Mining Diversity of the Natural Biorefinery Housed Within Tipula Abdominalis Larvae for Use in an Industrial Biorefinery For
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Durham E-Theses
Durham E-Theses The feeding ecology of certain larvae in the genus tipula (Tipulidae, Diptera), with special reference to their utilisation of Bryophytes Todd, Catherine Mary How to cite: Todd, Catherine Mary (1993) The feeding ecology of certain larvae in the genus tipula (Tipulidae, Diptera), with special reference to their utilisation of Bryophytes, Durham theses, Durham University. Available at Durham E-Theses Online: http://etheses.dur.ac.uk/5699/ Use policy The full-text may be used and/or reproduced, and given to third parties in any format or medium, without prior permission or charge, for personal research or study, educational, or not-for-prot purposes provided that: • a full bibliographic reference is made to the original source • a link is made to the metadata record in Durham E-Theses • the full-text is not changed in any way The full-text must not be sold in any format or medium without the formal permission of the copyright holders. Please consult the full Durham E-Theses policy for further details. Academic Support Oce, Durham University, University Oce, Old Elvet, Durham DH1 3HP e-mail: [email protected] Tel: +44 0191 334 6107 http://etheses.dur.ac.uk 2 THE FEEDING ECOLOGY OF CERTAIN LARVAE IN THE GENUS TIPULA (TIPULIDAE, DIPTERA), WITH SPECIAL REFERENCE TO THEIR UTILISATION OF BRYOPHYTES Catherine Mary Todd B.Sc. (London), M.Sc. (Durham) The copyright of this thesis rests with the author. No quotation from it should be published without his prior written consent and information derived from it should be acknowledged. A thesis presented in candidature for the degree of Doctor of Philosophy in the University of Durham, 1993 FEB t99^ Abstract Bryophytes are rarely used as a food source by any animal species, but the genus Tipula (Diptera, Tipulidae) contains some of the few insect species able to feed, and complete their life-cycle, on bryophytes. -

Analysis of Cellulolytic and Hemicellulolytic Enzyme Activity
Insect Science (2010) 17, 291–302, DOI 10.1111/j.1744-7917.2010.01346.x ORIGINAL ARTICLE Analysis of cellulolytic and hemicellulolytic enzyme activity within the Tipula abdominalis (Diptera: Tipulidae) larval gut and characterization of Crocebacterium ilecola gen. nov., sp. nov., isolated from the Tipula abdominalis larval hindgut Theresa E. Rogers and Joy Doran-Peterson Microbiology Department, University of Georgia, Athens, Georgia, USA Abstract In forested stream ecosystems of the north and eastern United States, larvae of the aquatic crane fly Tipula abdominalis are important shredders of leaf litter detritus. T. abdominalis larvae harbor a dense and diverse microbial community in their hindgut that may aide in the degradation of lignocellulose. In this study, the activities of cellulolytic and hemicellulolytic enzymes were demonstrated from hindgut extracts and from bacterial isolates using model sugar substrates. One of the bacterial isolates was further characterized as a member of the family Microbacteriaceae. Taxonomic position of the isolate within this family was determined by a polyphasic approach, as is commonly employed for the separation of genera within the family Microbacteriaceae. The bacterial isolate is Gram- type positive, motile, non-sporulating, and rod-shaped. The G + C content of the DNA is 64.9 mol%. The cell wall contains B2γ type peptidoglycan, D- and L-diaminobutyric acid as the diamino acid, and rhamnose as the predominant sugar. The predominant fatty acids are 12-methyltetradecanoic acid (ai-C15:0) and 14-methylhexadecanoic acid (ai-C17:0). The isolate forms a distinct lineage within the family Microbacteriaceae, as determined by 16S rRNA sequence analysis. We propose the name Crocebacterium ilecola gen. -

Diverse Allochthonous Resource Quality Effects on Headwater Stream Communities Through Insect-Microbe Interactions
DIVERSE ALLOCHTHONOUS RESOURCE QUALITY EFFECTS ON HEADWATER STREAM COMMUNITIES THROUGH INSECT-MICROBE INTERACTIONS By Courtney Larson A DISSERTATION Submitted to Michigan State University in partial fulfillment of the requirements for the degree of Entomology—Doctor of Philosophy Ecology, Evolutionary Biology and Behavior—Dual Major 2020 ABSTRACT DIVERSE ALLOCHTHONOUS RESOURCE QUALITY EFFECTS ON HEADWATER STREAM COMMUNITIES THROUGH INSECT-MICROBE INTERACTIONS By Courtney Larson Freshwater resources are vital to environmental sustainability and human health; yet, they are inundated by multiple stressors, leaving aquatic communities to face unknown consequences. Headwater streams are highly reliant on allochthonous sources of energy. Riparian trees shade the stream, limiting primary production, causing macroinvertebrates to consume an alternative food source. Traditionally, leaf litter fallen from riparian trees is the primary allochthonous resource, but other sources, such as salmon carrion associated with annual salmon runs, may also be important. An alteration in the quantity or quality of these sources may have far reaching effects not only on the organisms that directly consume the allochthonous resource (shredders), but also on other functional feeding groups. Allochthonous resources directly and indirectly change stream microbial communities, which are used by consumers with potential changes to their life histories and behavior traits. The objective of my research was to determine the influence allochthonous resources have on stream -

Bacterial Communities Associated with the Hindgut of Tipula Abdominalis Larvae
BACTERIAL COMMUNITIES ASSOCIATED WITH THE HINDGUT OF TIPULA ABDOMINALIS LARVAE (DIPTERA: TIPULIDAE), A NATURAL BIOREFINERY by DANA M. COOK (Under the Direction of Joy Doran Peterson) ABSTRACT Insects are the largest taxonomic group of animals on earth. Although a few thorough studies have shown insect guts host high microbial diversity, many insect-microbe associations have not been investigated. Tipula abdominalis is an aquatic crane fly ubiquitous in riparian environments. T. abdominalis larvae are shredders, a functional feeding group of insects that consume coarse particulate organic matter, primarily leaf litter. In small stream ecosystems, leaf litter comprises the majority of carbon and energy inputs; however, many organisms are unable to degrade this lignocellulosic material. By converting lignocellulose into a form that other organisms can use, T. abdominalis larvae influence the bioavailability of carbon and energy within the ecosystem. Evidence suggests that the bacterial community associated with the T. abdominalis larval hindgut facilitates the digestion of its recalcitrant lignocellulosic diet. Such lignocellulose-ecosystem-insect-microbiota interactions provide a model natural biorefinery and have become of special interest recently for the application of microbial conversion of lignocellulose to biofuels, including ethanol, butanol, and hydrogen. Bacterial isolates from the T. abdominalis larval hindgut were characterized, and many had enzymatic activity against plant polymer model substrates. Several isolates had low 16S rRNA gene sequence similarity to previous described bacteria, including the proposed novel genus Klugiella xanthotipulae gen. nov., sp. nov. Clone libraries of the 16S rRNA gene revealed a phylogenetically diverse bacterial community associated with the larval hindgut wall epithelial and lumen material. Clostridia and Bacteroidetes dominated both hindgut wall and lumen, while Betaproteobacteria dominated leaf diet- and cast-associated microbiota. -
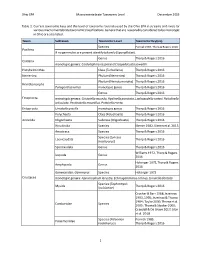
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 1 Table 1. Current Taxonomic Keys and the Level of Taxonomy Routinely U
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Table 1. Current taxonomic keys and the level of taxonomy routinely used by the Ohio EPA in streams and rivers for various macroinvertebrate taxonomic classifications. Genera that are reasonably considered to be monotypic in Ohio are also listed. Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Species Pennak 1989, Thorp & Rogers 2016 Porifera If no gemmules are present identify to family (Spongillidae). Genus Thorp & Rogers 2016 Cnidaria monotypic genera: Cordylophora caspia and Craspedacusta sowerbii Platyhelminthes Class (Turbellaria) Thorp & Rogers 2016 Nemertea Phylum (Nemertea) Thorp & Rogers 2016 Phylum (Nematomorpha) Thorp & Rogers 2016 Nematomorpha Paragordius varius monotypic genus Thorp & Rogers 2016 Genus Thorp & Rogers 2016 Ectoprocta monotypic genera: Cristatella mucedo, Hyalinella punctata, Lophopodella carteri, Paludicella articulata, Pectinatella magnifica, Pottsiella erecta Entoprocta Urnatella gracilis monotypic genus Thorp & Rogers 2016 Polychaeta Class (Polychaeta) Thorp & Rogers 2016 Annelida Oligochaeta Subclass (Oligochaeta) Thorp & Rogers 2016 Hirudinida Species Klemm 1982, Klemm et al. 2015 Anostraca Species Thorp & Rogers 2016 Species (Lynceus Laevicaudata Thorp & Rogers 2016 brachyurus) Spinicaudata Genus Thorp & Rogers 2016 Williams 1972, Thorp & Rogers Isopoda Genus 2016 Holsinger 1972, Thorp & Rogers Amphipoda Genus 2016 Gammaridae: Gammarus Species Holsinger 1972 Crustacea monotypic genera: Apocorophium lacustre, Echinogammarus ischnus, Synurella dentata Species (Taphromysis Mysida Thorp & Rogers 2016 louisianae) Crocker & Barr 1968; Jezerinac 1993, 1995; Jezerinac & Thoma 1984; Taylor 2000; Thoma et al. Cambaridae Species 2005; Thoma & Stocker 2009; Crandall & De Grave 2017; Glon et al. 2018 Species (Palaemon Pennak 1989, Palaemonidae kadiakensis) Thorp & Rogers 2016 1 Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Informal grouping of the Arachnida Hydrachnidia Smith 2001 water mites Genus Morse et al. -

Evolutionary Background Entities at the Cellular and Subcellular Levels in Bodies of Invertebrate Animals
The Journal of Theoretical Fimpology Volume 2, Issue 4: e-20081017-2-4-14 December 28, 2014 www.fimpology.com Evolutionary Background Entities at the Cellular and Subcellular Levels in Bodies of Invertebrate Animals Shu-dong Yin Cory H. E. R. & C. Inc. Burnaby, British Columbia, Canada Email: [email protected] ________________________________________________________________________ Abstract The novel recognition that individual bodies of normal animals are actually inhabited by subcellular viral entities and membrane-enclosed microentities, prokaryotic bacterial and archaeal cells and unicellular eukaryotes such as fungi and protists has been supported by increasing evidences since the emergence of culture-independent approaches. However, how to understand the relationship between animal hosts including human beings and those non-host microentities or microorganisms is challenging our traditional understanding of pathogenic relationship in human medicine and veterinary medicine. In recent novel evolution theories, the relationship between animals and their environments has been deciphered to be the interaction between animals and their environmental evolutionary entities at the same and/or different evolutionary levels;[1-3] and evolutionary entities of the lower evolutionary levels are hypothesized to be the evolutionary background entities of entities at the higher evolutionary levels.[1,2] Therefore, to understand the normal existence of microentities or microorganisms in multicellular animal bodies is becoming the first priority for elucidating the ecological and evolutiological relationships between microorganisms and nonhuman macroorganisms. The evolutionary background entities at the cellular and subcellular levels in bodies of nonhuman vertebrate animals have been summarized recently.[4] In this paper, the author tries to briefly review the evolutionary background entities (EBE) at the cellular and subcellular levels for several selected invertebrate animal species. -

Journal of Cave and Karst Studies
June 2020 Volume 82, Number 2 JOURNAL OF ISSN 1090-6924 A Publication of the National CAVE AND KARST Speleological Society STUDIES DEDICATED TO THE ADVANCEMENT OF SCIENCE, EDUCATION, EXPLORATION, AND CONSERVATION Published By BOARD OF EDITORS The National Speleological Society Anthropology George Crothers http://caves.org/pub/journal University of Kentucky Lexington, KY Office [email protected] 6001 Pulaski Pike NW Huntsville, AL 35810 USA Conservation-Life Sciences Julian J. Lewis & Salisa L. Lewis Tel:256-852-1300 Lewis & Associates, LLC. [email protected] Borden, IN [email protected] Editor-in-Chief Earth Sciences Benjamin Schwartz Malcolm S. Field Texas State University National Center of Environmental San Marcos, TX Assessment (8623P) [email protected] Office of Research and Development U.S. Environmental Protection Agency Leslie A. North 1200 Pennsylvania Avenue NW Western Kentucky University Bowling Green, KY Washington, DC 20460-0001 [email protected] 703-347-8601 Voice 703-347-8692 Fax [email protected] Mario Parise University Aldo Moro Production Editor Bari, Italy [email protected] Scott A. Engel Knoxville, TN Carol Wicks 225-281-3914 Louisiana State University [email protected] Baton Rouge, LA [email protected] Exploration Paul Burger National Park Service Eagle River, Alaska [email protected] Microbiology Kathleen H. Lavoie State University of New York Plattsburgh, NY [email protected] Paleontology Greg McDonald National Park Service Fort Collins, CO The Journal of Cave and Karst Studies , ISSN 1090-6924, CPM [email protected] Number #40065056, is a multi-disciplinary, refereed journal pub- lished four times a year by the National Speleological Society. -

Aquatic Insects: Holometabola – Diptera, Suborder Nematocera
Glime, J. M. 2017. Aquatic Insects: Holometabola – Diptera, Suborder Nematocera. Chapt. 11-13a. In: Glime, J. M. 11-13a-1 Bryophyte Ecology. Volume 2. Bryological Interaction. Ebook sponsored by Michigan Technological University and the International Association of Bryologists. Last updated 19 July 2020 and available at <http://digitalcommons.mtu.edu/bryophyte-ecology2/>. CHAPTER 11-13a AQUATIC INSECTS: HOLOMETABOLA – DIPTERA, SUBORDER NEMATOCERA TABLE OF CONTENTS DIPTERA – Flies .......................................................................................................................................... 11-13a-2 Suborder Nematocera ............................................................................................................................. 11-13a-5 Nymphomyiidae .............................................................................................................................. 11-13a-6 Cylindrotomidae – Long-bodied Craneflies .................................................................................... 11-13a-6 Limoniidae – Limoniid Craneflies .................................................................................................. 11-13a-8 Pediciidae – Hairy-eyed Craneflies ............................................................................................... 11-13a-11 Tipulidae – Craneflies ................................................................................................................... 11-13a-11 Anisopodidae – Wood Gnats, Window Gnats ............................................................................. -

Towards a Networks Based Approach to Biomonitoring Gray, Clare
Towards a networks based approach to biomonitoring Gray, Clare The copyright of this thesis rests with the author and no quotation from it or information derived from it may be published without the prior written consent of the author For additional information about this publication click this link. http://qmro.qmul.ac.uk/xmlui/handle/123456789/12820 Information about this research object was correct at the time of download; we occasionally make corrections to records, please therefore check the published record when citing. For more information contact [email protected] Towards a networks based approach to biomonitoring Clare Gray School of Biological and Chemical Sciences Queen Mary University of London A thesis submitted to the University of London for the degree of Doctor of Philosophy February 2016 Statement of originality I, Clare Gray, confirm that the research included within this thesis is my own work or that where it has been carried out in collaboration with, or supported by others, that this is duly acknowledged below and my contribution indicated. Previously published material is also acknowledged below. I attest that I have exercised reasonable care to ensure that the work is original, and does not to the best of my knowledge break any UK law, infringe any third party’s copyright or other Intellectual Property Right, or contain any confidential material. I accept that the College has the right to use plagiarism detection software to check the electronic version of the thesis. I confirm that this thesis has not been previously submitted for the award of a degree by this or any other university. -
Ohio EPA Macroinvertebrate Taxonomic Level January 2018 1 Table 1. Current Taxonomic Keys and the Level of Taxonomy Routinely U
Ohio EPA Macroinvertebrate Taxonomic Level January 2018 Table 1. Current taxonomic keys and the level of taxonomy routinely used by the Ohio EPA in streams and rivers for various macroinvertebrate taxonomic classifications. Genera that are reasonably considered to be monotypic in Ohio are also listed. Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Species Thorp & Rogers 2016 Porifera If no gemmules are present identify to family (Spongillidae). Genus Thorp & Rogers 2016 Cnidaria monotypic genera: Cordylophora caspia and Craspedacusta sowerbii Platyhelminthes Class (Turbellaria) Thorp & Rogers 2016 Nemertea Phylum (Nemertea) Thorp & Rogers 2016 Phylum (Nematomorpha) Thorp & Rogers 2016 Nematomorpha Paragordius varius monotypic genus Thorp & Rogers 2016 Genus Thorp & Rogers 2016 Ectoprocta monotypic genera: Cristatella mucedo, Hyalinella punctata, Lophopodella carteri, Paludicella articulata, Pectinatella magnifica, Pottsiella erecta Entoprocta Urnatella gracilis monotypic genus Thorp & Rogers 2016 Polychaeta Class (Polychaeta) Thorp & Rogers 2016 Annelida Oligochaeta Subclass (Oligochaeta) Thorp & Rogers 2016 Hirudinida Species Klemm 1982, Klemm et al. 2015 Anostraca Species Thorp & Rogers 2016 Species (Lynceus Laevicaudata Thorp & Rogers 2016 brachyurus) Spinicaudata Genus Thorp & Rogers 2016 Williams 1972, Thorp & Rogers Isopoda Genus 2016 Holsinger 1972, Thorp & Rogers Amphipoda Genus 2016 Gammaridae: Gammarus Species Holsinger 1972 Crustacea monotypic genera: Apocorophium lacustre, Echinogammarus ischnus, Synurella dentata Species (Taphromysis Mysida Thorp & Rogers 2016 louisianae) Jezerinac & Thoma 1984, Jezerinac 1995, Jezerinac 1993, Cambaridae Species Taylor 2000, Thoma et al. 2005, Thoma & Stocker 2009, Crocker & Barr 1968 Species (Palaemon Palaemonidae Thorp & Rogers 2016 kadiakensis) Informal grouping of the Arachnida Hydrachnidia Smith 2001 water mites 1 Ohio EPA Macroinvertebrate Taxonomic Level January 2018 Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Genus Morse et al. -

Rapid Bioassessment Protocols for Use in Streams and Wadeable Rivers
DRAFT REVISION—September 3, 1998 Merrimack Station AR-1164 EPA 841-B-99-002 Rapid Bioassessment Protocols For Use in Streams and Wadeable Rivers: Periphyton, Benthic Macroinvertebrates, and Fish Second Edition http://www.epa.gov/OWOW/monitoring/techmon.html By: Project Officer: Michael T. Barbour Chris Faulkner Jeroen Gerritsen Office of Water Blaine D. Snyder USEPA James B. Stribling 401 M Street, NW DRAFT REVISION—September 3, 1998 Washington, DC 20460 Rapid Bioassessment Protocols for Use in Streams and Rivers 2 DRAFT REVISION—September 3, 1998 NOTICE This document has been reviewed and approved in accordance with U.S. Environmental Protection Agency policy. Mention of trade names or commercial products does not constitute endorsement or recommendation for use. Appropriate Citation: Barbour, M.T., J. Gerritsen, B.D. Snyder, and J.B. Stribling. 1999. Rapid Bioassessment Protocols for Use in Streams and Wadeable Rivers: Periphyton, Benthic Macroinvertebrates and Fish, Second Edition. EPA 841-B-99-002. U.S. Environmental Protection Agency; Office of Water; Washington, D.C. This entire document, including data forms and other appendices, can be downloaded from the website of the USEPA Office of Wetlands, Oceans, and Watersheds: http://www.epa.gov/OWOW/monitoring/techmon.html DRAFT REVISION—September 3, 1998 FOREWORD In December 1986, U.S. EPA's Assistant Administrator for Water initiated a major study of the Agency's surface water monitoring activities. The resulting report, entitled "Surface Water Monitoring: A Framework for Change" (U.S. EPA 1987), emphasizes the restructuring of existing monitoring programs to better address the Agency's current priorities, e.g., toxics, nonpoint source impacts, and documentation of "environmental results." The study also provides specific recommendations on effecting the necessary changes. -

Diptera:Tipulidae)
Ai' ABSTRACT OF THE THESIS OF Thomas Leavitt Dudley for the degree ofMaster of Science 28 September 1982 in Entomology presented on Population and Production Ecology of Lipsothrix spp. Title: (Diptera: Tipulidae) Abstract approved: Redacted for Privacy /Dr. N.H. Ariderson Craneflies of the genus Lipsothrix inhabit decomposing wood in streams. The life cycles and ecological relationships of L. nigrilinea (Doane) and L. fenderi Alexander were investigated to determine how they exploit and respond to the wood habitat and their role in degradationof woody debris in western Oregon. Surveys of the western states and provinces provided data on geographical distribution of the four western species, and a key is given to the five species known from Americanorth of Mexico. The non-adult stages are spent within single logs, primarilyred alder (Alnus rubra), in headwater streams where disturbanceby abrasion is minimized. The two species are sympatric in soft logs in constant contact with water. L. fenderi larvae are found in a wider variety of wood types, including harder wood, other species of wood, andin marginal habitats in which they are more susceptible to desiccation and interactions with the semi-terrestrial community. Habitat selection may relate to the specificity of ovipositing females. Biological and behavioral characteristics of the life stages are described. Both species have a basic biennial life cycle with an ephemeral (1-8 days) adult stage. This long cycle may be possible because because the habitat is relatively predictable and constant. L. fenderi is smaller than L. nigrilinea, and some individuals emerge as annuals, probably in response to an indirect thermal cue in thefall.