Beryllium Chloride
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Working with Hazardous Chemicals
A Publication of Reliable Methods for the Preparation of Organic Compounds Working with Hazardous Chemicals The procedures in Organic Syntheses are intended for use only by persons with proper training in experimental organic chemistry. All hazardous materials should be handled using the standard procedures for work with chemicals described in references such as "Prudent Practices in the Laboratory" (The National Academies Press, Washington, D.C., 2011; the full text can be accessed free of charge at http://www.nap.edu/catalog.php?record_id=12654). All chemical waste should be disposed of in accordance with local regulations. For general guidelines for the management of chemical waste, see Chapter 8 of Prudent Practices. In some articles in Organic Syntheses, chemical-specific hazards are highlighted in red “Caution Notes” within a procedure. It is important to recognize that the absence of a caution note does not imply that no significant hazards are associated with the chemicals involved in that procedure. Prior to performing a reaction, a thorough risk assessment should be carried out that includes a review of the potential hazards associated with each chemical and experimental operation on the scale that is planned for the procedure. Guidelines for carrying out a risk assessment and for analyzing the hazards associated with chemicals can be found in Chapter 4 of Prudent Practices. The procedures described in Organic Syntheses are provided as published and are conducted at one's own risk. Organic Syntheses, Inc., its Editors, and its Board of Directors do not warrant or guarantee the safety of individuals using these procedures and hereby disclaim any liability for any injuries or damages claimed to have resulted from or related in any way to the procedures herein. -
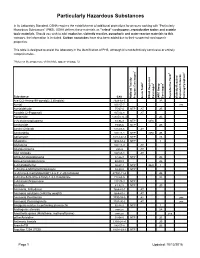
Particularly Hazardous Substances
Particularly Hazardous Substances In its Laboratory Standard, OSHA requires the establishment of additional protections for persons working with "Particularly Hazardous Substances" (PHS). OSHA defines these materials as "select" carcinogens, reproductive toxins and acutely toxic materials. Should you wish to add: explosive, violently reactive, pyrophoric and water-reactve materials to this category, the information is included. Carbon nanotubes have also been added due to their suspected carcinogenic properties. This table is designed to assist the laboratory in the identification of PHS, although it is not definitively conclusive or entirely comprehensive. *Notes on the proper use of this table appear on page 12. 1 6 5 2 3 4 Substance CAS National Toxicity National Program Carcinogen Toxin Acute Regulated OSHA Carcinogen Group IARC Carcinogen Toxin Reproductive Violently Reactive/ Explosive/Peroxide Forming/Pyrophoric A-a-C(2-Amino-9H-pyrido[2,3,b]indole) 2648-68-5 2B Acetal 105-57-7 yes Acetaldehyde 75-07-0 NTP AT 2B Acrolein (2-Propenal) 107-02-8 AT Acetamide 126850-14-4 2B 2-Acetylaminofluorene 53-96-3 NTP ORC Acrylamide 79-06-6 NTP 2B Acrylyl Chloride 814-68-6 AT Acrylonitrile 107-13-1 NTP ORC 2B Adriamycin 23214-92-8 NTP 2A Aflatoxins 1402-68-2 NTP 1 Allylamine 107-11-9 AT Alkylaluminums varies AT Allyl Chloride 107-05-1 AT ortho-Aminoazotoluene 97-56-3 NTP 2B para-aminoazobenzene 60-09-3 2B 4-Aminobiphenyl 92-67-1 NTP ORC 1 1-Amino-2-Methylanthraquinone 82-28-0 NTP (2-Amino-6-methyldipyrido[1,2-a:3’,2’-d]imidazole) 67730-11-4 2B -

Attachment 3-1 Guidance for Developing Ecological Soil
Attachment 3-1 Guidance for Developing Ecological Soil Screening Levels (Eco-SSLs) Eco-SSL Standard Operating Procedure (SOP # 1): Plant and Soil Invertebrate Literature Search and Acquisition OSWER Directive 92857-55 November 2003 This page intentionally left blank OVERVIEW Currently, there is a lack of clear guidance in setting terrestrial effect thresholds when conducting risk assessments. Without an EPA-approved, peer-reviewed, ecologically-based terrestrial effect database, the process to develop thresholds is problematic both to EPA, other federal agencies, states, and concerned private parties. Identification of published toxicity studies on invertebrates, microbial processes and plants is a key step in the derivation of benchmarks. The purpose of the Task Group 4, Standard Operating Procedure Number 1: Literature Search and Acquisition (referred to as TG4-SOP#1) is to document procedures used to identify and acquire potentially relevant toxicology literature for use in setting ecological soil screening levels. The literature search strategy is designed to locate worldwide terrestrial toxicity literature that includes the effects of chemicals of concern on terrestrial soil-dwelling invertebrates and plants. The literature acquisition process is designed to ensure timely acquisition of relevant publications. LITERATURE IDENTIFICATION Potentially relevant literature for developing ecological soil screening levels (Eco-SSLs) is identified by examining hard copies of relevant journals, bibliographies and guidance publications and through the use of a comprehensive computerized literature search strategy. These procedures are designed to locate worldwide terrestrial toxicology literature that includes the effects of specific toxic substances with an emphasis on exposure via soil. Paper-based Literature Identification The paper-based literature identification process includes the scanning of relevant review article bibliographies and key journals held in the U.S. -

Export Control Handbook for Chemicals
Export Control Handbook for Chemicals -Dual-use control list -Common Military List -Explosives precursors -Syria restrictive list -Psychotropics and narcotics precursors ARNES-NOVAU, X 2019 EUR 29879 This publication is a Technical report by the Joint Research Centre (JRC), the European Commission’s science and knowledge service. It aims to provide evidence-based scientific support to the European policymaking process. The scientific output expressed does not imply a policy position of the European Commission. Neither the European Commission nor any person acting on behalf of the Commission is responsible for the use that might be made of this publication. Contact information Xavier Arnés-Novau Joint Research Centre, Via Enrico Fermi 2749, 21027 Ispra (VA), Italy [email protected] Tel.: +39 0332-785421 Filippo Sevini Joint Research Centre, Via Enrico Fermi 2749, 21027 Ispra (VA), Italy [email protected] Tel.: +39 0332-786793 EU Science Hub https://ec.europa.eu/jrc JRC 117839 EUR 29879 Print ISBN 978-92-76-11971-5 ISSN 1018-5593 doi:10.2760/844026 PDF ISBN 978-92-76-11970-8 ISSN 1831-9424 doi:10.2760/339232 Luxembourg: Publications Office of the European Union, 2019 © European Atomic Energy Community, 2019 The reuse policy of the European Commission is implemented by Commission Decision 2011/833/EU of 12 December 2011 on the reuse of Commission documents (OJ L 330, 14.12.2011, p. 39). Reuse is authorised, provided the source of the document is acknowledged and its original meaning or message is not distorted. The European Commission shall not be liable for any consequence stemming from the reuse. -

The Elements.Pdf
A Periodic Table of the Elements at Los Alamos National Laboratory Los Alamos National Laboratory's Chemistry Division Presents Periodic Table of the Elements A Resource for Elementary, Middle School, and High School Students Click an element for more information: Group** Period 1 18 IA VIIIA 1A 8A 1 2 13 14 15 16 17 2 1 H IIA IIIA IVA VA VIAVIIA He 1.008 2A 3A 4A 5A 6A 7A 4.003 3 4 5 6 7 8 9 10 2 Li Be B C N O F Ne 6.941 9.012 10.81 12.01 14.01 16.00 19.00 20.18 11 12 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 3 Na Mg IIIB IVB VB VIB VIIB ------- VIII IB IIB Al Si P S Cl Ar 22.99 24.31 3B 4B 5B 6B 7B ------- 1B 2B 26.98 28.09 30.97 32.07 35.45 39.95 ------- 8 ------- 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 4 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 39.10 40.08 44.96 47.88 50.94 52.00 54.94 55.85 58.47 58.69 63.55 65.39 69.72 72.59 74.92 78.96 79.90 83.80 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 5 Rb Sr Y Zr NbMo Tc Ru Rh PdAgCd In Sn Sb Te I Xe 85.47 87.62 88.91 91.22 92.91 95.94 (98) 101.1 102.9 106.4 107.9 112.4 114.8 118.7 121.8 127.6 126.9 131.3 55 56 57 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86 6 Cs Ba La* Hf Ta W Re Os Ir Pt AuHg Tl Pb Bi Po At Rn 132.9 137.3 138.9 178.5 180.9 183.9 186.2 190.2 190.2 195.1 197.0 200.5 204.4 207.2 209.0 (210) (210) (222) 87 88 89 104 105 106 107 108 109 110 111 112 114 116 118 7 Fr Ra Ac~RfDb Sg Bh Hs Mt --- --- --- --- --- --- (223) (226) (227) (257) (260) (263) (262) (265) (266) () () () () () () http://pearl1.lanl.gov/periodic/ (1 of 3) [5/17/2001 4:06:20 PM] A Periodic Table of the Elements at Los Alamos National Laboratory 58 59 60 61 62 63 64 65 66 67 68 69 70 71 Lanthanide Series* Ce Pr NdPmSm Eu Gd TbDyHo Er TmYbLu 140.1 140.9 144.2 (147) 150.4 152.0 157.3 158.9 162.5 164.9 167.3 168.9 173.0 175.0 90 91 92 93 94 95 96 97 98 99 100 101 102 103 Actinide Series~ Th Pa U Np Pu AmCmBk Cf Es FmMdNo Lr 232.0 (231) (238) (237) (242) (243) (247) (247) (249) (254) (253) (256) (254) (257) ** Groups are noted by 3 notation conventions. -

Durham E-Theses
Durham E-Theses Co-ordination chemiatiy of beryllium Green, S.I.E. How to cite: Green, S.I.E. (1962) Co-ordination chemiatiy of beryllium, Durham theses, Durham University. Available at Durham E-Theses Online: http://etheses.dur.ac.uk/9108/ Use policy The full-text may be used and/or reproduced, and given to third parties in any format or medium, without prior permission or charge, for personal research or study, educational, or not-for-prot purposes provided that: • a full bibliographic reference is made to the original source • a link is made to the metadata record in Durham E-Theses • the full-text is not changed in any way The full-text must not be sold in any format or medium without the formal permission of the copyright holders. Please consult the full Durham E-Theses policy for further details. Academic Support Oce, Durham University, University Oce, Old Elvet, Durham DH1 3HP e-mail: [email protected] Tel: +44 0191 334 6107 http://etheses.dur.ac.uk Co-ordination Chemistry of Beryllium hy S. I. E. Green A thesis submitted for the Degree of Doctor of Philosophy in the University of Durham. June 1962 Acknowledgements The author would like to thank Professor 6. E. Coates for hie help and constant encouragement given during the course of this work. Thanks are also given to Sr. K. Wade and Br. 6. P. Cox for many helpful discussions, and Professor R. S. Nyholm and Dr. C. S. Pande for magnetic susceptibility measurements. The author is indebted to the Department of Scientific and Industrial Research for a Researoh Studentship. -

Distribution of This Report Is Provided in the Interest of Information Exchange, Responsibility for the Contents Resides In
Distribution of this report is provided in the interest of information exchange, Responsibility for the contents resides in the author or organization that prepared it, h- oar. T Prepared under 00 NASL-7418 by -I I NASA CR-66344 THE DEVEWPMENT OF TECHNOLOGY REQUIRED FOR THE SUCCESSFUL DEPOSITION OF ALUMINUM ALWYS AND BERYLLIUM By Ronald Guidotti and Kenneth Lui Distribution of this report is provided in the interest of information exchange, Responsibility for the contents resides in the author or organization that prepared it. Prepared under Contract No, NAS1-7418 by ELECTRO-OPTICAL SYSTEMS, INC A Xerox Company for NATIONAL AERONAUTICS AND SPACE ADMINISTRATION ABSTRACT Trial syntheses of beryllium Grignards yielded insufficient quantities of material required for the successful electrodeposition of beryllium. Aluminum-alloy and hardened-aluminum plating baths were developed through the use of inorganic and organic compound additives to an aluminum plating bath (AN1 and LiAtH in diethyl 3 4 ether). The ultimate yield strength of the aluminum deposits from 2 the improved baths was in the range of 145 - 228 MN/m (21K to 33K psi). A voltammetric technique using a triangular waveform was used to scan a large number of electrolytes in uncovering the im- proved plating baths. The triangular-wave voltametric scan also was found to be a useful method in monitoring for signs of deteri- oration of the plating bath. 7248-Final iii ACKNOWLEDGMENT The authors wish to aclcnowledge the support given by Jimmie Brower in the preparation of the large number of test solutions. 7 248- F ina 1 V s CONTENTS 1. -

1462 Element of Month Beryllium
All About Elements: Beryllium 1 Ward’s All About Elements Series Fun Facts Building Real-World Connections to About… 4 the Building Blocks of Chemistry Beryllium PERIODIC TABLE OF THE ELEMENTS 1. Prior to being named beryllium, this element GROUP 1/IA 18/VIIIA 1 2 was known as glucinium, which originated H KEY He from the Greek word glykys, meaning sweet. Atomic Number 1.01 2/IIA 35 13/IIIA 14/IVA 15/VA 16/VIA 17/VIIA 4.00 3 4 5 6 7 8 9 10 Symbol It was so named due to it characteristic Li Be Br B C N O F Ne 6.94 9.01 79.90 Atomic Weight 10.81 12.01 14.01 16.00 19.00 20.18 11 12 13 14 15 16 17 18 sweet taste. Na Mg Al Si P S Cl Ar 8 9 10 22.99 24.31 3/IIIB 4/IVB 5/VB 6/VIB 7/VIIB VIIIBVIII 11/IB 12/IIB 26.98 28.09 30.97 32.07 35.45 39.95 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 Be K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 2. Emerald, morganite and aquamarine are 39.10 40.08 44.96 47.87 50.94 52.00 54.94 55.85 58.93 58.69 63.55 65.41 69.72 72.64 74.92 78.9678.96 79.90 83.80 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 precious forms of beryl. -

Block Elements?
Chapter 10 S -BLOCK ELEMENTS Question and answers carrying 1 mark 1. What are s- block elements? s-block elements are those in which the last electron enters the outermost s-orbital . As the s-orbital can accommodate only two electrons, two groups (1 & 2) belong to the s-block of the Periodic Table. 2. Name the elements present in the 1 st Group of the Periodic Table lithium, sodium, potassium, rubidium, caesium and francium. They are collectively known as the alkali metals . 3. Why I group elements are called alkali metals ? These are called so because they form hydroxides on reaction with water which are strongly alkaline in nature. 4. Name the elements present in the 2 nd Group of the Periodic Table: beryllium, magnesium, calcium, strontium, barium and radium. These elements with the exception of beryllium are commonly known as the alkaline earth metals . 5. Why II group elements are called alkaline earth metals ? These are called so because their oxides and hydroxides are alkaline in nature and these metal oxides are found in the earth’s crust. 6. What is the reason for the diagonal relationship ? Diagonal relationship is due to the similarity in ionic sizes and /or charge/radius ratio of the elements. 7. Which is smaller in size between a metal ion and its parent atom? The monovalent ions (M +) are smaller than the parent atom. 8. Which group elements show very low ionization enthalpy in the periodic table? First group elements (alkali metals) 9. How the ionization enthalpy varies in alkali metals Ionization enthalpy decrease down the group from Li to Cs. -

ROHS Annex II Dossier for Beryllium and Its Compounds. Restriction Proposal for Substances in Electrical and Electronic Equipment Under Rohs
www.oeko.de ROHS Annex II Dossier for Beryllium and its compounds. Restriction proposal for substances in electrical and electronic equipment under RoHS Report No. 5 Version 3 Substance Name: Beryllium and its compounds 25/03/2020 EC Numbers: Beryllium metal: 231-150-7 Beryllium oxide (BeO): 215-133-1 and other Beryllium compounds CAS Numbers: Beryllium metal: 7440-41-7 Beryllium oxide (BeO): 1304-56-9 and other beryllium compounds Head Office Freiburg P.O. Box 17 71 79017 Freiburg Street address Merzhauser Strasse 173 79100 Freiburg Tel. +49 761 45295-0 Office Berlin Schicklerstrasse 5-7 10179 Berlin Tel. +49 30 405085-0 Office Darmstadt Rheinstrasse 95 64295 Darmstadt Tel. +49 6151 8191-0 [email protected] www.oeko.de RoHS Annex II Dossier, Version 3 Beryllium and its compounds Table of Contents List of Figures 5 List of Tables 5 1 IDENTIFICATION, CLASSIFICATION AND LABELLING, LEGAL STATUS AND USE RESTRICTIONS 8 1.1 Identification 8 1.1.1 Name, other identifiers, and composition of the substance 9 1.1.2 Physico-chemical properties 10 1.2 Classification and labelling status 12 1.3 Legal status and use restrictions 14 1.3.1 Regulation of the substance under REACH 14 1.3.2 Occupational Exposure Limits (OEL) 14 1.3.3 Other legislative measures 15 1.3.4 Non-governmental and non-regulatory initiatives 15 2 USE IN ELECTRICAL AND ELECTRONIC EQUIPMENT 17 2.1 Function of the substance 17 2.2 Types of applications / types of materials 19 2.3 Quantities of the substance used 25 3 HUMAN HEALTH HAZARD PROFILE 29 3.1 Critical endpoint 29 3.2 Existing Guidance -

Group 2 Organometallic Chemistry
GROUP 2 ORGANOMETALLIC CHEMISTRY: AN EXCURSION INTO THE CHEMISTRY OF HIGHLY REACTIVE ORGANOBERYLLIUM COMPLEXES by ROBERT JERARD GILLIARD, JR. (Under the direction of Gregory H. Robinson) ABSTRACT The syntheses and molecular structures of N-heterocyclic carbene-stabilized derivatives 1 1 i 1 2 of BeCl2 ([L :BeCl2] (1); L : = :C{N(2, 6- Pr2C6H3)CH}2), [L :BeCl2·THF] (2), ([L :BeCl2·THF] 2 1 1 (3); L : = :C{N(2, 4, 6-Me3C6H2)CH}2, [L :BeCl2]2·dioxane (4)) and Be(BH4)2 ([L :Be(BH4)2] (5)), are described herein. The reaction of lithium borohydride with 1 afforded carbene-stabilized beryllium borohydride, (5). Notably, compound 5 is the first structurally characterized compound containing the highly reactive Be(BH4)2 monomer. Structural analysis of 5 reveals a five-coordinate beryllium atom with distorted square pyramidal geometry. To probe the reactivity of 5, it was combined with a well-known reducing agent, disodium tetracarbonylferrate [Na2Fe(CO)4·dioxane]. The addition of one equivalent of Na2Fe(CO)4·dioxane to a toluene solution of 5 yielded a dually reduced compound containing a BH2 unit, (6). The BH2 fragment is stabilized by an N-heterocyclic carbene ligand and a reduced N-heterocyclic carbene moiety. The isolation of 6 represents the first example of the “dual reduction” of both the C=C carbene backbone (hydroboration) and the C2 carbene center (hydrogenation) of an N-heterocyclic carbene ligand. In comparison to carbene-complexed beryllium chlorides, the chemistry of electron-rich silicon compounds with beryllium chloride is also presented. The reaction of carbene-stabilized 1 1 1 i disilicon, L :Si=Si:L [L : = :C{N(2, 6- Pr2C6H3)CH}2], with BeCl2 in hexane quantitatively 1 1 yields [L :Si=Si:L ]BeCl2, (7). -

Beryllium, Inorganic and Soluble Salts
Beryllium, inorganic and soluble salts Evaluation of health hazards and proposal of a health based quality criterion for drinking water Environmental Project No. 1517, 2013 Title: Author: Beryllium, inorganic and soluble salts. Elsa Nielsen Evaluation of health hazards by exposure and Krestine Greve proposal of a health based quality criterion for Ole Ladefoged drinking water John Christian Larsen Division of Toxicology and Risk Assessment. National Food Institute, Technical University of Denmark Published by: The Danish Environmental Protection Agency Strandgade 29 1401 Copenhagen K Denmark www.mst.dk/english Year: ISBN no. Authored in 2009 978-87-93026-72-8 Published in 2013 Disclaimer: When the occasion arises, the Danish Environmental Protection Agency will publish reports and papers concerning research and development projects within the environmental sector, financed by study grants provided by the Danish Environmental Protection Agency. It should be noted that such publications do not necessarily reflect the position or opinion of the Danish Environmental Protection Agency. However, publication does indicate that, in the opinion of the Danish Environmental Protection Agency, the content represents an important contribution to the debate surrounding Danish environmental policy. Sources must be acknowledged. Content CONTENT 3 PREFACE 5 1 GENERAL DESCRIPTION 6 1.1 IDENTITY AND PHYSICAL / CHEMICAL PROPERTIES 6 1.2 PRODUCTION AND USE 6 1.3 ENVIRONMENTAL OCCURRENCE AND FATE 9 1.3.1 Air 9 1.3.2 Water 10 1.3.3 Soil 10 1.3.4 Foodstuffs 11