[email protected] +1-703-527-3887 (International) Website
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-
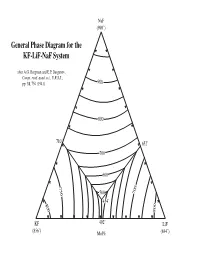
General Phase Diagram for the KF-Lif-Naf System
NaF (990˚) General Phase Diagram for the KF-LiF-NaF System after A.G. Bergman and E.P. Dergunov, Compt. rend. acad. sci., U.R.S.S., pp. 31, 754 (1941). 900 800 710˚ 652˚ 700 600 750 500 700 454˚ 800 800 KF 492˚ LiF (856˚) Mol% (844˚) Sodium Fluoride NaF 950˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 940˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 930˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 920˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 910˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 900˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 890˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 880˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 870˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 860˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 850˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 840˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 830˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 820˚ KF LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 810˚ 810˚ ˚ KF 810 LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 800˚ 800˚ ˚ KF 800 LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 790˚ 790˚ ˚ KF 790 LiF Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 780˚ 780˚ ˚ LiF KF 780 Potassium Fluoride Lithium Fluoride Sodium Fluoride NaF 770˚ 770˚ ˚ LiF KF 770 Potassium Fluoride -

Chemical Storage Cabinets
Storage cabinets Incompatible chemicals must be kept separately to reduce the risk of mixing in case of accidental breakage or response to an emergency within the laboratory. (A starting point is to check the Safety Data Sheet for hazard and storage requirements.) Containers must be kept tightly closed and stored in cabinets which are suitable for the chemical. It is important to check materials which have a shelf life and safely dispose of before the date expires, and generally check the label to ensure it is still suitable and provides the necessary safety information. The condition of the cabinets needs to be checked to ensure that they are providing adequate containment of the materials being stored. Ensure spill trays are used which are capable of holding 110% volume of the largest container being stored in the tray. There may be occasions where the quantities being stored are very small and secondary containment of the chemicals within a cabinet provided would be deemed as providing adequate separation. The table below provides guidance on the type of materials being stored and the type of cabinet which is suitable for that material. This is not a definitive list and does not include ‘Highly Toxic’ materials. Separate guidance is in development. Hazard Group Type of cabinet Safety Sign and suggested Other information Max allowed (where wording applicable) Flammable Liquids Metal flammable storage Flammable liquids must 50 litres of highly flammable cabinet to BS EN 14470- never be stored in a liquid stored in any one E.g. alcohols, toluene, 1:2004, offering minimum refrigerator or freezer laboratory. -

Treating Irrigation Systems with Chlorine 1
Archival copy: for current recommendations see http://edis.ifas.ufl.edu or your local extension office. CIR1039 Treating Irrigation Systems with Chlorine 1 Kati W. Migliaccio, Brian Boman and Gary A. Clark2 Introduction Irrigation systems can become partially or completely clogged from biological growths of bacteria or algae which are often present in surface water and ground water. Bacteria and algae use chemical elements such as nitrogen, phosphorus, sulfur, or iron as nutrient sources to grow and develop (Figures 1 and 2) (Pitts et al., 2003). Thus, irrigation systems that also receive nutrients (such as natural background concentrations in the water or from fertigation) may experience greater rates of clogging. While all irrigation systems should have some Figure 1. Irrigation emitter clogged with algal growth. type of filtration system, this alone cannot effectively Credits: Brian Boman UF/IFAS remove microorganisms. Microorganism growth can result in clogged pipes, fittings, and emission devices of the chlorine chemical must be used to provide an (sprinklers, drippers, spray jets, etc.), decreasing effective water treatment program without damaging water application amounts and reducing application the irrigation system or the agricultural crop. uniformity and efficiency. The results of these are This publication provides a guide for using generally reduced agriculture productivity. A chlorine to treat inhibiting microorganism buildup in chemical method for removing microbial growth is irrigation systems. chlorination. Proper injection methods and amounts 1. This document is CIR1039, one of a series of the Agricultural and Biological Engineering Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida. Original publication date July 1992. -

186 SESSION LAWS [Chap
186 SESSION LAWS [Chap. Sec. 2. This act shall take effect and be in force from and after its passage. Approved April 13, 1925. CHAPTER 187—S. F. No. 984. An act to safeguard the distribution and sale of certain danger- ous caustic or corrosive acids, alkalis, and other substances. Be it enacted by the Legislature of the State of Minnesota: Section 1. Definitions.—That in this act, unless the context or subject-matter otherwise requires, A. The term "dangerous caustic or corrosive substance" means each and all of the acids, alkalis, and substances named below: (a) Hydrochloric acid and any preparation containing free or chemi- cally unneutralized hydrochloric acid (HC1) in a concentration of ten per centum or more; (b) Sulphuric acid and any preparation containing free or chemically unneulralized sulphuric acid (H2SO4) in a concentration of ten per centum or more; (c) Nitric acid or any preparation containing free or chemically unneutralized nitric acid (HNO3) in a concentration of five per centum or more; (d) Carbolic acid (C6H5OH), otherwise known as phenol, and any preparation containing carbolic acid in a concentration of five per centum or more; (e) Oxalic acid and any preparation containing free or chemically unneutralized oxalic acid (H2C2O4) in a con- centration of ten per centum or more; (f) Any salt of oxalic acid and any preparation containing any such salt in a concentration of ten per centum or more; (g) Acetic acid or any preparation con- taining free or chemically unneutralized acetic acid (HC2H3O2) in a concentration of -

N-BUTYL ALCOHOL
Right to Know Hazardous Substance Fact Sheet Common Name: n-BUTYL ALCOHOL Synonyms: Propyl Carbinol; n-Butanol CAS Number: 71-36-3 Chemical Name: 1-Butanol RTK Substance Number: 1330 Date: November 1998 Revision: January 2008 DOT Number: UN 1120 Description and Use EMERGENCY RESPONDERS >>>> SEE BACK PAGE n-Butyl Alcohol is a colorless liquid with a strong, sweet Hazard Summary alcohol odor. It is used as a solvent for fats, waxes, shellacs, Hazard Rating NJDOH NFPA resins, gums, and varnish, in making hydraulic fluids, and in HEALTH - 2 medications for animals. FLAMMABILITY - 3 REACTIVITY - 0 f ODOR THRESHOLD = 1 to 15 ppm FLAMMABLE f Odor thresholds vary greatly. Do not rely on odor alone to POISONOUS GASES ARE PRODUCED IN FIRE determine potentially hazardous exposures. CONTAINERS MAY EXPLODE IN FIRE Hazard Rating Key: 0=minimal; 1=slight; 2=moderate; 3=serious; Reasons for Citation 4=severe f n-Butyl Alcohol is on the Right to Know Hazardous f n-Butyl Alcohol can affect you when inhaled and by Substance List because it is cited by OSHA, ACGIH, DOT, passing through the skin. NIOSH, DEP, IRIS, NFPA and EPA. f Contact can irritate and burn the skin. f This chemical is on the Special Health Hazard Substance f n-Butyl Alcohol can irritate and burn the eyes with possible List. eye damage. f Inhaling n-Butyl Alcohol can irritate the nose, throat and lungs. f Exposure to n-Butyl Alcohol can cause headache, dizziness, nausea and vomiting. SEE GLOSSARY ON PAGE 5. f n-Butyl Alcohol can damage the liver, kidneys, hearing, and sense of balance. -

United States Patent Office Patented July 1, 1969
3,453,337 United States Patent Office Patented July 1, 1969 1. 2 3,453,337 FLUORINATION OF HALOGENATED The presence in the reaction mixture of the two fluo ORGANIC COMPOUNDS rides, or the complex fluoride enables better yields of Royston Henry Bennett and David Walter Cottrell, Ayon highly fluorinated products to be obtained under less mouth, England, assignors to Imperial Smelting Cor severe reaction condions, markedly increases the amount poration (N.S.C.) Limited, London, England, a British of fluorination reagent reacted under otherwise similar company conditions and enables the fluorination reaction to be No brawing. Filed Feb. 19, 1965, Ser. No. 434,128 carried out (for the same yield of product) at a lower Claims priority, application Great Britain, Feb. 26, 1964, temperature with the consequent use of less costly ma 7,932/64 terials and techniques of reactor construction. The pres Int, C. C07c 25/04 10 ence of the fluorides enables the vapor phase reaction U.S. C. 260-650 2 Claims to be carried out (for the same yields) at lower pressure This invention relates to the fluorination of organic than the pressure involved in the reactions using only halogen compounds and more especially to a process the alkali metal fluorides as proposed hitherto. The fur for the production of highly fluorinated aromatic com ther possibility of using a continuous flow apparatus such pounds by the replacement of higher halogen atoms in 15 as a fluidised reactor will be apparent to those familiar halogeno-aromatic compounds by fluorine atoms. with the art. The presence of the two fluorides or com Aromatic halogenocarbons containing carbon and halo plex fluoride enables a lower temperature to be em gen atoms only can be reacted with alkali fluorides ployed than was hitherto believed to be necessary, with under various conditions to give yields of halofluoro a consequent reduction in the extent of thermal degrada aromatic compounds. -

N-Hexyl-Chloroformate-NHEXCF.Pdf
Date of issue: 11. 02. 2004. Date of revision: 15.01.2015. Version: CLP_ C msds-europe.com SAFETY DATA SHEET SECTION 1 : IDENTIFICATION OF THE SUBSTANCE AND OF THE COMPANY/UNDERTAKING 1.1. Product identifier: HEXYL CHLOROFORMATE Synonym: Chloroformic Acid Hexyl Ester CAS number: 6092-54-2 EU number: 228-036-4 Index number: 607-064-00-4 Registration number: Registration number is not available for this substance since this substance or its use is exempted from registration based on Article 2 of REACH regulation, or registration is not necessary due to the annual tonnage band, or the registration is scheduled to a later registration date. 1.2. Relevant identified uses of the substance and uses advised against: Organic synthesis intermediate for industrial use. 1.3. Details of the supplier of the safety data sheet: FRAMOCHEM FRENCH-HUNGARIAN FINE CHEMICALS LTD. 3700 Kazincbarcika, Szerviz str. 5., POB. 504 Telephone: +36 (48) 311-991 Fax: +36 (48) 512-162 1.3.1. Responsible person: - E-mail: [email protected] 1.4. Emergency telephone number: Public Toxicological Health Service (ETTSZ) 1096 Budapest, Nagyvárad tér 2. Tel.: 06 1 476 6464, 06 80 201 199 (0-24 h) SECTION 2: HAZARDS IDENTIFICATION 2.1. Classification of the substance: Classification according to Regulation 1272/2008/EC (CLP): Acute Tox (oral) 3 – H301 Acute Tox. 3 (dermal) – H311 Skin Corr. 1B – H314 Acute Tox. (inhalative) – H331 Warning H statements: H301 – Toxic if swallowed. H311 – Toxic in contact with skin. H314 – Causes severe skin burns and eye damage. H331 – Toxic if inhaled. Classification according to Directive 67/548/EEC: T; Toxic – R 23/24/25 C; Corrosive – R34 R phrases referring to the hazards/risks: FRAMOCHEM FRENCH-HUNGARIAN FINE CHEMICALS LTD. -

July 2014 1/15 ASSESSMENT of the PERFORMANCE
July 2014 ASSESSMENT OF THE PERFORMANCE CHARACTERISTICS OF PROPOSED IN VITRO MEMBRANE BARRIER TEST METHOD FOR SKIN CORROSION This document contains Performance Standards (PS) for determining the validation status (reliability and relevance) of similar and modified skin corrosion test methods that are structurally and mechanistically similar to the in vitro membrane barrier test method in OECD Test Guideline 435 (1), in accordance with the principles of Guidance Document No. 34 (3). These PS include a list of 40 Reference Chemicals by which to evaluate assay performance, the essential test method components by which to evaluate the structural, mechanistic and procedural similarity of a new proposed test method, and the minimum reliability and accuracy values necessary for the test method to be considered comparable to the validated reference method (VRM). INTRODUCTION 1. Test methods proposed for use under this Test Guideline should be evaluated to determine their Field Code Changed reliability and accuracy using substances of known corrosivity and non-corrosivity. When evaluated using the recommended reference chemicals (Table 1), the proposed test methods should have reliability and accuracy that are comparable to that of the validated reference test method (VRM) (2)(Table 2). The reliability and accuracy standards that should be achieved are provided in paragraphs 3 and 4. Non- corrosive and corrosive substances, ranging from strong to weak and representing relevant chemical classes are included so that the reliability and accuracy, i.e., sensitivity, specificity, false negative rates, and false positive rates, of the proposed test method can be compared to that of the validated reference test method (2)(3). -

Aluminum Phosphate
Right to Know Hazardous Substance Fact Sheet Common Name: ALUMINUM PHOSPHATE Synonyms: Aluminum Monophosphate CAS Number: 7784-30-7 Chemical Name: Phosphoric Acid, Aluminum Salt (1:1) RTK Substance Number: 0062 Date: June 1998 Revision: July 2007 DOT Number: UN 1760 Description and Use EMERGENCY RESPONDERS >>>> SEE BACK PAGE Aluminum Phosphate is an odorless, white crystalline solid Hazard Summary which is often used in liquid or gel form. It is used in ceramics, Hazard Rating NJDOH NFPA dental cements, cosmetics, paints, paper and pharmaceuticals. HEALTH 2 - FLAMMABILITY 0 - REACTIVITY 0 - CORROSIVE POISONOUS GASES ARE PRODUCED IN FIRE CONTAINERS MAY EXPLODE IN FIRE DOES NOT BURN Reason for Citation Hazard Rating Key: 0=minimal; 1=slight; 2=moderate; 3=serious; f Aluminum Phosphate is on the Right to Know Hazardous 4=severe Substance List because it is cited by OSHA, ACGIH, DOT f Aluminum Phosphate can affect you when inhaled. and NIOSH. f Contact can irritate and burn the skin and eyes. f This chemical is on the Special Health Hazard Substance f Inhaling Aluminum Phosphate can irritate the nose, throat List. and lungs. SEE GLOSSARY ON PAGE 5. FIRST AID Eye Contact Workplace Exposure Limits f Immediately flush with large amounts of cool water. The following exposure limits are for inorganic Aluminum Continue for at least 15 minutes, occasionally lifting upper compounds (measured as Aluminum): and lower lids. Remove contact lenses, if worn, while rinsing. Immediate medical attention is necessary. OSHA: The legal airborne permissible exposure limit (PEL) is 15 mg/m3 (as total dust), and 5 mg/m3 (as respirable Skin Contact dust) averaged over an 8-hour workshift. -

Guidelines for Using Perchloric Acid
Guidelines for Using Perchloric Acid Perchloric acid (HClO4) is a strong mineral acid. Under some circumstances it may act as an oxidizer and/or present an explosion hazards. These guidelines present information on how to handle and store perchloric acid safely. Please notify EH&S at 8-8182 if you are using perchloric acid in your laboratory. Using Perchloric Acid (< 72%) at Room Temperature At room temperature, perchloric acid up to concentrations of 72% has properties similar to other strong mineral acids. It is a highly corrosive substance and causes severe burns on contact with the eyes, skin, and mucous membranes. When used under these conditions, perchloric acid reacts as a strong non-oxidizing acid. The following precautions should be taken when using perchloric acid under these conditions: · Substitute with less hazardous chemicals when appropriate. Use dilute solutions (< 60%) whenever possible. · Conduct operations involving cold perchloric acid in a properly functioning chemical fume hood with current EH&S certification. If operations are conducted frequently or in large quantities contact EH&S to determine if a specially designed fume hood dedicated to perchloric acid use is required. · Always use impact-resistant chemical goggles, a face shield, neoprene gloves, and a rubber apron when handling perchloric acid. · When using or storing even dilute perchloric acid solutions avoid contact with strong dehydrating agents (concentrated sulfuric acid, anhydrous phosphorous pentoxide, etc.). These chemicals may concentrate the perchloric acid and make it unstable. · Always transfer perchloric acid over a sink or other suitable containment in order to catch any spills and afford a ready means of cleanup and disposal. -

Reaction of Potassium Fluoride with Organic Halogen Compounds. I
Reaction of Potassium Fluoride with Organic Halogen Compounds. I) Reactions of Potassium Fluoride with Organic Halides, Acids, aad Esters in presence ef Dimethyl Formamide and their Pyrolytic Decaboxylation in presence of Potassium Fluoride By You Sun Kim Atomic Energy Research Institute, Korea 有機 할로겐 化合物과 弗化加里의 反應 (第1報) 有機 할라어드, 酸 및 에스테르와 弗化加里의 디메칠 호쁨아마이드 溶蝶系 反應 및 高混■■脫炭酸-熱分解反應 金 裕 *善 (1963. 6. 19 受理) Abstract Reactions between potassium fluride with organic halogen-containning carboxylic acids in dimethyl formamide solvent gave a decarboxylation reaction for the case of fluoro carboxylic acids of the type of CF3 COOH, C3F7COOH, and C2F5COOH, whereas an additional partial fluorination together with dimeri zation reaction occured for the chlorine containning acids of the type of CH2CICOOH, CH3CHCICOOH, CHCI2COOH and o-Cl-CeHi-COOH. The phenyl halides showed no reactivity, but the halides with two electron attracting substituents on the benzene ring gave mainly dimerization reaction. The esters and alcohols gave an usual fluorination reaction. The same reactions in absence of the solvent at the elevated temperature increase the yield of the dimerized product and gave the cyclized product, fluorenone, in case of o-chlorobenzoic acid. It was found that the fluorination usually precede the decarboxylation reaction by checking the stiochemical sequence of reaction. Catalytic influence of potassium fluoride were discussed and the mechanism of the reaction was considered. 耍 約 「디메望호름아마이드」溶媒系에서 有機含할로겐化合物을 弗化加里와 反應시켜 본 結果 CFsCOOH, CsF’COOH, CzFQOOH 와 같은 含弗素有機酸에서는 脫炭酸反應이 일어나며, 含鹽素有機酸, CH2C1COOH. CH3CHC1COOH, CHC12- COOH 및 o-CK사LCOOH 은 一部 弗化反應이 일어 나고 雙合어imerization) 反應이 隨伴된다는 것을 究明하였다. -

C3- Chemical Reactions LINK
Section 1 - Types of Reactions Section 2-Testing for Gases Key Words Phrase Definition Phrase Definition (highlighted) A reaction where energy is given out to the To test for hydrogen use a lit splint, if it surroundings. The surroundings then have more 2a Hydrogen test makes a ‘squeaky pop’ sound then 1a Exothermic energy than it started with. This shows as an hydrogen is present. increase in temperature. To test for oxygen use a glowing splint, 2b Oxygen test LINK A reaction where energy is taken in. The reaction if it reignites oxygen is present. requires more energy than the reactants so it 1b Endothermic Bubble the gas through limewater and Extended absorbs energy from the surroundings. This shows 2c Carbon dioxide test if it turns cloudy then carbon dioxide is reading as a temperature decrease present. The process of burning a fuel in the presence of 1c Combustion oxygen. 2d Limewater Calcium hydroxide solution. It turns Thermal Type of reaction in which a compound breaks down milky in the presence of carbon dioxide LINK 1d decomposition to form two or more substances when it is heated. Exam 1f- products Substances present at the start of a chemical 1e- reactants questions 1e Reactants reaction. C3- Chemical 1f Products A substance formed in a chemical reaction. Reactions LINK When a substance bonds with oxygen it is 1g Oxidation oxidised. Video e.g magnesium + oxygen → magnesium oxide. links A compound made of only hydrogen and carbon. 1h Hydrocarbon These are common as fuels. When a more reactive element displaces a less 1i Displacement reactive element.