Increased Expression of HIST1H1D in Esophageal Carcinoma Predicts Poor Survival: a Study Base on TCGA Database
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Download Download
Supplementary Figure S1. Results of flow cytometry analysis, performed to estimate CD34 positivity, after immunomagnetic separation in two different experiments. As monoclonal antibody for labeling the sample, the fluorescein isothiocyanate (FITC)- conjugated mouse anti-human CD34 MoAb (Mylteni) was used. Briefly, cell samples were incubated in the presence of the indicated MoAbs, at the proper dilution, in PBS containing 5% FCS and 1% Fc receptor (FcR) blocking reagent (Miltenyi) for 30 min at 4 C. Cells were then washed twice, resuspended with PBS and analyzed by a Coulter Epics XL (Coulter Electronics Inc., Hialeah, FL, USA) flow cytometer. only use Non-commercial 1 Supplementary Table S1. Complete list of the datasets used in this study and their sources. GEO Total samples Geo selected GEO accession of used Platform Reference series in series samples samples GSM142565 GSM142566 GSM142567 GSM142568 GSE6146 HG-U133A 14 8 - GSM142569 GSM142571 GSM142572 GSM142574 GSM51391 GSM51392 GSE2666 HG-U133A 36 4 1 GSM51393 GSM51394 only GSM321583 GSE12803 HG-U133A 20 3 GSM321584 2 GSM321585 use Promyelocytes_1 Promyelocytes_2 Promyelocytes_3 Promyelocytes_4 HG-U133A 8 8 3 GSE64282 Promyelocytes_5 Promyelocytes_6 Promyelocytes_7 Promyelocytes_8 Non-commercial 2 Supplementary Table S2. Chromosomal regions up-regulated in CD34+ samples as identified by the LAP procedure with the two-class statistics coded in the PREDA R package and an FDR threshold of 0.5. Functional enrichment analysis has been performed using DAVID (http://david.abcc.ncifcrf.gov/) -

Supplementary Information
Supplementary Information Table S1. Gene Ontology analysis of affected biological processes/pathways/themes in H1 hESCs based on sets of statistically significant differentially expressed genes. Exposures Overrepresented Categories (Upregulation) EASE Score Negative regulation of cell differentiation 0.0049 Lipid biosynthetic process 0.015 Negative regulation of cell proliferation 0.02 5 cGy, 2 h Transcription factor binding 0.037 Regulation of apoptosis 0.038 Positive regulation of anti-apoptosis 0.048 P53 signaling pathway 4.2 × 10−10 Positive regulation of apoptosis 7.5 × 10−8 Response to DNA damage stimulus 1.5 × 10−6 Cellular response to stress 9.6 × 10−6 1 Gy, 2 h Negative regulation of cell proliferation 9.8 × 10−6 Cell cycle arrest 7.0 × 10−4 Negative regulation of cell differentiation 1.5 × 10−3 Regulation of protein kinase activity 0.011 I-kappaB kinase/NF-kappaB cascade 0.025 Metallothionein superfamily 9.9 × 10−18 Induction of apoptosis 8.8 × 10−5 DNA damage response 2.6 × 10−4 1 Gy, 16 h Positive regulation of anti-apoptosis 0.001 Positive regulation of cell death 0.005 Cellular response to stress 0.012 Exposures Overrepresented Categories (Downregulation) EASE Score Alternative splicing 0.016 1 Gy, 2 h Chromatin organization 0.020 Chromatin assembly 2.0 × 10−6 Cholesterol biosynthesis 5.1 × 10−5 Macromolecular complex assembly 9.3 × 10−4 1 Gy, 16 h PPAR signaling pathway 0.007 Hemopoietic organ development 0.033 Immune system development 0.040 S2 Table S2. Gene Ontology analysis of affected biological processes/pathways/themes in H7 based on sets of statistically significant differentially expressed genes. -
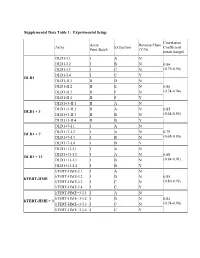
Table 3: Average Gene Expression Profiles by Chromosome
Supplemental Data Table 1: Experimental Setup Correlation Array Reverse Fluor Array Extraction Coefficient Print Batch (Y/N) mean (range) DLD1-I.1 I A N DLD1-I.2 I B N 0.86 DLD1-I.3 I C N (0.79-0.90) DLD1-I.4 I C Y DLD1 DLD1-II.1 II D N DLD1-II.2 II E N 0.86 DLD1-II.3 II F N (0.74-0.94) DLD1-II.4 II F Y DLD1+3-II.1 II A N DLD1+3-II.2 II A N 0.85 DLD1 + 3 DLD1+3-II.3 II B N (0.64-0.95) DLD1+3-II.4 II B Y DLD1+7-I.1 I A N DLD1+7-I.2 I A N 0.79 DLD1 + 7 DLD1+7-I.3 I B N (0.68-0.90) DLD1+7-I.4 I B Y DLD1+13-I.1 I A N DLD1+13-I.2 I A N 0.88 DLD1 + 13 DLD1+13-I.3 I B N (0.84-0.91) DLD1+13-I.4 I B Y hTERT-HME-I.1 I A N hTERT-HME-I.2 I B N 0.85 hTERT-HME hTERT-HME-I.3 I C N (0.80-0.92) hTERT-HME-I.4 I C Y hTERT-HME+3-I.1 I A N hTERT-HME+3-I.2 I B N 0.84 hTERT-HME + 3 hTERT-HME+3-I.3 I C N (0.74-0.90) hTERT-HME+3-I.4 I C Y Supplemental Data Table 2: Average gene expression profiles by chromosome arm DLD1 hTERT-HME Ratio.7 Ratio.1 Ratio.3 Ratio.3 Chrom. -

Application of Next-Generation Sequencing for the Genomic
Article Application of Next‐Generation Sequencing for the Genomic Characterization of Patients with Smoldering Myeloma Martina Manzoni, Valentina Marchica, Paola Storti, Bachisio Ziccheddu, Gabriella Sammarelli, Giannalisa Todaro, Francesca Pelizzoni, Simone Salerio, Laura Notarfranchi, Alessandra Pompa, Luca Baldini, Niccolò Bolli, Antonino Neri, Nicola Giuliani and Marta Lionetti Supplementary Materials Supplementary Methods Library Design, CAPP‐Seq Library Preparation, and Ultra‐Deep Next‐Generation Sequencing (NGS) The targeted resequencing gene panel included coding exons and splice sites of 56 genes that emerged as drivers from a recent analysis of whole genome/exome data in more than 800 MM patients [1] (target region: 112 kb: ACTG1, BCL7A, BHLHE41, BRAF, BTG1, CCND1, CDKN1B, CYLD, DIS3, DTX1, DUSP2, EGR1, FAM46C, FGFR3, FUBP1, HIST1H1B, HIST1H1D, HIST1H1E, HIST1H2BK, IGLL5, IRF1, IRF4, KLHL6, KMT2B, KRAS, LCE1D, LTB, MAX, NFKB2, NFKBIA, NRAS, PABPC1, PIM1, POT1, PRDM1, PRKD2, PTPN11, RASA2, RB1, RFTN1, RPL10, RPL5, RPRD1B, RPS3A, SAMHD1, SETD2, SP140, TBC1D29, TCL1A, TGDS, TP53, TRAF2, TRAF3, XBP1, ZNF462, ZNF292). Tumor gDNA (median 400 ng) was sheared through enzymatic fragmentation before library construction to obtain 150–200‐bp fragments. Targeted ultra‐deep‐next generation sequencing was performed by using the CAPP‐seq approach, as described in Newman A.M. et al., Nat. Med. 2014 [2]. The NGS libraries were constructed using the KAPA Library Preparation Kit (Kapa Biosystems, Wilmington, MA, USA) and hybrid selection was performed with the custom SeqCap EZ Choice Library (Roche NimbleGen, Madison, WI, USA), allowing for enrichment by the capture of genomic regions of interest up to 7 Mb for human resequencing studies. Multiplexed libraries were sequenced using 200‐bp paired‐end runs on an Illumina MiSeq sequencer (Illumina, Hayward, CA, USA). -

Supplemental Data.Pdf
Supplementary material -Table of content Supplementary Figures (Fig 1- Fig 6) Supplementary Tables (1-13) Lists of genes belonging to distinct biological processes identified by GREAT analyses to be significantly enriched with UBTF1/2-bound genes Supplementary Table 14 List of the common UBTF1/2 bound genes within +/- 2kb of their TSSs in NIH3T3 and HMECs. Supplementary Table 15 List of gene identified by microarray expression analysis to be differentially regulated following UBTF1/2 knockdown by siRNA Supplementary Table 16 List of UBTF1/2 binding regions overlapping with histone genes in NIH3T3 cells Supplementary Table 17 List of UBTF1/2 binding regions overlapping with histone genes in HMEC Supplementary Table 18 Sequences of short interfering RNA oligonucleotides Supplementary Table 19 qPCR primer sequences for qChIP experiments Supplementary Table 20 qPCR primer sequences for reverse transcription-qPCR Supplementary Table 21 Sequences of primers used in CHART-PCR Supplementary Methods Supplementary Fig 1. (A) ChIP-seq analysis of UBTF1/2 and Pol I (POLR1A) binding across mouse rDNA. UBTF1/2 is enriched at the enhancer and promoter regions and along the entire transcribed portions of rDNA with little if any enrichment in the intergenic spacer (IGS), which separates the rDNA repeats. This enrichment coincides with the distribution of the largest subunit of Pol I (POLR1A) across the rDNA. All sequencing reads were mapped to the published complete sequence of the mouse rDNA repeat (Gene bank accession number: BK000964). The graph represents the frequency of ribosomal sequences enriched in UBTF1/2 and Pol I-ChIPed DNA expressed as fold change over those of input genomic DNA. -

Clinical and Genetic Characterization of Basal Cell Carcinoma and Breast
Morelle et al. SpringerPlus 2014, 3:454 http://www.springerplus.com/content/3/1/454 a SpringerOpen Journal CASE STUDY Open Access Clinical and genetic characterization of basal cell carcinoma and breast cancer in a single patient Alessandra Morelle1, Rodrigo Cericatto1, Ana Cristina Victorino Krepischi2 and Itamar Romano Garcia Ruiz3* Abstract Introduction: Multiple environmental and genetic factors are involved with the development of basal cell carcinomas (BCC), as well as with breast cancers. Tumor initiation and progression are often associated with genomic instability such as aneuploidies, and gains or losses of large chromosomal segments, known as copy number alterations (CNAs). CNAs have been successfully detected using the microarray comparative genomic hybridization technique (array-CGH) at high resolution. Data thus obtained are useful to identify specific genomic aberrations, to classify tumor stages, and to stratify subgroups of patients with different prognosis and clinical behaviors. Case description: Clinical study of a 66-year-old white female identified two primary tumors, a ductal invasive grade-II carcinoma of the breast, and one nodular BCC. Germline and tumor genomic survey utilized the 180 K array-CGH analysis to investigate chromosomal alterations. Discussion and evaluation: Several chromosomal anomalies were detected in the breast tumor genome, including focal ~422 Kb 13q13.3 microdeletion. In the BCC, amplification of a chromosome 6 spanning the centromere region between the cytobands 6p23 and 6q12 was identified. Several 6p amplified genes correspond to families of histone and human leukocyte antigen genes, whereas some of the CNAs found in the breast tumor are uncommon. No germline CNA was detected in the normal skin of the patient at this technical resolution. -

Histone H1.3 (HIST1H1D) (NM 005320) Human Tagged ORF Clone Product Data
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for RC211187L3 Histone H1.3 (HIST1H1D) (NM_005320) Human Tagged ORF Clone Product data: Product Type: Expression Plasmids Product Name: Histone H1.3 (HIST1H1D) (NM_005320) Human Tagged ORF Clone Tag: Myc-DDK Symbol: H1-3 Synonyms: H1.3; H1D; H1F3; H1s-2; HIST1H1D Vector: pLenti-C-Myc-DDK-P2A-Puro (PS100092) E. coli Selection: Chloramphenicol (34 ug/mL) Cell Selection: Puromycin ORF Nucleotide The ORF insert of this clone is exactly the same as(RC211187). Sequence: Restriction Sites: SgfI-MluI Cloning Scheme: ACCN: NM_005320 ORF Size: 663 bp This product is to be used for laboratory only. Not for diagnostic or therapeutic use. View online » ©2021 OriGene Technologies, Inc., 9620 Medical Center Drive, Ste 200, Rockville, MD 20850, US 1 / 2 Histone H1.3 (HIST1H1D) (NM_005320) Human Tagged ORF Clone – RC211187L3 OTI Disclaimer: Due to the inherent nature of this plasmid, standard methods to replicate additional amounts of DNA in E. coli are highly likely to result in mutations and/or rearrangements. Therefore, OriGene does not guarantee the capability to replicate this plasmid DNA. Additional amounts of DNA can be purchased from OriGene with batch-specific, full-sequence verification at a reduced cost. Please contact our customer care team at [email protected] or by calling 301.340.3188 option 3 for pricing and delivery. The molecular sequence of this clone aligns with the gene accession number as a point of reference only. -

Usbiological Datasheet
HIST1H1D, ID (HIST1H1D, H1F3, Histone H1.3, Histone H1c, Histone H1s-2) (Azide free) (HRP) Catalog number 036581-HRP Supplier United States Biological Histones are basic nuclear proteins responsible for nucleosome structure of the chromosomal fiber in eukaryotes. Two molecules of each of the four core histones (H2A, H2B, H3, and H4) form an octamer, around which approximately 146 bp of DNA is wrapped in repeating units, called nucleosomes. The linker histone, H1, interacts with linker DNA between nucleosomes and functions in the compaction of chromatin into higher order structures. This gene is intronless and encodes a member of the histone H1 family. Transcripts from this gene lack polyA tails but instead contain a palindromic termination element. This gene is found in the large histone gene cluster on chromosome 6. Applications Suitable for use in Western Blot, Immunohistochemistry, ELISA Recommended Dilution ELISA: 1:1,000 Western Blot: 1:100-500 Immunohistochemistry: 1:10-50 Storage and Stability Store product at 4°C if to be used immediately within two weeks. For long-term storage, aliquot to avoid repeated freezing and thawing and store at -20°C. Aliquots are stable at -20°C for 12 months after receipt. Dilute required amount only prior to immediate use. Further dilutions can be made in assay buffer. Note: Sodium azide is a potent inhibitor of peroxidase and should not be added to HRP conjugates. For maximum recovery of product, centrifuge the original vial after thawing and prior to removing the cap. Note Applications are based on unconjugated antibody. Immunogen HIST1H1D antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 134-164 amino acids from the Central region of human HIST1H1D. -

TET1 Is a Tumor Suppressor of Hematopoietic Malignancy
TET1 is a tumor suppressor of hematopoietic malignancy The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation Cimmino, Luisa et al. “TET1 Is a Tumor Suppressor of Hematopoietic Malignancy.” Nature Immunology 16.6 (2015): 653– 662. As Published http://dx.doi.org/10.1038/ni.3148 Publisher Nature Publishing Group Version Author's final manuscript Citable link http://hdl.handle.net/1721.1/108307 Terms of Use Article is made available in accordance with the publisher's policy and may be subject to US copyright law. Please refer to the publisher's site for terms of use. HHS Public Access Author manuscript Author Manuscript Author ManuscriptNat Immunol Author Manuscript. Author manuscript; Author Manuscript available in PMC 2015 August 21. Published in final edited form as: Nat Immunol. 2015 June ; 16(6): 653–662. doi:10.1038/ni.3148. Tet1 is a tumor suppressor of hematopoietic malignancy Luisa Cimmino1,2,9, Meelad M. Dawlaty3,4,9, Delphine Ndiaye-Lobry1,2, Yoon Sing Yap1,2, Sofia Bakogianni1,2, Yiting Yu5, Sanchari Bhattacharyya5, Rita Shaknovich6, Huimin Geng7, Camille Lobry1,2, Jasper Mullenders1,2, Bryan King1,2, Thomas Trimarchi1,2, Beatriz Aranda-Orgilles1,2, Cynthia Liu1, Steven Shen8, Amit K. Verma5, Rudolf Jaenisch3,4, and Iannis Aifantis1,2 1Howard Hughes Medical Institute and Department of Pathology, NYU School of Medicine, New York, NY, 10016, USA 2NYU Cancer Institute and Helen L. and Martin S. Kimmel Center for Stem Cell Biology, NYU School of Medicine, New -
Product Data Sheet
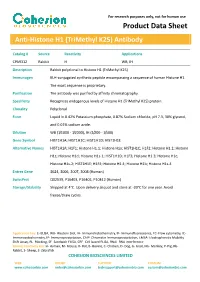
For research purposes only, not for human use Product Data Sheet Anti-Histone H1 (TriMethyl K25) Antibody Catalog # Source Reactivity Applications CPA9312 Rabbit H WB, IH Description Rabbit polyclonal to Histone H1 (TriMethyl K25) Immunogen KLH-conjugated synthetic peptide encompassing a sequence of human Histone H1. The exact sequence is proprietary. Purification The antibody was purified by affinity chromatography. Specificity Recognizes endogenous levels of Histone H1 (TriMethyl K25) protein. Clonality Polyclonal Form Liquid in 0.42% Potassium phosphate, 0.87% Sodium chloride, pH 7.3, 30% glycerol, and 0.01% sodium azide. Dilution WB (1/1000 - 1/2000), IH (1/200 - 1/500) Gene Symbol HIST1H1A; HIST1H1C; HIST1H1D; HIST1H1E Alternative Names HIST1H1A; H1F1; Histone H1.1; Histone H1a; HIST1H1C; H1F2; Histone H1.2; Histone H1c; Histone H1d; Histone H1s-1; HIST1H1D; H1F3; Histone H1.3; Histone H1c; Histone H1s-2; HIST1H1E; H1F4; Histone H1.4; Histone H1b; Histone H1s-4 Entrez Gene 3024, 3006, 3007, 3008 (Human) SwissProt Q02539, P16403, P16402, P10412 (Human) Storage/Stability Shipped at 4°C. Upon delivery aliquot and store at -20°C for one year. Avoid freeze/thaw cycles. Application key: E- ELISA, WB- Western blot, IH- Immunohistochemistry, IF- Immunofluorescence, FC- Flow cytometry, IC- Immunocytochemistry, IP- Immunoprecipitation, ChIP- Chromatin Immunoprecipitation, EMSA- Electrophoretic Mobility Shift Assay, BL- Blocking, SE- Sandwich ELISA, CBE- Cell-based ELISA, RNAi- RNA interference Species reactivity key: H- Human, M- Mouse, R- Rat, B- Bovine, C- Chicken, D- Dog, G- Goat, Mk- Monkey, P- Pig, Rb- Rabbit, S- Sheep, Z- Zebrafish COHESION BIOSCIENCES LIMITED WEB ORDER SUPPORT CUSTOM www.cohesionbio.com [email protected] [email protected] [email protected] For research purposes only, not for human use Product Data Sheet Western blot analysis of Histone H1 (TriMethyl K25) expression in Hela (A) whole cell lysates. -

Downloaded from the Cancer Genome Atlas (TCGA) Database
5507 Original Article TNFRSF12A and a new prognostic model identified from methylation combined with expression profiles to predict overall survival in hepatocellular carcinoma Yu Fang1, Lin Xiang1, La-Mei Chen1, Wei-Juan Sun1, Yu-Jia Zhai1, Yu-Chen Fan1,2, Kai Wang1,2 1Department of Hepatology, Qilu Hospital of Shandong University, Jinan, China; 2Institute of Hepatology, Shandong University, Jinan, China Contributions: (I) Conception and design: Y Fang, K Wang; (II) Administrative support: K Wang; (III) Provision of study materials or patients: Y Fang, L Xiang; (IV) Collection and assembly of data: Y Fang, L Xiang, LM Chen; (V) Data analysis and interpretation: YC Fan, WJ Sun, YJ Zhai; (VI) Manuscript writing: All authors; (VII) Final approval of manuscript: All authors. Correspondence to: Kai Wang, MD, PhD. Department of Hepatology, Qilu Hospital of Shandong University; Hepatology Institute of Shandong University; Shenzhen Research Institute of Shandong University, Wenhuaxi Road 107#, Jinan 250012, China. Email: [email protected]; [email protected]. Background: It has been proved that DNA methylation, as an epigenetic regulatory mode, plays a crucial role in the initiation, progression and invasion of hepatocellular carcinoma (HCC). However, there still are some pathways and factors that regulates the carcinogenesis of HCC remains unclear. Methods: The original datasets comparing DNA methylation, clinical information and transcriptome profiling between HCC and normal controls were downloaded from The Cancer Genome Atlas (TCGA) database. R software was used to screen for methylation-differential genes (MDGs) and methylation-driven genes. Gene-functional enrichment analysis, ConsensusPathDB pathway analysis, protein-protein interaction (PPI) network construction and survival analysis were performed; methylation-specific polymerase chain reaction (MSP) and real-time quantitative polymerase chain reaction (RT-qPCR) were used for validation. -

Transcriptomic Response of Breast Cancer Cells to Anacardic Acid David J
www.nature.com/scientificreports OPEN Transcriptomic response of breast cancer cells to anacardic acid David J. Schultz1, Abirami Krishna2, Stephany L. Vittitow2, Negin Alizadeh-Rad2, Penn Muluhngwi2, Eric C. Rouchka 3 & Carolyn M. Klinge 2 Received: 5 December 2017 Anacardic acid (AnAc), a potential dietary agent for preventing and treating breast cancer, inhibited Accepted: 10 May 2018 the proliferation of estrogen receptor α (ERα) positive MCF-7 and MDA-MB-231 triple negative Published: xx xx xxxx breast cancer cells. To characterize potential regulators of AnAc action, MCF-7 and MDA-MB-231 cells were treated for 6 h with purifed AnAc 24:1n5 congener followed by next generation transcriptomic sequencing (RNA-seq) and network analysis. We reported that AnAc-diferentially regulated miRNA transcriptomes in each cell line and now identify AnAc-regulated changes in mRNA and lncRNA transcript expression. In MCF-7 cells, 80 AnAc-responsive genes were identifed, including lncRNA MIR22HG. More AnAc-responsive genes (886) were identifed in MDA-MB-231 cells. Only six genes were commonly altered by AnAc in both cell lines: SCD, INSIG1, and TGM2 were decreased and PDK4, GPR176, and ZBT20 were increased. Modeling of AnAc-induced gene changes suggests that AnAc inhibits monounsaturated fatty acid biosynthesis in both cell lines and increases endoplasmic reticulum stress in MDA-MB-231 cells. Since modeling of downregulated genes implicated NFκB in MCF-7, we confrmed that AnAc inhibited TNFα-induced NFκB reporter activity in MCF-7 cells. These data identify new targets and pathways that may account for AnAc’s anti-proliferative and pro-apoptotic activity.