Phosphorus Insecticides Against Organophosphorus- Resistant Rice Stem Borers*
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Routine Multipesticide Analysis Orbitrap
Routine Multi-Pesticide Residue Analysis by Orbitrap MS Technology Osama Abu-Nimreh CMD Sales Support Specialist MECEC , Dubai The world leader in serving science Challenges of Pesticide-Residues Analysis • Sample variability (matrix) • Different compound characteristics • Large number of samples • Hundreds of analytes monitored • Low levels controlled • Baby food (MRL for all pesticides = 0.01 mg/kg) • Fast response required 2 Former Pesticide Multi-Residue Method Setup . Extraction Acetonitrile, Ethyl Mostly replaced acetate, Methanol... by QuEChERS today . Clean-up GPC, SPE, LLE, LC . Determination GC, LC, GC-MS, LC-MS, Thermo Scientific™ QuEChERS™ GC-MS/MS, LC-MS/MS... method 3 Simplified Extraction Procedure Applied 10 g of sample is weighed into Quechers extraction tube + 20 mL of water + 10 mL of ACN shaking 10 min Centrifugation 5 min @ 5000 rpm Injection to LC-HRAM 4 Consumables Used Consumables/Chemicals Part Number Acetonitrile A/0638/17 QuEChERS extraction tube, 50 mL, 250 pack 60105-216 QuEChERS pouches, 50 pack 60105-344 Apparatus/Columns Part Number Horizontal shaker 1069-3391 Horizontal shaker plate 1053-0102 Thermo Scientific™ Barnstead™ EASYpure™II water 3125753 Thermo Scientific™ Heraeus™ Fresco™ 17 micro centrifuge 3208590 Thermo Scientific™ Accucore™ aQ column 100x2.1, 2.6 µm 17326-102130 5 Improving QuEChERS Extraction Tips & Tricks: • Dry food (cereals/dried food, < 25 % water content): • Addition of water to enable adequate partitioning and reducing interaction of pesticides with matrix • Food containing fat/wax (avocado/oil): • After extraction step add a freezing out step and transfer supernatant to clean-up tube • More clean-up might be needed of raw extract (PSA+C18) • Food containing complex matrix (tea/spices) • Additional clean-up with GCB might be necessary (potential loss of planar structure pesticides like thiabendazole) • Acidic food (citrus): • Adjust pH (5-5.5) to increase recovery (e.g. -

Novel Approach to Fast Determination of 64 Pesticides Using of Ultra-Performance Liquid Chromatography-Tandem Mass Spectrometry
Novel approach to fast determination of 64 pesticides using of ultra-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS) Tomas Kovalczuk, Ondrej Lacina, Martin Jech, Jan Poustka, Jana Hajslova To cite this version: Tomas Kovalczuk, Ondrej Lacina, Martin Jech, Jan Poustka, Jana Hajslova. Novel approach to fast determination of 64 pesticides using of ultra-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS). Food Additives and Contaminants, 2008, 25 (04), pp.444-457. 10.1080/02652030701570156. hal-00577414 HAL Id: hal-00577414 https://hal.archives-ouvertes.fr/hal-00577414 Submitted on 17 Mar 2011 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Food Additives and Contaminants For Peer Review Only Novel approach to fast determination of 64 pesticides using of ultra-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS) Journal: Food Additives and Contaminants Manuscript ID: TFAC-2007-065.R1 Manuscript Type: Original Research Paper Date Submitted by the 07-Jul-2007 Author: Complete List of Authors: -

COMBINED LIST of Particularly Hazardous Substances
COMBINED LIST of Particularly Hazardous Substances revised 2/4/2021 IARC list 1 are Carcinogenic to humans list compiled by Hector Acuna, UCSB IARC list Group 2A Probably carcinogenic to humans IARC list Group 2B Possibly carcinogenic to humans If any of the chemicals listed below are used in your research then complete a Standard Operating Procedure (SOP) for the product as described in the Chemical Hygiene Plan. Prop 65 known to cause cancer or reproductive toxicity Material(s) not on the list does not preclude one from completing an SOP. Other extremely toxic chemicals KNOWN Carcinogens from National Toxicology Program (NTP) or other high hazards will require the development of an SOP. Red= added in 2020 or status change Reasonably Anticipated NTP EPA Haz list COMBINED LIST of Particularly Hazardous Substances CAS Source from where the material is listed. 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide Acutely Toxic Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- Acutely Toxic 1-(2-Chloroethyl)-3-(4-methylcyclohexyl)-1-nitrosourea (Methyl-CCNU) Prop 65 KNOWN Carcinogens NTP 1-(2-Chloroethyl)-3-cyclohexyl-1-nitrosourea (CCNU) IARC list Group 2A Reasonably Anticipated NTP 1-(2-Chloroethyl)-3-cyclohexyl-1-nitrosourea (CCNU) (Lomustine) Prop 65 1-(o-Chlorophenyl)thiourea Acutely Toxic 1,1,1,2-Tetrachloroethane IARC list Group 2B 1,1,2,2-Tetrachloroethane Prop 65 IARC list Group 2B 1,1-Dichloro-2,2-bis(p -chloropheny)ethylene (DDE) Prop 65 1,1-Dichloroethane -

Development of a CEN Standardised Method for Liquid Chromatography Coupled to Accurate Mass Spectrometry
Development of a CEN standardised method for liquid chromatography coupled to accurate mass spectrometry CONTENTS 1. Aim and scope ................................................................................................................. 2 2. Short description ................................................................................................................ 2 3. Apparatus and consumables ......................................................................................... 2 4. Chemicals ........................................................................................................................... 2 5. Procedure ........................................................................................................................... 3 5.1. Sample preparation ................................................................................................... 3 5.2. Recovery experiments for method validation ...................................................... 3 5.3. Extraction method ...................................................................................................... 3 5.4. Measurement .............................................................................................................. 3 5.5. Instrumentation and analytical conditions ............................................................ 4 5.5.1. Dionex Ultimate 3000 .......................................................................................... 4 5.5.2. QExactive Focus HESI source parameters ..................................................... -
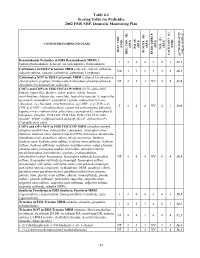
2002 NRP Section 6, Tables 6.1 Through
Table 6.1 Scoring Table for Pesticides 2002 FSIS NRP, Domestic Monitoring Plan } +1 0.05] COMPOUND/COMPOUND CLASS * ) (EPA) (EPA) (EPA) (EPA) (EPA) (FSIS) (FSIS) PSI (P) TOX.(T) L-1 HIST. VIOL. BIOCON. (B) {[( (2*R+P+B)/4]*T} REG. CON. (R) * ENDO. DISRUP. LACK INFO. (L) LACK INFO. {[ Benzimidazole Pesticides in FSIS Benzimidazole MRM (5- 131434312.1 hydroxythiabendazole, benomyl (as carbendazim), thiabendazole) Carbamates in FSIS Carbamate MRM (aldicarb, aldicarb sulfoxide, NA44234416.1 aldicarb sulfone, carbaryl, carbofuran, carbofuran 3-hydroxy) Carbamates NOT in FSIS Carbamate MRM (carbaryl 5,6-dihydroxy, chlorpropham, propham, thiobencarb, 4-chlorobenzylmethylsulfone,4- NT 4 1 3 NV 4 4 13.8 chlorobenzylmethylsulfone sulfoxide) CHC's and COP's in FSIS CHC/COP MRM (HCB, alpha-BHC, lindane, heptachlor, dieldrin, aldrin, endrin, ronnel, linuron, oxychlordane, chlorpyrifos, nonachlor, heptachlor epoxide A, heptachlor epoxide B, endosulfan I, endosulfan I sulfate, endosulfan II, trans- chlordane, cis-chlordane, chlorfenvinphos, p,p'-DDE, p, p'-TDE, o,p'- 3444NV4116.0 DDT, p,p'-DDT, carbophenothion, captan, tetrachlorvinphos [stirofos], kepone, mirex, methoxychlor, phosalone, coumaphos-O, coumaphos-S, toxaphene, famphur, PCB 1242, PCB 1248, PCB 1254, PCB 1260, dicofol*, PBBs*, polybrominated diphenyl ethers*, deltamethrin*) (*identification only) COP's and OP's NOT in FSIS CHC/COP MRM (azinphos-methyl, azinphos-methyl oxon, chlorpyrifos, coumaphos, coumaphos oxon, diazinon, diazinon oxon, diazinon met G-27550, dichlorvos, dimethoate, dimethoate -

Chemical Name Federal P Code CAS Registry Number Acutely
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extremely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extremely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extremely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extremely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extremely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extremely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extremely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extremely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extremely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -

Assessment of the Endocrine-Disrupting Effects Of
Food and Chemical Toxicology 133 (2019) 110759 Contents lists available at ScienceDirect Food and Chemical Toxicology journal homepage: www.elsevier.com/locate/foodchemtox Assessment of the endocrine-disrupting effects of organophosphorus T pesticide triazophos and its metabolites on endocrine hormones biosynthesis, transport and receptor binding in silico ⁎ Fang-Wei Yanga, Yi-Xuan Lia, Fa-Zheng Rena,b, Jie Luoa,c, Guo-Fang Panga,d, a Beijing Advanced Innovation Center for Food Nutrition and Human Health, College of Food Science and Nutritional Engineering, China Agricultural University, Beijing, 100083, China b Key Laboratory of Functional Dairy, Co-constructed by Ministry of Education and Beijing Government, Beijing Laboratory of Food Quality and Safety, China Agricultural University, Beijing, 100083, China c College of Food Science and Technology, Hunan Agricultural University, Changsha, 410114, China d Chinese Academy of Inspection and Quarantine, Beijing, 100176, China ARTICLE INFO ABSTRACT Keywords: Triazophos (TAP) was a widely used organophosphorus insecticide in developing countries. TAP could produce Triazophos specific metabolites triazophos-oxon (TAPO) and 1-phenyl-3-hydroxy-1,2,4-triazole (PHT) and non-specific Metabolites metabolites diethylthiophosphate (DETP) and diethylphosphate (DEP). The objective of this study involved Endocrine disruption computational approaches to discover potential mechanisms of molecular interaction of TAP and its major Hormones metabolites with endocrine hormone-related proteins using molecular docking in silico. We found that TAP, Molecular docking TAPO and DEP showed high binding affinity with more proteins and enzymes than PHT and DETP. TAPmight interfere with the endocrine function of the adrenal gland, and TAP might also bind strongly with glucocorticoid receptors and thyroid hormone receptors. -

Application of Dried Blood Spots for the Determination of Organophosphorus Pesticides by GC-MS/MS
UNIVERSIDADE DA BEIRA INTERIOR Ciências Application of dried blood spots for the determination of organophosphorus pesticides by GC-MS/MS Sofia Pires Seixo Soares Dissertação para obtenção do Grau de Mestre em Bioquímica (2º ciclo de estudos) Orientador: Doutor Mário Jorge Dinis Barroso Co-orientador: Professora Doutora Maria Eugenia Gallardo Alba Covilhã, junho de 2018 Agradecimentos A realização de uma dissertação de mestrado é o culminar de muitas horas de estudo, reflexão e trabalho pessoal, mas representa também uma etapa que não seria possível sem o contributo fundamental de várias pessoas. À Doutora Eugénia Gallardo e ao Doutor Mário Barroso, como excelentes mentores que são, quero agradecer toda a orientação, motivação, incentivo e confiança. Toda a ajuda e partilha de experiência e conhecimento durante todo este percurso. Ao Tiago Rosado, pela imprescindível ajuda e disponibilidade constante, bem como pela amizade. À Joana Gonçalves e ao Ângelo Luís um agradecimento especial pela amizade e carinho demonstrado ao longo deste ano. A todos, quero agradecer pelo bom ambiente proporcionado. Aos meus pais Ana Soares e José Soares, a quem dedico este feito, por me darem a possibilidade de concretizar conquistas como esta, pelo apoio incondicional, por sempre terem acreditado em mim e por fazerem parte deste momento. Aos meus avós e restante família, por todo o apoio e preocupação. Um agradecimento particular à Inês Ramos, à Rita Marques, à Maria Cunha e ao Pedro Batista pelo companheirismo, amizade e apoio de há tantos anos. E por último, mas nunca menos importante, um enorme obrigada ao meu namorado António Fernandes por toda a paciência, pelo incansável apoio, carinho e ajuda. -

Eupt-Fv17- Target Pesticide List
EUPT-FV17- TARGET PESTICIDE LIST MRRL Pesticide (mg/Kg) 3-hydroxy-carbofuran 0.01 Acephate 0.01 Acetamiprid 0.01 Acrinathrin 0.01 Aldicarb 0.01 Aldicarb Sulfone 0.01 Aldicarb Sulfoxide 0.01 Azinphos-methyl 0.01 Azoxystrobin 0.01 Benfuracarb 0.01 Benomyl 0.01 Bifenthrin 0.01 Bitertanol 0.01 Boscalid 0.01 Bromopropylate 0.01 Bromuconazole 0.01 Bupirimate 0.01 Buprofezin 0.01 Cadusafos 0.006 Carbaryl 0.01 Carbendazim 0.01 Carbofuran 0.01 Carbosulfan 0.01 Chlorfenapyr 0.01 Chlorfenvinphos 0.01 Chlorobenzilate 0.01 Chlorothalonil 0.01 Chlorpropham (only parent compound) 0.01 Chlorpyrifos 0.01 Chlorpyrifos-methyl 0.01 Clofentezine (only parent compound) 0.01 Clothianidin 0.01 Cyfluthrin (cyfluthrin incl. other mixtures of constituent isomers (sum of isomers)) 0.01 Cypermethrin (cypermethrin incl. other mixtures of constituent isomers (sum of isomers)) 0.01 Cyproconazole 0.01 Cyprodinil 0.01 Deltamethrin 0.01 Demeton-S-methylsulfone 0.006 Desmethyl-pirimicarb 0.01 Diazinon 0.01 Dichlofluanid (only parent compound) 0.01 Dichlorvos 0.01 Dicloran 0.01 Dicofol 0.01 Diethofencarb 0.01 Difenoconazole 0.01 Diflubenzuron 0.01 Dimethoate 0.003 Dimethomorph 0.01 Dimethylaminosulfotoluidide (DMST) 0.01 Diniconazole 0.01 Diphenylamine 0.01 Endosulfan alpha 0.01 Endosulfan beta 0.01 Endosulfan sulfate 0.01 EPN 0.01 Epoxiconazole 0.01 Ethion 0.01 Ethirimol 0.01 Ethoprophos 0.008 Etofenprox 0.01 Fenamidone 0.01 Fenamiphos 0.01 Fenamiphos sulfone 0.01 Fenamiphos sulfoxide 0.01 Fenarimol 0.01 Fenazaquin 0.01 Fenbuconazole 0.01 Fenhexamid 0.01 Fenitrothion 0.01 -

744 Hydrolysis of Chiral Organophosphorus Compounds By
[Frontiers in Bioscience, Landmark, 26, 744-770, Jan 1, 2021] Hydrolysis of chiral organophosphorus compounds by phosphotriesterases and mammalian paraoxonase-1 Antonio Monroy-Noyola1, Damianys Almenares-Lopez2, Eugenio Vilanova Gisbert3 1Laboratorio de Neuroproteccion, Facultad de Farmacia, Universidad Autonoma del Estado de Morelos, Morelos, Mexico, 2Division de Ciencias Basicas e Ingenierias, Universidad Popular de la Chontalpa, H. Cardenas, Tabasco, Mexico, 3Instituto de Bioingenieria, Universidad Miguel Hernandez, Elche, Alicante, Spain TABLE OF CONTENTS 1. Abstract 2. Introduction 2.1. Organophosphorus compounds (OPs) and their toxicity 2.2. Metabolism and treatment of OP intoxication 2.3. Chiral OPs 3. Stereoselective hydrolysis 3.1. Stereoselective hydrolysis determines the toxicity of chiral compounds 3.2. Hydrolysis of nerve agents by PTEs 3.2.1. Hydrolysis of V-type agents 3.3. PON1, a protein restricted in its ability to hydrolyze chiral OPs 3.4. Toxicity and stereoselective hydrolysis of OPs in animal tissues 3.4.1. The calcium-dependent stereoselective activity of OPs associated with PON1 3.4.2. Stereoselective hydrolysis commercial OPs pesticides by alloforms of PON1 Q192R 3.4.3. PON1, an enzyme that stereoselectively hydrolyzes OP nerve agents 3.4.4. PON1 recombinants and stereoselective hydrolysis of OP nerve agents 3.5. The activity of PTEs in birds 4. Conclusions 5. Acknowledgments 6. References 1. ABSTRACT Some organophosphorus compounds interaction of the racemic OPs with these B- (OPs), which are used in the manufacturing of esterases (AChE and NTE) and such interactions insecticides and nerve agents, are racemic mixtures have been studied in vivo, ex vivo and in vitro, using with at least one chiral center with a phosphorus stereoselective hydrolysis by A-esterases or atom. -

Liquid and Gas Chromatographic Multi-Residue Pesticide Determination in Animal Tissues
Pestic. Sci. 1997, 49,56È64 Liquid and Gas Chromatographic Multi-residue Pesticide Determination in Animal Tissues Aurora Navas Dj az,* Angeles Garcj a Pareja & Francisco Garcj aSanchez Departamento de Qu•mica Anal•tica, Facultad de Ciencias, Universidad de Malaga, 29071-Malaga, Spain (Received 13 June 1995; revised version received 17 April 1996; accepted 16 August 1996) Abstract: A liquid chromatography multi-residue method with photometric detection has been developed. The method is applicable to the quantitative deter- mination of organochlorine (tetradifon, dicofol, chlorfenson, chlorobenzilate), organophosphorus (fenitrothion, azinphos-ethyl) and carbamate (pirimicarb) pesticides in animal tissues. The extracted residues are cleaned up by gel- permeation chromatography. A further fractionation on silica Sep-Pack car- tridges is included in the procedure. A gas chromatographic method with electron-capture detection for the analysis of the same pesticides was carried out and the results in the two cases compared. Lower detection and quantitation limits and similar recoveries of pesticides from spiked pig liver and brain samples were obtained by the LC method. Key words: liquid chromatography, gas chromatography, multi-residue pesti- cides, animal tissues 1 INTRODUCTION by various element-sensitive detectors, leading to reli- ance on mass spectrometry because of its ability to help Pest control in modern agriculture includes treatment to deÐne the structure. This has especially been true in of crops pre- and post-harvest with a variety of chemi- the case of the combination of gas chromatography cals, such as herbicides and insecticides in the pre- with mass spectrometry (GC/MS). One of the obvious harvest stage and with fungicides and rodenticides in Ðelds of application for this technique was the analysis the storage stage of the total harvest process. -

Spatial Variation in Non-Target Effects of the Insecticides Chlorpyrifos
Pesticide Science Pestic Sci 55:875±886 (1999) Spatial variation in non-target effects of the insecticides chlorpyrifos, cypermethrin and pirimicarb on Collembola in winter wheat Geoffrey K Frampton* Biodiversity and Ecology Division, School of Biological Sciences, University of Southampton, Bassett Crescent East, Southampton, SO16 7PX, UK Abstract: Contiguous winter wheat ®elds of similar cropping history and soil type were used in a study of the responses of Collembola to summer sprays of cypermethrin and pirimicarb in southern England. Chlorpyrifos was included in the study as a toxic standard. Epigeic arthropods were captured by suction sampling and crop-inhabiting species obtained by dissecting wheat ears. Eight genera of Collembola responded signi®cantly to the insecticide treatments. Collembolan abundance decreased after chlorpyrifos was applied but increased after use of cypermethrin. Negative effects of cypermethrin and pirimicarb on Collembola were not detected in this study. Effects of chlorpyrifos varied spatially as a result of faunal heterogeneity among the ®elds, despite apparent homogeneity of the site. Some species known to be susceptible to chlorpyrifos were absent from one or more of the ®elds. The implications of these ®ndings for the interpretation of non-target pesticide effects and the potential use of Collembola as bioindicators in ®eld studies with pesticides are discussed. # 1999 Society of Chemical Industry Keywords: insecticides; chlorpyrifos; cypermethrin; pirimicarb; Collembola; side-effects; bioindicators