prior to publication of the rule in requirements of the Federal Food, Drug, the Federal Register.