Cellulase and Hemicellulase Activity Within the Tipula
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Mining Diversity of the Natural Biorefinery Housed Within Tipula Abdominalis Larvae for Use in an Industrial Biorefinery For
Insect Science (2010) 13, 303–312, DOI 10.1111/j.1744-7917.2010.01343.x ORIGINAL ARTICLE Mining diversity of the natural biorefinery housed within Tipula abdominalis larvae for use in an industrial biorefinery for production of lignocellulosic ethanol Dana M. Cook and Joy Doran-Peterson Microbiology Department, University of Georgia, Athens, Georgia, USA Abstract Although they are the largest taxonomic group of animals, relatively few in- sects have been examined for symbiotic relationships with micro-organisms. However, this is rapidly changing because of the potential for examination of the natural insect– microbe–lignocellulose interactions to provide insights for biofuel technology. Micro- organisms associated with lignocellulose-consuming insects often facilitate the digestion of the recalcitrant plant diet; therefore these microbial communities may be mined for novel lignocellulose-degrading microbes, or for robust and inexpensive biocatalysts necessary for economically feasible biofuel production from lignocellulose. These insect–microbe interactions are influenced by the ecosystem and specific lignocellulose diet, and appre- ciating the whole ecosystem–insect–microbiota–lignocellulose as a natural biorefinery provides a rich and diverse framework from which to design novel industrial processes. One such natural biorefinery, the Tipula abdominalis larvae in riparian ecosystems, is reviewed herein with applications for biochemical processes and overcoming challenges involved in conversion of lignocellulosic biomass to fuel ethanol. From the dense and diverse T. abdominalis larval hindgut microbial community, a cellulolytic bacterial iso- late, 27C64, demonstrated enzymatic activity toward many model plant polymers and also produced a bacterial antibiotic. 27C64 was co-cultured with yeast in fermentation of pine to ethanol, which allowed for a 20% reduction of commercial enzyme. -

Cellulases: Characteristics, Sources, Production, and Applications
8 CELLULASES: CHARACTERISTICS, SOURCES, PRODUCTION, AND APPLICATIONS Xiao-Zhou Zhang and Yi-Heng Percival Zhang 8.1 INTRODUCTION lulases: (1) endoglucanases (EC 3.2.1.4), (2) exogluca- nases, including cellobiohydrolases (CBHs) (EC Cellulose is the most abundant renewable biological 3.2.1.91), and (3) β -glucosidase (BG) (EC 3.2.1.21). To resource and a low-cost energy source based on energy hydrolyze and metabolize insoluble cellulose, the micro- content ($3–4/GJ) ( Lynd et al., 2008 ; Zhang, 2009 ). The organisms must secrete the cellulases (possibly except production of bio-based products and bioenergy from BG) that are either free or cell-surface-bound. Cellu- less costly renewable lignocellulosic materials would lases are increasingly being used for a large variety of bring benefi ts to the local economy, environment, and industrial purposes—in the textile industry, pulp and national energy security ( Zhang, 2008 ). paper industry, and food industry, as well as an additive High costs of cellulases are one of the largest obsta- in detergents and improving digestibility of animal cles for commercialization of biomass biorefi neries feeds. Now cellulases account for a signifi cant share of because a large amount of cellulase is consumed for the world ’ s industrial enzyme market. The growing con- biomass saccharifi cation, for example, ∼ 100 g enzymes cerns about depletion of crude oil and the emissions of per gallon of cellulosic ethanol produced ( Zhang et al., greenhouse gases have motivated the production of bio- 2006b ; Zhu et al., 2009 ). In order to decrease cellulase ethanol from lignocellulose, especially through enzy- use, increase volumetric productivity, and reduce capital matic hydrolysis of lignocelluloses materials—sugar investment, consolidated bioprocessing ( CBP ) has been platform ( Bayer et al., 2007 ; Himmel et al., 1999 ; Zaldi- proposed by integrating cellulase production, cellulose var et al., 2001 ). -

United States Patent (19) 11 Patent Number: 5,981,835 Austin-Phillips Et Al
USOO598.1835A United States Patent (19) 11 Patent Number: 5,981,835 Austin-Phillips et al. (45) Date of Patent: Nov. 9, 1999 54) TRANSGENIC PLANTS AS AN Brown and Atanassov (1985), Role of genetic background in ALTERNATIVE SOURCE OF Somatic embryogenesis in Medicago. Plant Cell Tissue LIGNOCELLULOSC-DEGRADING Organ Culture 4:107-114. ENZYMES Carrer et al. (1993), Kanamycin resistance as a Selectable marker for plastid transformation in tobacco. Mol. Gen. 75 Inventors: Sandra Austin-Phillips; Richard R. Genet. 241:49-56. Burgess, both of Madison; Thomas L. Castillo et al. (1994), Rapid production of fertile transgenic German, Hollandale; Thomas plants of Rye. Bio/Technology 12:1366–1371. Ziegelhoffer, Madison, all of Wis. Comai et al. (1990), Novel and useful properties of a chimeric plant promoter combining CaMV 35S and MAS 73 Assignee: Wisconsin Alumni Research elements. Plant Mol. Biol. 15:373-381. Foundation, Madison, Wis. Coughlan, M.P. (1988), Staining Techniques for the Detec tion of the Individual Components of Cellulolytic Enzyme 21 Appl. No.: 08/883,495 Systems. Methods in Enzymology 160:135-144. de Castro Silva Filho et al. (1996), Mitochondrial and 22 Filed: Jun. 26, 1997 chloroplast targeting Sequences in tandem modify protein import specificity in plant organelles. Plant Mol. Biol. Related U.S. Application Data 30:769-78O. 60 Provisional application No. 60/028,718, Oct. 17, 1996. Divne et al. (1994), The three-dimensional crystal structure 51 Int. Cl. ............................. C12N 15/82; C12N 5/04; of the catalytic core of cellobiohydrolase I from Tricho AO1H 5/00 derma reesei. Science 265:524-528. -

Durham E-Theses
Durham E-Theses The feeding ecology of certain larvae in the genus tipula (Tipulidae, Diptera), with special reference to their utilisation of Bryophytes Todd, Catherine Mary How to cite: Todd, Catherine Mary (1993) The feeding ecology of certain larvae in the genus tipula (Tipulidae, Diptera), with special reference to their utilisation of Bryophytes, Durham theses, Durham University. Available at Durham E-Theses Online: http://etheses.dur.ac.uk/5699/ Use policy The full-text may be used and/or reproduced, and given to third parties in any format or medium, without prior permission or charge, for personal research or study, educational, or not-for-prot purposes provided that: • a full bibliographic reference is made to the original source • a link is made to the metadata record in Durham E-Theses • the full-text is not changed in any way The full-text must not be sold in any format or medium without the formal permission of the copyright holders. Please consult the full Durham E-Theses policy for further details. Academic Support Oce, Durham University, University Oce, Old Elvet, Durham DH1 3HP e-mail: [email protected] Tel: +44 0191 334 6107 http://etheses.dur.ac.uk 2 THE FEEDING ECOLOGY OF CERTAIN LARVAE IN THE GENUS TIPULA (TIPULIDAE, DIPTERA), WITH SPECIAL REFERENCE TO THEIR UTILISATION OF BRYOPHYTES Catherine Mary Todd B.Sc. (London), M.Sc. (Durham) The copyright of this thesis rests with the author. No quotation from it should be published without his prior written consent and information derived from it should be acknowledged. A thesis presented in candidature for the degree of Doctor of Philosophy in the University of Durham, 1993 FEB t99^ Abstract Bryophytes are rarely used as a food source by any animal species, but the genus Tipula (Diptera, Tipulidae) contains some of the few insect species able to feed, and complete their life-cycle, on bryophytes. -

Cells of Commelina Communis1 Received for Publication April 8, 1987 and in Revised Form June 13, 1987 NINA L
Plant Physiol. (1987) 85, 360-364 0032-0889/87/85/0360/05/$01.00/0 Localization of Carbohydrate Metabolizing Enzymes in Guard Cells of Commelina communis1 Received for publication April 8, 1987 and in revised form June 13, 1987 NINA L. ROBINSON2 AND JACK PREISS*3 Department ofBiochemistry and Biophysics, University ofCalifornia, Davis, California 95616 ABSTRACI leaves. The sucrose is either degraded in the apoplast or in the cytoplasm of the storage cell. Sucrose, or its degradation prod- The lliztion ofenzymes involved in the flow of carbon into and out ucts, can be further metabolized to the triose-P or 3-PGA level. of starch was determined in guard cells of Commelina communis. The These compounds may then move into the amyloplast via the guard cell chloroplasts were separated from the rest of the cellular triose-P/Pi translocator and are converted into starch. However, components by a modification of published microfuge methods. The at present, the presence of the triose-P/Pi translocator in amy- enzymes of interest were then assayed in the supernatant and chloroplast loplasts has not been demonstrated. Assuming that the triose-P/ fractions. The chloroplast yield averaged 75% with 10% cytoplasmic Pi translocator is present, the movement of carbon into starch contamination. The enzymes involved in starch biosynthesis, ADPglucose would be a reversal of the enzymic steps occurring in the cyto- pyrophosphorylase, starch synthase, and branching enzyme, are located plasm with the last several steps resulting in the direct incorpo- exclusively in the chloroplast fraction. The enzymes involved in starch ration of carbon into starch. -

Xylooligosaccharides Production, Quantification, and Characterization
19 Xylooligosaccharides Production, Quantification, and Characterization in Context of Lignocellulosic Biomass Pretreatment Qing Qing1, Hongjia Li2,3,4,Ã, Rajeev Kumar2,4 and Charles E. Wyman2,3,4 1 Pharmaceutical Engineering & Life Science, Changzhou University, Changzhou, China 2 Center for Environmental Research and Technology, University of California, Riverside, USA 3 Department of Chemical and Environmental Engineering, University of California, Riverside, USA 4 BioEnergy Science Center, Oak Ridge, USA 19.1 Introduction 19.1.1 Definition of Oligosaccharides Oligosaccharides, also termed sugar oligomers, refer to short-chain polymers of monosaccharide units con- nected by a and/or b glycosidic bonds. In structure, oligosaccharides represent a class of carbohydrates between polysaccharides and monosaccharides, but the range of degree of polymerization (DP, chain length) spanned by oligosaccharides has not been consistently defined. For example, the Medical Subject Headings (MeSH) database of the US National Library of Medicine defines oligosaccharides as carbohy- drates consisting of 2–10 monosaccharide units; in other literature, sugar polymers with DPs of up to 30–40 have been included as oligosaccharides [1–3]. ÃPresent address: DuPont Industrial Biosciences, Palo Alto, USA Aqueous Pretreatment of Plant Biomass for Biological and Chemical Conversion to Fuels and Chemicals, First Edition. Edited by Charles E. Wyman. Ó 2013 John Wiley & Sons, Ltd. Published 2013 by John Wiley & Sons, Ltd. 392 Aqueous Pretreatment of Plant Biomass for -

Analysis of Cellulolytic and Hemicellulolytic Enzyme Activity
Insect Science (2010) 17, 291–302, DOI 10.1111/j.1744-7917.2010.01346.x ORIGINAL ARTICLE Analysis of cellulolytic and hemicellulolytic enzyme activity within the Tipula abdominalis (Diptera: Tipulidae) larval gut and characterization of Crocebacterium ilecola gen. nov., sp. nov., isolated from the Tipula abdominalis larval hindgut Theresa E. Rogers and Joy Doran-Peterson Microbiology Department, University of Georgia, Athens, Georgia, USA Abstract In forested stream ecosystems of the north and eastern United States, larvae of the aquatic crane fly Tipula abdominalis are important shredders of leaf litter detritus. T. abdominalis larvae harbor a dense and diverse microbial community in their hindgut that may aide in the degradation of lignocellulose. In this study, the activities of cellulolytic and hemicellulolytic enzymes were demonstrated from hindgut extracts and from bacterial isolates using model sugar substrates. One of the bacterial isolates was further characterized as a member of the family Microbacteriaceae. Taxonomic position of the isolate within this family was determined by a polyphasic approach, as is commonly employed for the separation of genera within the family Microbacteriaceae. The bacterial isolate is Gram- type positive, motile, non-sporulating, and rod-shaped. The G + C content of the DNA is 64.9 mol%. The cell wall contains B2γ type peptidoglycan, D- and L-diaminobutyric acid as the diamino acid, and rhamnose as the predominant sugar. The predominant fatty acids are 12-methyltetradecanoic acid (ai-C15:0) and 14-methylhexadecanoic acid (ai-C17:0). The isolate forms a distinct lineage within the family Microbacteriaceae, as determined by 16S rRNA sequence analysis. We propose the name Crocebacterium ilecola gen. -

Diverse Allochthonous Resource Quality Effects on Headwater Stream Communities Through Insect-Microbe Interactions
DIVERSE ALLOCHTHONOUS RESOURCE QUALITY EFFECTS ON HEADWATER STREAM COMMUNITIES THROUGH INSECT-MICROBE INTERACTIONS By Courtney Larson A DISSERTATION Submitted to Michigan State University in partial fulfillment of the requirements for the degree of Entomology—Doctor of Philosophy Ecology, Evolutionary Biology and Behavior—Dual Major 2020 ABSTRACT DIVERSE ALLOCHTHONOUS RESOURCE QUALITY EFFECTS ON HEADWATER STREAM COMMUNITIES THROUGH INSECT-MICROBE INTERACTIONS By Courtney Larson Freshwater resources are vital to environmental sustainability and human health; yet, they are inundated by multiple stressors, leaving aquatic communities to face unknown consequences. Headwater streams are highly reliant on allochthonous sources of energy. Riparian trees shade the stream, limiting primary production, causing macroinvertebrates to consume an alternative food source. Traditionally, leaf litter fallen from riparian trees is the primary allochthonous resource, but other sources, such as salmon carrion associated with annual salmon runs, may also be important. An alteration in the quantity or quality of these sources may have far reaching effects not only on the organisms that directly consume the allochthonous resource (shredders), but also on other functional feeding groups. Allochthonous resources directly and indirectly change stream microbial communities, which are used by consumers with potential changes to their life histories and behavior traits. The objective of my research was to determine the influence allochthonous resources have on stream -

Bacterial Communities Associated with the Hindgut of Tipula Abdominalis Larvae
BACTERIAL COMMUNITIES ASSOCIATED WITH THE HINDGUT OF TIPULA ABDOMINALIS LARVAE (DIPTERA: TIPULIDAE), A NATURAL BIOREFINERY by DANA M. COOK (Under the Direction of Joy Doran Peterson) ABSTRACT Insects are the largest taxonomic group of animals on earth. Although a few thorough studies have shown insect guts host high microbial diversity, many insect-microbe associations have not been investigated. Tipula abdominalis is an aquatic crane fly ubiquitous in riparian environments. T. abdominalis larvae are shredders, a functional feeding group of insects that consume coarse particulate organic matter, primarily leaf litter. In small stream ecosystems, leaf litter comprises the majority of carbon and energy inputs; however, many organisms are unable to degrade this lignocellulosic material. By converting lignocellulose into a form that other organisms can use, T. abdominalis larvae influence the bioavailability of carbon and energy within the ecosystem. Evidence suggests that the bacterial community associated with the T. abdominalis larval hindgut facilitates the digestion of its recalcitrant lignocellulosic diet. Such lignocellulose-ecosystem-insect-microbiota interactions provide a model natural biorefinery and have become of special interest recently for the application of microbial conversion of lignocellulose to biofuels, including ethanol, butanol, and hydrogen. Bacterial isolates from the T. abdominalis larval hindgut were characterized, and many had enzymatic activity against plant polymer model substrates. Several isolates had low 16S rRNA gene sequence similarity to previous described bacteria, including the proposed novel genus Klugiella xanthotipulae gen. nov., sp. nov. Clone libraries of the 16S rRNA gene revealed a phylogenetically diverse bacterial community associated with the larval hindgut wall epithelial and lumen material. Clostridia and Bacteroidetes dominated both hindgut wall and lumen, while Betaproteobacteria dominated leaf diet- and cast-associated microbiota. -
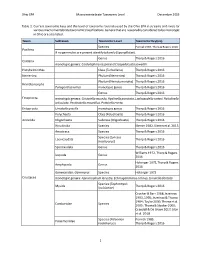
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 1 Table 1. Current Taxonomic Keys and the Level of Taxonomy Routinely U
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Table 1. Current taxonomic keys and the level of taxonomy routinely used by the Ohio EPA in streams and rivers for various macroinvertebrate taxonomic classifications. Genera that are reasonably considered to be monotypic in Ohio are also listed. Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Species Pennak 1989, Thorp & Rogers 2016 Porifera If no gemmules are present identify to family (Spongillidae). Genus Thorp & Rogers 2016 Cnidaria monotypic genera: Cordylophora caspia and Craspedacusta sowerbii Platyhelminthes Class (Turbellaria) Thorp & Rogers 2016 Nemertea Phylum (Nemertea) Thorp & Rogers 2016 Phylum (Nematomorpha) Thorp & Rogers 2016 Nematomorpha Paragordius varius monotypic genus Thorp & Rogers 2016 Genus Thorp & Rogers 2016 Ectoprocta monotypic genera: Cristatella mucedo, Hyalinella punctata, Lophopodella carteri, Paludicella articulata, Pectinatella magnifica, Pottsiella erecta Entoprocta Urnatella gracilis monotypic genus Thorp & Rogers 2016 Polychaeta Class (Polychaeta) Thorp & Rogers 2016 Annelida Oligochaeta Subclass (Oligochaeta) Thorp & Rogers 2016 Hirudinida Species Klemm 1982, Klemm et al. 2015 Anostraca Species Thorp & Rogers 2016 Species (Lynceus Laevicaudata Thorp & Rogers 2016 brachyurus) Spinicaudata Genus Thorp & Rogers 2016 Williams 1972, Thorp & Rogers Isopoda Genus 2016 Holsinger 1972, Thorp & Rogers Amphipoda Genus 2016 Gammaridae: Gammarus Species Holsinger 1972 Crustacea monotypic genera: Apocorophium lacustre, Echinogammarus ischnus, Synurella dentata Species (Taphromysis Mysida Thorp & Rogers 2016 louisianae) Crocker & Barr 1968; Jezerinac 1993, 1995; Jezerinac & Thoma 1984; Taylor 2000; Thoma et al. Cambaridae Species 2005; Thoma & Stocker 2009; Crandall & De Grave 2017; Glon et al. 2018 Species (Palaemon Pennak 1989, Palaemonidae kadiakensis) Thorp & Rogers 2016 1 Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Informal grouping of the Arachnida Hydrachnidia Smith 2001 water mites Genus Morse et al. -

How Liquorilactobacillus Hordei TMW 1.1822 Changes Its Behavior in the Presence of Sucrose in Comparison to Glucose
foods Article Living the Sweet Life: How Liquorilactobacillus hordei TMW 1.1822 Changes Its Behavior in the Presence of Sucrose in Comparison to Glucose Julia Bechtner 1 , Christina Ludwig 2, Michael Kiening 3, Frank Jakob 1 and Rudi F. Vogel 1,* 1 Lehrstuhl für Technische Mikrobiologie, Technische Universität München (TUM), 85354 Freising, Germany; [email protected] (J.B.); [email protected] (F.J.) 2 Bavarian Center for Biomolecular Mass Spectrometry (BayBioMS), 85354 Freising, Germany; [email protected] 3 Lehrstuhl für Genomorientierte Bioinformatik, Technische Universität München (TUM), 85354 Freising, Germany; [email protected] * Correspondence: [email protected] Received: 2 August 2020; Accepted: 17 August 2020; Published: 21 August 2020 Abstract: Liquorilactobacillus (L.) hordei (formerly Lactobacillus hordei) is one of the dominating lactic acid bacteria within the water kefir consortium, being highly adapted to survive in this environment, while producing high molecular weight dextrans from sucrose. In this work, we extensively studied the physiological response of L. hordei TMW 1.1822 to sucrose compared to glucose, applying label-free, quantitative proteomics of cell lysates and exoproteomes. This revealed the differential expression of 53 proteins within cellular proteomes, mostly associated with carbohydrate uptake and metabolism. Supported by growth experiments, this suggests that L. hordei TMW 1.1822 favors fructose over other sugars. The dextransucrase was expressed irrespectively of the present carbon source, while it was significantly more released in the presence of sucrose (log2FC = 3.09), being among the most abundant proteins within exoproteomes of sucrose-treated cells. Still, L. hordei TMW 1.1822 expressed other sucrose active enzymes, predictively competing with the dextransucrase reaction. -

Evolutionary Background Entities at the Cellular and Subcellular Levels in Bodies of Invertebrate Animals
The Journal of Theoretical Fimpology Volume 2, Issue 4: e-20081017-2-4-14 December 28, 2014 www.fimpology.com Evolutionary Background Entities at the Cellular and Subcellular Levels in Bodies of Invertebrate Animals Shu-dong Yin Cory H. E. R. & C. Inc. Burnaby, British Columbia, Canada Email: [email protected] ________________________________________________________________________ Abstract The novel recognition that individual bodies of normal animals are actually inhabited by subcellular viral entities and membrane-enclosed microentities, prokaryotic bacterial and archaeal cells and unicellular eukaryotes such as fungi and protists has been supported by increasing evidences since the emergence of culture-independent approaches. However, how to understand the relationship between animal hosts including human beings and those non-host microentities or microorganisms is challenging our traditional understanding of pathogenic relationship in human medicine and veterinary medicine. In recent novel evolution theories, the relationship between animals and their environments has been deciphered to be the interaction between animals and their environmental evolutionary entities at the same and/or different evolutionary levels;[1-3] and evolutionary entities of the lower evolutionary levels are hypothesized to be the evolutionary background entities of entities at the higher evolutionary levels.[1,2] Therefore, to understand the normal existence of microentities or microorganisms in multicellular animal bodies is becoming the first priority for elucidating the ecological and evolutiological relationships between microorganisms and nonhuman macroorganisms. The evolutionary background entities at the cellular and subcellular levels in bodies of nonhuman vertebrate animals have been summarized recently.[4] In this paper, the author tries to briefly review the evolutionary background entities (EBE) at the cellular and subcellular levels for several selected invertebrate animal species.