Pressure Vs Temperature (Boiling Point)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
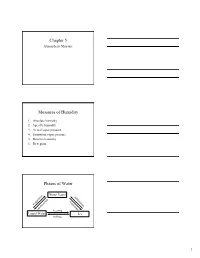
Chapter 5 Measures of Humidity Phases of Water
Chapter 5 Atmospheric Moisture Measures of Humidity 1. Absolute humidity 2. Specific humidity 3. Actual vapor pressure 4. Saturation vapor pressure 5. Relative humidity 6. Dew point Phases of Water Water Vapor n o su ti b ra li o n m p io d a a at ep t v s o io e en s n d it n io o n c freezing Liquid Water Ice melting 1 Coexistence of Water & Vapor • Even below the boiling point, some water molecules leave the liquid (evaporation). • Similarly, some water molecules from the air enter the liquid (condense). • The behavior happens over ice too (sublimation and condensation). Saturation • If we cap the air over the water, then more and more water molecules will enter the air until saturation is reached. • At saturation there is a balance between the number of water molecules leaving the liquid and entering it. • Saturation can occur over ice too. Hydrologic Cycle 2 Air Parcel • Enclose a volume of air in an imaginary thin elastic container, which we will call an air parcel. • It contains oxygen, nitrogen, water vapor, and other molecules in the air. 1. Absolute Humidity Mass of water vapor Absolute humidity = Volume of air The absolute humidity changes with the volume of the parcel, which can change with temperature or pressure. 2. Specific Humidity Mass of water vapor Specific humidity = Total mass of air The specific humidity does not change with parcel volume. 3 Specific Humidity vs. Latitude • The highest specific humidities are observed in the tropics and the lowest values in the polar regions. -

Equation of State for Benzene for Temperatures from the Melting Line up to 725 K with Pressures up to 500 Mpa†
High Temperatures-High Pressures, Vol. 41, pp. 81–97 ©2012 Old City Publishing, Inc. Reprints available directly from the publisher Published by license under the OCP Science imprint, Photocopying permitted by license only a member of the Old City Publishing Group Equation of state for benzene for temperatures from the melting line up to 725 K with pressures up to 500 MPa† MONIKA THOL ,1,2,* ERIC W. Lemm ON 2 AND ROLAND SPAN 1 1Thermodynamics, Ruhr-University Bochum, Universitaetsstrasse 150, 44801 Bochum, Germany 2National Institute of Standards and Technology, 325 Broadway, Boulder, Colorado 80305, USA Received: December 23, 2010. Accepted: April 17, 2011. An equation of state (EOS) is presented for the thermodynamic properties of benzene that is valid from the triple point temperature (278.674 K) to 725 K with pressures up to 500 MPa. The equation is expressed in terms of the Helmholtz energy as a function of temperature and density. This for- mulation can be used for the calculation of all thermodynamic properties. Comparisons to experimental data are given to establish the accuracy of the EOS. The approximate uncertainties (k = 2) of properties calculated with the new equation are 0.1% below T = 350 K and 0.2% above T = 350 K for vapor pressure and liquid density, 1% for saturated vapor density, 0.1% for density up to T = 350 K and p = 100 MPa, 0.1 – 0.5% in density above T = 350 K, 1% for the isobaric and saturated heat capaci- ties, and 0.5% in speed of sound. Deviations in the critical region are higher for all properties except vapor pressure. -

Determination of the Identity of an Unknown Liquid Group # My Name the Date My Period Partner #1 Name Partner #2 Name
Determination of the Identity of an unknown liquid Group # My Name The date My period Partner #1 name Partner #2 name Purpose: The purpose of this lab is to determine the identity of an unknown liquid by measuring its density, melting point, boiling point, and solubility in both water and alcohol, and then comparing the results to the values for known substances. Procedure: 1) Density determination Obtain a 10mL sample of the unknown liquid using a graduated cylinder Determine the mass of the 10mL sample Save the sample for further use 2) Melting point determination Set up an ice bath using a 600mL beaker Obtain a ~5mL sample of the unknown liquid in a clean dry test tube Place a thermometer in the test tube with the sample Place the test tube in the ice water bath Watch for signs of crystallization, noting the temperature of the sample when it occurs Save the sample for further use 3) Boiling point determination Set up a hot water bath using a 250mL beaker Begin heating the water in the beaker Obtain a ~10mL sample of the unknown in a clean, dry test tube Add a boiling stone to the test tube with the unknown Open the computer interface software, using a graph and digit display Place the temperature sensor in the test tube so it is in the unknown liquid Record the temperature of the sample in the test tube using the computer interface Watch for signs of boiling, noting the temperature of the unknown Dispose of the sample in the assigned waste container 4) Solubility determination Obtain two small (~1mL) samples of the unknown in two small test tubes Add an equal amount of deionized into one of the samples Add an equal amount of ethanol into the other Mix both samples thoroughly Compare the samples for solubility Dispose of the samples in the assigned waste container Observations: The unknown is a clear, colorless liquid. -

Physics, Chapter 17: the Phases of Matter
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Robert Katz Publications Research Papers in Physics and Astronomy 1-1958 Physics, Chapter 17: The Phases of Matter Henry Semat City College of New York Robert Katz University of Nebraska-Lincoln, [email protected] Follow this and additional works at: https://digitalcommons.unl.edu/physicskatz Part of the Physics Commons Semat, Henry and Katz, Robert, "Physics, Chapter 17: The Phases of Matter" (1958). Robert Katz Publications. 165. https://digitalcommons.unl.edu/physicskatz/165 This Article is brought to you for free and open access by the Research Papers in Physics and Astronomy at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Robert Katz Publications by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln. 17 The Phases of Matter 17-1 Phases of a Substance A substance which has a definite chemical composition can exist in one or more phases, such as the vapor phase, the liquid phase, or the solid phase. When two or more such phases are in equilibrium at any given temperature and pressure, there are always surfaces of separation between the two phases. In the solid phase a pure substance generally exhibits a well-defined crystal structure in which the atoms or molecules of the substance are arranged in a repetitive lattice. Many substances are known to exist in several different solid phases at different conditions of temperature and pressure. These solid phases differ in their crystal structure. Thus ice is known to have six different solid phases, while sulphur has four different solid phases. -

The Basics of Vapor-Liquid Equilibrium (Or Why the Tripoli L2 Tech Question #30 Is Wrong)
The Basics of Vapor-Liquid Equilibrium (Or why the Tripoli L2 tech question #30 is wrong) A question on the Tripoli L2 exam has the wrong answer? Yes, it does. What is this errant question? 30.Above what temperature does pressurized nitrous oxide change to a gas? a. 97°F b. 75°F c. 37°F The correct answer is: d. all of the above. The answer that is considered correct, however, is: a. 97ºF. This is the critical temperature above which N2O is neither a gas nor a liquid; it is a supercritical fluid. To understand what this means and why the correct answer is “all of the above” you have to know a little about the behavior of liquids and gases. Most people know that water boils at 212ºF. Most people also know that when you’re, for instance, boiling an egg in the mountains, you have to boil it longer to fully cook it. The reason for this has to do with the vapor pressure of water. Every liquid wants to vaporize to some extent. The measure of this tendency is known as the vapor pressure and varies as a function of temperature. When the vapor pressure of a liquid reaches the pressure above it, it boils. Until then, it evaporates until the fraction of vapor in the mixture above it is equal to the ratio of the vapor pressure to the total pressure. At that point it is said to be in equilibrium and no further liquid will evaporate. The vapor pressure of water at 212ºF is 14.696 psi – the atmospheric pressure at sea level. -

Liquid-Vapor Equilibrium in a Binary System
Liquid-Vapor Equilibria in Binary Systems1 Purpose The purpose of this experiment is to study a binary liquid-vapor equilibrium of chloroform and acetone. Measurements of liquid and vapor compositions will be made by refractometry. The data will be treated according to equilibrium thermodynamic considerations, which are developed in the theory section. Theory Consider a liquid-gas equilibrium involving more than one species. By definition, an ideal solution is one in which the vapor pressure of a particular component is proportional to the mole fraction of that component in the liquid phase over the entire range of mole fractions. Note that no distinction is made between solute and solvent. The proportionality constant is the vapor pressure of the pure material. Empirically it has been found that in very dilute solutions the vapor pressure of solvent (major component) is proportional to the mole fraction X of the solvent. The proportionality constant is the vapor pressure, po, of the pure solvent. This rule is called Raoult's law: o (1) psolvent = p solvent Xsolvent for Xsolvent = 1 For a truly ideal solution, this law should apply over the entire range of compositions. However, as Xsolvent decreases, a point will generally be reached where the vapor pressure no longer follows the ideal relationship. Similarly, if we consider the solute in an ideal solution, then Eq.(1) should be valid. Experimentally, it is generally found that for dilute real solutions the following relationship is obeyed: psolute=K Xsolute for Xsolute<< 1 (2) where K is a constant but not equal to the vapor pressure of pure solute. -

It's Just a Phase!
BASIS Lesson Plan Lesson Name: It’s Just a Phase! Grade Level Connection(s) NGSS Standards: Grade 2, Physical Science FOSS CA Edition: Grade 3 Physical Science: Matter and Energy Module *Note to teachers: Detailed standards connections can be found at the end of this lesson plan. Teaser/Overview Properties of matter are illustrated through a series of demonstrations and hands-on explorations. Students will learn to identify solids, liquids, and gases. Water will be used to demonstrate the three phases. Students will learn about sublimation through a fun experiment with dry ice (solid CO2). Next, they will compare the propensity of several liquids to evaporate. Finally, they will learn about freezing and melting while making ice cream. Lesson Objectives ● Students will be able to identify the three states of matter (solid, liquid, gas) based on the relative properties of those states. ● Students will understand how to describe the transition from one phase to another (melting, freezing, evaporation, condensation, sublimation) ● Students will learn that matter can change phase when heat/energy is added or removed Vocabulary Words ● Solid: A phase of matter that is characterized by a resistance to change in shape and volume. ● Liquid: A phase of matter that is characterized by a resistance to change in volume; a liquid takes the shape of its container ● Gas: A phase of matter that can change shape and volume ● Phase change: Transformation from one phase of matter to another ● Melting: Transformation from a solid to a liquid ● Freezing: Transformation -
ESSENTIALS of METEOROLOGY (7Th Ed.) GLOSSARY
ESSENTIALS OF METEOROLOGY (7th ed.) GLOSSARY Chapter 1 Aerosols Tiny suspended solid particles (dust, smoke, etc.) or liquid droplets that enter the atmosphere from either natural or human (anthropogenic) sources, such as the burning of fossil fuels. Sulfur-containing fossil fuels, such as coal, produce sulfate aerosols. Air density The ratio of the mass of a substance to the volume occupied by it. Air density is usually expressed as g/cm3 or kg/m3. Also See Density. Air pressure The pressure exerted by the mass of air above a given point, usually expressed in millibars (mb), inches of (atmospheric mercury (Hg) or in hectopascals (hPa). pressure) Atmosphere The envelope of gases that surround a planet and are held to it by the planet's gravitational attraction. The earth's atmosphere is mainly nitrogen and oxygen. Carbon dioxide (CO2) A colorless, odorless gas whose concentration is about 0.039 percent (390 ppm) in a volume of air near sea level. It is a selective absorber of infrared radiation and, consequently, it is important in the earth's atmospheric greenhouse effect. Solid CO2 is called dry ice. Climate The accumulation of daily and seasonal weather events over a long period of time. Front The transition zone between two distinct air masses. Hurricane A tropical cyclone having winds in excess of 64 knots (74 mi/hr). Ionosphere An electrified region of the upper atmosphere where fairly large concentrations of ions and free electrons exist. Lapse rate The rate at which an atmospheric variable (usually temperature) decreases with height. (See Environmental lapse rate.) Mesosphere The atmospheric layer between the stratosphere and the thermosphere. -

Freezing and Boiling Point Changes
Freezing Point and Boiling Point Changes for Solutions and Solids Lesson Plans and Labs By Ranald Bleakley Physics and Chemistry teacher Weedsport Jnr/Snr High School RET 2 Cornell Center for Materials Research Summer 2005 Freezing Point and Boiling Point Changes for Solutions and Solids Lesson Plans and Labs Summary: Major skills taught: • Predicting changes in freezing and boiling points of water, given type and quantity of solute added. • Solving word problems for changes to boiling points and freezing points of solutions based upon data supplied. • Application of theory to real samples in lab to collect and analyze data. Students will describe the importance to industry of the depression of melting points in solids such as metals and glasses. • Students will describe and discuss the evolution of glass making through the ages and its influence on social evolution. Appropriate grade and level: The following lesson plans and labs are intended for instruction of juniors or seniors enrolled in introductory chemistry and/or physics classes. It is most appropriately taught to students who have a basic understanding of the properties of matter and of solutions. Mathematical calculations are intentionally kept to a minimum in this unit and focus is placed on concepts. The overall intent of the unit is to clarify the relationship between changes to the composition of a mixture and the subsequent changes in the eutectic properties of that mixture. Theme: The lesson plans and labs contained in this unit seek to teach and illustrate the concepts associated with the eutectic of both liquid solutions, and glasses The following people have been of great assistance: Professor Louis Hand Professor of Physics. -

Methods for the Determination of the Normal Boiling Point of a High
Metalworking Fluids & VOC, Today and Tomorrow A Joint Symposium by SCAQMD & ILMA South Coast Air Quality Management District Diamond Bar, CA, USA March 8, 2012 Understanding & Determining the Normal Boiling Point of a High Boiling Liquid Presentation Outline • Relationship of Vapor Pressure to Temperature • Examples of VP/T Curves • Calculation of Airborne Vapor Concentration • Binary Systems • Relative Volatility as a function of Temperature • GC Data and Volatility • Everything Needs a Correlation • Conclusions Vapor Pressure Models (pure vapor over pure liquid) Correlative: • Clapeyron: Log(P) = A/T + B • Antoine: Log(P) = A/(T-C) + B • Riedel: LogP = A/T + B + Clog(T) + DTE Predictive: • ACD Group Additive Methods • Riedel: LogP = A/T + B + Clog(T) + DTE Coefficients defined, Reduced T = T/Tc • Variations: Frost-Kalkwarf-Thodos, etc. Two Parameters: Log(P) = A/T + B Vaporization as an CH OH(l) CH OH(g) activated process 3 3 G = -RTln(K) = -RTln(P) K = [CH3OH(g)]/[CH3OH(l)] G = H - TS [CH3OH(g)] = partial P ln(P) = -G/RT [CH3OH(l)] = 1 (pure liquid) ln(P) = -H/RT+ S/R K = P S/R = B ln(P) = ln(K) H/R = -A Vapor pressure Measurement: Direct versus Distillation • Direct vapor pressure measurement (e.g., isoteniscope) requires pure material while distillation based determination can employ a middle cut with a relatively high purity. Distillation allows for extrapolation and/or interpolation of data to approximate VP. • Direct vapor pressure measurement requires multiple freeze-thaw cycles to remove atmospheric gases while distillation (especially atmospheric distillation) purges atmospheric gases as part of the process. • Direct measurement OK for “volatile materials” (normal BP < 100 oC) but involved for “high boilers” (normal BP > 100 oC). -

Liquid Equilibrium Measurements in Binary Polar Systems
DIPLOMA THESIS VAPOR – LIQUID EQUILIBRIUM MEASUREMENTS IN BINARY POLAR SYSTEMS Supervisors: Univ. Prof. Dipl.-Ing. Dr. Anton Friedl Associate Prof. Epaminondas Voutsas Antonia Ilia Vienna 2016 Acknowledgements First of all I wish to thank Doctor Walter Wukovits for his guidance through the whole project, great assistance and valuable suggestions for my work. I have completed this thesis with his patience, persistence and encouragement. Secondly, I would like to thank Professor Anton Friedl for giving me the opportunity to work in TU Wien and collaborate with him and his group for this project. I am thankful for his trust from the beginning until the end and his support during all this period. Also, I wish to thank for his readiness to help and support Professor Epaminondas Voutsas, who gave me the opportunity to carry out this thesis in TU Wien, and his valuable suggestions and recommendations all along the experimental work and calculations. Additionally, I would like to thank everybody at the office and laboratory at TU Wien for their comprehension and selfless help for everything I needed. Furthermore, I wish to thank Mersiha Gozid and all the students of Chemical Engineering Summer School for their contribution of data, notices, questions and solutions during my experimental work. And finally, I would like to thank my family and friends for their endless support and for the inspiration and encouragement to pursue my goals and dreams. Abstract An experimental study was conducted in order to investigate the vapor – liquid equilibrium of binary mixtures of Ethanol – Butan-2-ol, Methanol – Ethanol, Methanol – Butan-2-ol, Ethanol – Water, Methanol – Water, Acetone – Ethanol and Acetone – Butan-2-ol at ambient pressure using the dynamic apparatus Labodest VLE 602. -

Problem Set #10 Assigned November 8, 2013 – Due Friday, November 15, 2013 Please Show All Work for Credit
Problem Set #10 Assigned November 8, 2013 – Due Friday, November 15, 2013 Please show all work for credit To Hand in 1. 1 2. –1 A least squares fit of ln P versus 1/T gives the result Hvaporization = 25.28 kJ mol . 3. Assuming constant pressure and temperature, and that the surface area of the protein is reduced by 25% due to the hydrophobic interaction: 2 G 0.25 N A 4 r Convert to per mole, ↓determine size per molecule 4 r 3 V M N 0.73mL/ g 60000g / mol (6.02 1023) 3 2 2 A r 2.52 109 m 2 23 9 2 G 0.25 N A 4 r 0.0720N / m 0.25 6.02 10 / mol (4 ) (2.52 10 m) 865kJ / mol We think this is a reasonable approach, but the value seems high 2 4. The vapor pressure of an unknown solid is approximately given by ln(P/Torr) = 22.413 – 2035(K/T), and the vapor pressure of the liquid phase of the same substance is approximately given by ln(P/Torr) = 18.352 – 1736(K/T). a. Calculate Hvaporization and Hsublimation. b. Calculate Hfusion. c. Calculate the triple point temperature and pressure. a) Calculate Hvaporization and Hsublimation. From Equation (8.16) dPln H sublimation dT RT 2 dln P d ln P dT d ln P H T 2 sublimation 11 dT dT R dd TT For this specific case H sublimation 2035 H 16.92 103 J mol –1 R sublimation Following the same proedure as above, H vaporization 1736 H 14.43 103 J mol –1 R vaporization b.