1 © <2021>. This Manuscript Version Is Made Available Under the CC-BY
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Status of Insectivorous Plants in Northeast India
Technical Refereed Contribution Status of insectivorous plants in northeast India Praveen Kumar Verma • Shifting Cultivation Division • Rain Forest Research Institute • Sotai Ali • Deovan • Post Box # 136 • Jorhat 785 001 (Assam) • India • [email protected] Jan Schlauer • Zwischenstr. 11 • 60594 Frankfurt/Main • Germany • [email protected] Krishna Kumar Rawat • CSIR-National Botanical Research Institute • Rana Pratap Marg • Lucknow -226 001 (U.P) • India Krishna Giri • Shifting Cultivation Division • Rain Forest Research Institute • Sotai Ali • Deovan • Post Box #136 • Jorhat 785 001 (Assam) • India Keywords: Biogeography, India, diversity, Red List data. Introduction There are approximately 700 identified species of carnivorous plants placed in 15 genera of nine families of dicotyledonous plants (Albert et al. 1992; Ellison & Gotellli 2001; Fleischmann 2012; Rice 2006) (Table 1). In India, a total of five genera of carnivorous plants are reported with 44 species; viz. Utricularia (38 species), Drosera (3), Nepenthes (1), Pinguicula (1), and Aldrovanda (1) (Santapau & Henry 1976; Anonymous 1988; Singh & Sanjappa 2011; Zaman et al. 2011; Kamble et al. 2012). Inter- estingly, northeastern India is the home of all five insectivorous genera, namely Nepenthes (com- monly known as tropical pitcher plant), Drosera (sundew), Utricularia (bladderwort), Aldrovanda (waterwheel plant), and Pinguicula (butterwort) with a total of 21 species. The area also hosts the “ancestral false carnivorous” plant Plumbago zelayanica, often known as murderous plant. Climate Lowland to mid-altitude areas are characterized by subtropical climate (Table 2) with maximum temperatures and maximum precipitation (monsoon) in summer, i.e., May to September (in some places the highest temperatures are reached already in April), and average temperatures usually not dropping below 0°C in winter. -

Carnivorous Plant Responses to Resource Availability
Carnivorous plant responses to resource availability: environmental interactions, morphology and biochemistry Christopher R. Hatcher A doctoral thesis submitted in partial fulfilment of requirements for the award of Doctor of Philosophy of Loughborough University November 2019 © by Christopher R. Hatcher (2019) Abstract Understanding how organisms respond to resources available in the environment is a fundamental goal of ecology. Resource availability controls ecological processes at all levels of organisation, from molecular characteristics of individuals to community and biosphere. Climate change and other anthropogenically driven factors are altering environmental resource availability, and likely affects ecology at all levels of organisation. It is critical, therefore, to understand the ecological impact of environmental variation at a range of spatial and temporal scales. Consequently, I bring physiological, ecological, biochemical and evolutionary research together to determine how plants respond to resource availability. In this thesis I have measured the effects of resource availability on phenotypic plasticity, intraspecific trait variation and metabolic responses of carnivorous sundew plants. Carnivorous plants are interesting model systems for a range of evolutionary and ecological questions because of their specific adaptations to attaining nutrients. They can, therefore, provide interesting perspectives on existing questions, in this case trait-environment interactions, plant strategies and plant responses to predicted future environmental scenarios. In a manipulative experiment, I measured the phenotypic plasticity of naturally shaded Drosera rotundifolia in response to disturbance mediated changes in light availability over successive growing seasons. Following selective disturbance, D. rotundifolia became more carnivorous by increasing the number of trichomes and trichome density. These plants derived more N from prey and flowered earlier. -

Redalyc.Overcoming DNA Extraction Problems from Carnivorous Plants
Anales del Jardín Botánico de Madrid ISSN: 0211-1322 [email protected] Consejo Superior de Investigaciones Científicas España Fleischmann, Andreas; Heubl, Günther Overcoming DNA extraction problems from carnivorous plants Anales del Jardín Botánico de Madrid, vol. 66, núm. 2, julio-diciembre, 2009, pp. 209-215 Consejo Superior de Investigaciones Científicas Madrid, España Available in: http://www.redalyc.org/articulo.oa?id=55612913003 How to cite Complete issue Scientific Information System More information about this article Network of Scientific Journals from Latin America, the Caribbean, Spain and Portugal Journal's homepage in redalyc.org Non-profit academic project, developed under the open access initiative Anales del Jardín Botánico de Madrid Vol. 66(2): 209-215 julio-diciembre 2009 ISSN: 0211-1322 doi: 10.3989/ajbm.2198 Overcoming DNA extraction problems from carnivorous plants by Andreas Fleischmann & Günther Heubl LMU Munich, Systematic Botany and Mycology, Menzinger Strasse 67, D-80638 Munich, Germany [email protected] Abstract Resumen Fleischmann, A. & Heubl, G. 2009. Overcoming DNA extraction Fleischmann, A. & Heubl, G. 2009. Superando problemas de ex- problems from carnivorous plants. Anales Jard. Bot. Madrid tracción de ADN de plantas carnívoras. Anales Jard. Bot. Madrid 66(2): 209-215. 66(2): 209-215 (en inglés). We tested previously published protocols for DNA isolation from Probamos algunos protocolos publicados previamente para el plants with high contents of polyphenols and polysaccharides aislamiento del ADN de plantas con alto contenido de polifeno- for several taxa of carnivorous plants. However, we did not get les y polisacáridos para varios táxones de plantas carnívoras. Sin satisfying results with fresh or silica dried leaf tissue obtained embargo, no conseguimos muy buenos resultados ni con tejidos from field collected or greenhouse grown plants, nor from de hojas frescas, ni con tejidos de hojas secadas en gel de sílice herbarium specimens. -

1 EPC Exhibit 138-19.2 May 15, 2015 the LIBRARY of CONGRESS
EPC Exhibit 138-19.2 May 15, 2015 THE LIBRARY OF CONGRESS Dewey Section To: Jonathan Furner, Chair Decimal Classification Editorial Policy Committee Cc: Members of the Decimal Classification Editorial Policy Committee Karl E. Debus-López, Chief, U.S. Programs, Law, and Literature Division From: Rebecca Green, Assistant Editor Dewey Decimal Classification OCLC Online Computer Library Center, Inc. Via: Michael Panzer, Editor in Chief Dewey Decimal Classification OCLC Online Computer Library Center, Inc Re: 583–584 Angiosperms [Note: In this exhibit, • “APG III” refers to the taxonomy of angiosperms in: Angiosperm Phylogeny Group. (2009). An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG III. Botanical Journal of the Linnean Society, 161 (2): 105–121. • “LAPG” refers to the numbered list of angiosperm families in Haston et al. (2009). The Linear Angiosperm Phylogeny Group (LAPG) III: a linear sequence of the families in APG III. Botanical Journal of the Linnean Society 161 (2), 128–131.] This exhibit is the culmination of work over several years, following from these previous exhibits: • EPC Exhibit 135-17.1 Angiosperms, a request from Magdalena Svanberg that we (a) give consideration to the Angiosperm Phylogeny Group’s 2009 classification for flowering plants (APG III) and (b) revise the DDC where appropriate; • EPC Exhibit 135-17.1.1, 583–584 Angiosperms: Discussion paper, which explored issues and options in the possible accommodation of APG III in the DDC; • EPC Exhibit 136-19.1, a preprint of a paper presented at the 2013 SIG/CR Classification Research Workshop, which addressed issues raised during the discussion of EPC Exhibit 135- 17.1.1. -

The Function of Secondary Metabolites in Plant Carnivory 1 Christopher R
1 Review Paper: The function of secondary metabolites in plant carnivory 2 Christopher R. Hatcher*, Dr David B. Ryves, Dr Jonathan Millett 3 Geography and Environment, Loughborough University, Loughborough, LE11 3TU 4 * Corresponding author: 5 Email: [email protected] 6 1 1 ABSTRACT 2 Background: Carnivorous plants are an ideal model system for evaluating the role of 3 secondary metabolites in plant ecology and evolution. Carnivory is a striking example of 4 convergent evolution to attract, capture and digest prey for nutrients to enhance growth and 5 reproduction and has evolved independently at least ten times. Though the role of many traits 6 in plant carnivory has been well studied, the role of secondary metabolites in the carnivorous 7 habit are considerably less understood. 8 Scope: This review provides the first synthesis of research in which secondary plant 9 metabolites have been demonstrated to have a functional role in plant carnivory. From these 10 studies we identify key metabolites for plant carnivory, their functional role, and highlight 11 biochemical similarities across taxa. From this synthesis we provide new research directions 12 for integrating secondary metabolites into understanding of the ecology and evolution of 13 plant carnivory. 14 Conclusions: Carnivorous plants use secondary metabolites to facilitate prey attraction, 15 capture, digestion and assimilation. We found ~170 metabolites for which a functional role in 16 carnivory has been demonstrated. Of these, 26 compounds are present across genera that 17 independently evolved a carnivorous habit, suggesting convergent evolution. Some secondary 18 metabolites have been co-opted from other processes such as defence or pollinator attraction. -

Metabolomics Analysis Reveals Tissue-Specific Metabolite
International Journal of Molecular Sciences Article Metabolomics Analysis Reveals Tissue-Specific Metabolite Compositions in Leaf Blade and Traps of Carnivorous Nepenthes Plants 1,2, 3, 3 Alberto Dávila-Lara y , Carlos E. Rodríguez-López y , Sarah E. O’Connor and Axel Mithöfer 1,* 1 Research Group Plant Defense Physiology, Max Planck Institute for Chemical Ecology, 07745 Jena, Germany; [email protected] 2 Departamento de Biología, Universidad Nacional Autónoma de Nicaragua-León (UNAN), 21000 León, Nicaragua 3 Department of Natural Product Biosynthesis, Max Planck Institute for Chemical Ecology, 07745 Jena, Germany; [email protected] (C.E.R.-L.); [email protected] (S.E.O.) * Correspondence: [email protected] These authors contributed equally to this work. y Received: 18 May 2020; Accepted: 17 June 2020; Published: 19 June 2020 Abstract: Nepenthes is a genus of carnivorous plants that evolved a pitfall trap, the pitcher, to catch and digest insect prey to obtain additional nutrients. Each pitcher is part of the whole leaf, together with a leaf blade. These two completely different parts of the same organ were studied separately in a non-targeted metabolomics approach in Nepenthes x ventrata, a robust natural hybrid. The first aim was the analysis and profiling of small (50–1000 m/z) polar and non-polar molecules to find a characteristic metabolite pattern for the particular tissues. Second, the impact of insect feeding on the metabolome of the pitcher and leaf blade was studied. Using UPLC-ESI-qTOF and cheminformatics, about 2000 features (MS/MS events) were detected in the two tissues. -

Micropropagation of Carnivorous Plants
University of Rhode Island DigitalCommons@URI Open Access Dissertations 2003 Micropropagation of Carnivorous Plants Kenneth Stephen Uhnak University of Rhode Island Follow this and additional works at: https://digitalcommons.uri.edu/oa_diss Recommended Citation Uhnak, Kenneth Stephen, "Micropropagation of Carnivorous Plants" (2003). Open Access Dissertations. Paper 471. https://digitalcommons.uri.edu/oa_diss/471 This Dissertation is brought to you for free and open access by DigitalCommons@URI. It has been accepted for inclusion in Open Access Dissertations by an authorized administrator of DigitalCommons@URI. For more information, please contact [email protected]. MICROPROPAGATION OF CARNIVOROUS PLANTS BY KENNETH STEPHEN UHNAK A DISSERTATION IN PARTIAL FULFILLMENT OF THE REQUIREMENT FOR THE DEGREE OF DOCTOR OF PHILOSOPHY IN BIOLOGICAL SCIENCES • UNIVERSITY OF RHODE ISLAND 2003 DOCTOR OF PHILOSOPHY DISSERTATION OF KENNETH S. UHNAK APPROVED: Dissertation Committee Major Pro fes so r_ ___,#"=<f----'--:;_:__;;~----l'-----''---"'--'-------,-..,.._- UNIVERSITY OF RHODE ISLAND 2003 ABSTRACT A simple, efficient system for seed surface disinfestation and in vitro germination was developed for the carnivorous pitcher plants Darlingtonia californica and Sarracenia leucophylla. Of the disinfectants tested, hydrogen peroxide or 10% Clorox® were most effective for disinfesting seeds of D. californica, while concentrated sulfuric acid worked best for S. leucophylla. Differences in the effectiveness of sterilants were associated by differences in seed coat morphology. Seeds of D. californica imbibed at 4-7°C in sterile deionized water with surfactant and gibberellin germinated earlier than seeds without exposure to gibberellin. Unimbibed seeds of S. leucophylla germinated rapidly in sterile water after treatment in concentrated sulfuric acid. Scanning electron microscopy of D. -

Progress and Problems in the Assessment of Flower Morphology In
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by RERO DOC Digital Library Plant Syst Evol (2012) 298:257–276 DOI 10.1007/s00606-011-0576-2 REVIEW Progress and problems in the assessment of flower morphology in higher-level systematics Peter K. Endress • Merran L. Matthews Received: 1 September 2011 / Accepted: 21 November 2011 / Published online: 7 January 2012 Ó Springer-Verlag 2012 Abstract Floral features used for characterization of Introduction higher-level angiosperm taxa (families, orders, and above) are assessed following a comparison of earlier (pre- Plant species, genera, families, orders, and even higher cladistic/premolecular) and current classifications. Cron- categories have long been characterized by structural fea- quist (An integrated system of classification of flowering tures, mainly by floral morphology. Certain features have plants. Columbia University Press, New York, 1981) and generally been regarded as of special value to characterize APG (Angiosperm Phylogeny Group) (Bot J Linn Soc higher-level taxa (families and above) in traditional clas- 161:105–121, 2009) were mainly used as the basis for this sifications, with the assumption that they are relatively comparison. Although current circumscriptions of taxo- stable. Earlier, classifications were developed whereby nomic groups (clades) are largely based on molecular larger primary groups were formed based upon shared markers, it is also important to morphologically charac- structural similarity. These groups then constituted the terize these new groups, as, in many cases, they are com- nuclei around which other groups were assembled by pletely novel assemblages, especially at the level of orders concatenation according to their least dissimilarity. -
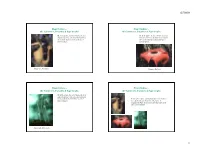
Plant Oddities - ! Plant Oddities - ! the Carnivores, Parasites & Saprotrophs ! the Carnivores, Parasites & Saprotrophs !
6/29/09! Plant Oddities - ! Plant Oddities - ! the Carnivores, Parasites & Saprotrophs ! the Carnivores, Parasites & Saprotrophs ! Of all the plants, the most bizarre, the least Of all the plants, the most bizarre, the least understood, but yet the most interesting are understood, but yet the most interesting are those plants that have unusual modes of those plants that have unusual modes of nutrient uptake.! nutrient uptake.! Carnivore: Nepenthes! Parasite: Rafflesia! Plant Oddities - ! Plant Oddities - ! the Carnivores, Parasites & Saprotrophs ! the Carnivores, Parasites & Saprotrophs ! Of all the plants, the most bizarre, the least understood, but yet the most interesting are those plants that have unusual modes of At the state, federal, and international levels, many of nutrient uptake.! these plants are extremely rare, threatened, or endangered. Many are protected internationally from collection by CITES.! Saprotroph: Monotropa! 1! 6/29/09! Plant Oddities - ! Plant Oddities - ! the Carnivores, Parasites & Saprotrophs ! the Carnivores, Parasites & Saprotrophs ! Three aspects of these plants present problems. ! Two factors for this systematic confusion! 1. The biology of how they trap animals, recognize and penetrate other plants, and how they interact with fungi is • !the specialized roles often involve reductions in both often little understood.! vegetative and floral features! 2. The basic anatomical and morphological structures of both • !often unrelated members of these groups converge unto the vegetative and floral parts of these -

DDC) Stemming from the Adoption of the APG (Angiosperm Phylogeny Group) III Classification As the Basis for the DDC’S Treatment of Flowering Plants
This PDF documents proposed changes throughout the Dewey Decimal Classification (DDC) stemming from the adoption of the APG (Angiosperm Phylogeny Group) III classification as the basis for the DDC’s treatment of flowering plants. We request comment from any interested party, to be sent to Rebecca Green ([email protected]) by 31 January 2016. Please include “Angiosperm review comments” in your subject line. -------------------------------------------------------------- Why is the DDC adopting a new basis for classifying angiosperms (flowering plants)? During the latter half of the 20th century, biological classification turned from establishing taxa predominantly on the basis of morphological similarities to establishing taxa predominantly on the basis of shared ancestry / shared derived characters, with biological taxonomies mirroring evolutionary relationships. Phylogenetic analysis typically underlies modern evolutionary classifications, but has resulted in the development of many competing classifications. Within the domain of flowering plants, different classification systems have been favored in different countries. The Angiosperm Phylogeny Group, a global consortium of botanists, has addressed this issue by developing a “consensus” classification that is monophyletic (i.e., its taxa include all but only the descendants of a common ancestor). Now in its third version, the APG III classification is considered relatively stable and useful for both research and practice (e.g., for organizing plants in herbaria). The development for flowering plants presented here is the culmination of DDC editorial work over a span of several years. An early version revised 583–584 to make the schedule compatible with the APG III classification, while trying to minimize relocations and using see references to establish the APG III logical hierarchy. -

Flora of the Guianas Project
Leiden, October 2012 The Flora of the Guianas is a co-operative programme of: Botanischer Garten und Botanisches Museum Berlin-Dahlem, Berlin; Institut de Recherche pour le Développement, IRD, Centre de Cayenne, Cayenne; Department of Biology, University of Guyana, Georgetown; Herbarium, Royal Botanic Gardens, Kew; New York Botanical Garden, New York; Nationaal Herbarium Suriname, Paramaribo; Muséum National d’Histoire Naturelle, Paris; Nationaal Herbarium Nederland, Utrecht University branch, Utrecht, and Department of Botany, Smithsonian Institution, Washington, D.C. For further information see the website: http://www.nationaalherbarium.nl/FoGWebsite/index.html Published on June 2014 Flora of the Guianas Newsletter no 18. Nationaal Herbarium Nederland, Naturalis Biodiversity Center, Einsteiweg 2, 2333 CC, Leiden, The Netherlands 2 CONTENTS In Memorian M.F.Prévost, J. Florschutz 1. MEETING PROGRAM ...................................................................................................... 6 2. MINUTES OF THE ADVISORY BOARD MEETING .............................................................. 7 2.1. Opening and report on previous meeting in Washigton .............................................. 7 2.2. Board personnel changes ............................................................................................ 7 2.3. Memorandum of Understanding ................................................................................. 7 2.4. Report by the executive editor ................................................................................... -

Year Sem. Subject Code Title of the Paper Hours/ Week 2018 -2019 Onwards III 18MBO31C PAPER VII PLANT SYSTEMATICS, RESOURCES A
Year Sem. Subject Code Title of the paper Hours/ Week III 18MBO31C PAPER VII PLANT SYSTEMATICS, RESOURCES 7 2018 -2019 AND ETHNOBOTANY onwards Objectives: 1. To acquire the fundamental values of plant systematics 2. To know about the basic concepts and principles of plant systematics 3. To establish a suitable method for correct identification and adequate characterization of plants 4. To aware of the importance of taxonomic relationships in plant systematic studies 5. To understand the utility of different plant species 6. To have a first- hand knowledge on Economic Botany and Ethnobotany Unit – I History of classification; Systems of classification: Bentham and Hooker and Cronquist; Angiosperm Phylogeny Group 2011; International Code for Botanical Nomenclature; Typification, Valid publication, Citation, Retention choice and Rejection of names; Priority. Unit –II Plant molecular systematics; Chemotaxonomy and Numerical taxonomy; Taxonomic evidences from Morphology, Anatomy, Embryology, Palynology and Cytology; Concepts of Taxa and Taxonomic hierarchy; Construction and uses of different types of key for plant identification (indented and bracket keys); Basic concepts of Flora, Revisions, Monographs, Herbaria and Data information system; Botanical Gardens. Unit – III Comparative and detailed study of the following families: Nymphaceae, Capparidaceae Polygalaceae, Portulacaceae, Zygophyllaceae, Rhamnaceae, Sapindaceae, Combretaceae, Passiflorae, Ebenaceae, Ficodeae, Rubiaceae, Oleaceae and Boraginaceae. Unit – IV Comparative and detailed